Abstract
Background and Objective
Henagliflozin, a selective inhibitor of the renal sodium glucose cotransporter-2, was developed for type 2 diabetes mellitus (T2DM). This study characterized single- and multiple-dose pharmacokinetics and pharmacodynamics of henagliflozin in Chinese patients with T2DM.
Methods
Thirty T2DM patients were randomized in a 4:1 ratio to orally receive either henagliflozin 5, 10, 20 mg/day or placebo for 10 days, except on day 2 and day 3. Pharmacokinetic and pharmacodynamic profiles were measured on day 1 and day 10.
Results
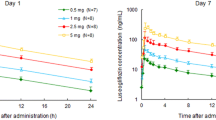
Henagliflozin exhibited dose-proportional plasma concentrations with a half-life ranging from 9.1 to 14 h. Steady-state plasma henagliflozin concentration was reached by day 7 in all active treatment groups. Henagliflozin decreased the 24-h mean plasma glucose by −0.3, −1.0 and −1.0 mmol/L with doses of 5, 10 and 20 mg on day 1, respectively. The corresponding values on day 10 were −0.8, −0.9 and −1.2 mmol/L. Twenty-four-hour urinary glucose excretion increased by 11, 65 and 82 times with doses of 5, 10 and 20 mg on day 1, respectively, with a similar trend on day 10. No treatment-related serious adverse events or discontinuations due to adverse events occurred.
Conclusions
The observed pharmacokinetic and pharmacodynamic profiles of henagliflozin support a once-daily dosing regimen in Chinese T2DM patients.


Similar content being viewed by others
References
Freeman H, Cox RD. Type-2 diabetes: a cocktail of genetic discovery. Hum Mol Genet. 2006;15(2):R202–9.
Naser KA, Gruber A, Thomson GA. The emerging pandemic of obesity and diabetes: are we doing enough to prevent a disaster? Int J Clin Pract. 2006;60(9):1093–7.
Nathan DM, Buse JB, Davidson MB, Ferrannini E, Holman RR, Sherwin R, et al. Medical management of hyperglycemia in type 2 diabetes: a consensus algorithm for the initiation and adjustment of therapy: a consensus statement of the American Diabetes Association and the European Association for the study of diabetes. Diabetes Care. 2009;32(5):193–203.
Wright EM, Hirayama BA, Loo DF. Active sugar transport in health and disease. J Intern Med. 2007;261(1):32–43.
Katsuno K, Fujimori Y, Takemura Y, Hiratochi M, Itoh F, Komatsu Y, et al. Sergliflozin, a novel selective inhibitor of low-affinity sodium glucose cotransporter (SGLT2), validates the critical role of SGLT2 in renal glucose reabsorption and modulates plasma glucose level. J Pharmacol Exp Ther. 2007;320(1):323–30.
Oku A, Ueta K, Arakawa K, Kano-Ishihara T, Matsumoto M, Adachi T, et al. Antihyperglycemic effect of T-1095 via inhibition of renal Na+-glucose cotransporters in streptozotocin-induced diabetic rats. Biol Pharm Bull. 2000;23(12):1434–7.
Devineni D, Curtin CR, Polidori D, Gutierrez MJ, Murphy J, Rusch S, et al. Pharmacokinetics and pharmacodynamics of canagliflozin, a sodium glucose co-transporter 2 inhibitor, in subjects with type 2 diabetes mellitus. J Clin Pharmacol. 2013;53(6):601–10.
US Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER). Guidance for industry. Pharmacokinetics in patients with impaired renal function-study design, data analysis, and impact on dosing and labeling. 2010; Clincial Pharmacology; Revision 1.
Kasichayanula S, Chang M, Hasegawa M, Liu X, Yamahira N, LaCreta FP, et al. Pharmacokinetics and pharmacodynamics of dapagliflozin, a novel selective inhibitor of sodium-glucose co-transporter type 2, in Japanese subjects without and with type 2 diabetes mellitus. Diabetes Obes Metab. 2011;13(4):357–65.
Komoroski B, Vachharajani N, Feng Y, Li L, Kornhauser D, Pfister M. Dapagliflozin, a novel, selective SGLT2 inhibitor, improved glycemic control over 2 weeks in patients with type 2 diabetes mellitus. Clin Pharmacol Ther. 2009;85(5):513–9.
Komoroski B, Vachharajani N, Boulton D, Kornhauser D, Geraldes M, Li L, et al. Dapagliflozin, a novel SGLT2 inhibitor, induces dose-dependent glucosuria in healthy subjects. Clin Pharmacol Ther. 2009;85(5):520–6.
Kasichayanula S, Liu X, LaCreta F, Griffen SC, Boulton DW. Clincial pharmacokinetics and pharmacodynamics of dapagliflozin, a selective inhibitor of sodium-glucose co-transport type 2. Clin Pharmacokinet. 2014;53(1):17–27.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was sponsored by Jiangsu Hengrui Medicine Co., Ltd.
Conflict of interest
Haiyan Liu, Yan-Ping Liu, Ying Chen, Minquan Wang, Jia Li and Huaqiong Shen were full-time employees of Jiangsu Hengrui Medicine Co., Ltd., when the study was conducted. All other authors have no conflicts of interest.
Ethical approval
The study was conducted in accordance with Good Clinical Practice as defined by the International Conference on Harmonisation, and Chinese State Food and Drug Administration, and Declaration of Helsinki. The study protocol and all amendments were reviewed and approved by the Clinical Research Ethics Committee of Chengdu Military General Hospital and Xijing Hospital.
Informed consent
All subjects were informed of the nature and purpose of the study, and gave written consent to participate before any screening procedures.
Rights and permissions
About this article
Cite this article
Yong, X., Wen, A., Liu, X. et al. Pharmacokinetics and Pharmacodynamics of Henagliflozin, a Sodium Glucose Co-Transporter 2 Inhibitor, in Chinese Patients with Type 2 Diabetes Mellitus. Clin Drug Investig 36, 195–202 (2016). https://doi.org/10.1007/s40261-015-0366-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40261-015-0366-7