Abstract
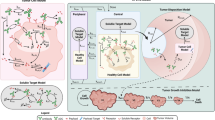
Exploiting the highly targeted nature of monoclonal antibodies to deliver selectively to tumor cells a cytotoxic payload is an attractive concept and the successful precedents of the recent past set the stage for broader applications in the future. Antibody–drug conjugates may currently hold an unprecedented potential; however, there are multiple unique challenges in their development, and the recent successes have come hand in hand with significant technologic advances in their chemistry and manufacturing. Over the years, multiple factors have been identified to affect the pharmacokinetic and pharmacodynamic properties of an antibody–drug conjugate, but many important details remain to be further investigated. These factors pertain to the target antigen, antibody, conjugate, linker, as well as the nature of the malignancy under treatment. Glembatumumab vedotin is an antibody–drug conjugate targeting glycoprotein non-metastatic B (GPNMB) expressed in multiple malignancies, including breast cancer. The expression of this protein has been associated with an aggressive malignant phenotype, invasive growth, angiogenesis, and generation of skeletal metastases. Glembatumumab vedotin is currently in early stages of clinical development in melanoma and breast cancer. Although in unselected patients with metastatic breast cancer glembatumumab vedotin was not superior to other agents, by virtue of its target being frequently and highly expressed in triple-negative breast cancer, its activity was particularly promising in this subset of patients. Results from the clinical studies in breast cancer as well as companion studies in melanoma indicate that a biomarker-informed approach is the optimal pathway for the future development of this drug.



Similar content being viewed by others
Abbreviations
- ADAM10:
-
A disintegrin and metalloproteinase 10
- ADC:
-
Antibody drug conjugate
- CDX-011 or CR011-vcMMAE:
-
Glembatumumab vedotin
- CHO:
-
Chinese Hamster Ovary
- CMC-544:
-
Inotuzumab ozogamicin
- DC-HIL (GPNMB):
-
Dendritic cell - heparin integrin ligand
- GPNMB:
-
Glycoprotein Nonmetastatic B
- GPNMB-ECD:
-
GPNMB extracellular domain
- HGFIN (GPNMB):
-
Hematopoietic growth factor inducible neurokinin-1 type
- Ig:
-
Immunoglobulin
- MITF:
-
Microphthalmia transcription factor
- MMAE:
-
Monomethylauristatin E
- MMP-3:
-
Matrix metalloproteinase 3
- sRANKL:
-
Soluble receptor activator of NF-κB ligand
- TGF-β:
-
Transforming growth factor beta
- VEGF:
-
Vascular endothelial growth factor
References
Verma S, Miles D, Gianni L, Krop IE, Welslau M, Baselga J, et al. Trastuzumab emtansine for HER2-positive advanced breast cancer. N Engl J Med. 2012;367(19):1783–91. doi:10.1056/NEJMoa1209124.
Younes A, Bartlett NL, Leonard JP, Kennedy DA, Lynch CM, Sievers EL, et al. Brentuximab vedotin (SGN-35) for relapsed CD30-positive lymphomas. N Engl J Med. 2010;363(19):1812–21. doi:10.1056/NEJMoa1002965.
Teicher BA, Chari RV. Antibody conjugate therapeutics: challenges and potential. Clin Cancer Res. 2011;17(20):6389–97. doi:10.1158/1078-0432.CCR-11-1417.
Rose AA, Annis MG, Dong Z, Pepin F, Hallett M, Park M, et al. ADAM10 releases a soluble form of the GPNMB/Osteoactivin extracellular domain with angiogenic properties. PLoS One. 2010;5(8):e12093.
Rose AA, Grosset AA, Dong Z, Russo C, Macdonald PA, Bertos NR, et al. Glycoprotein nonmetastatic B is an independent prognostic indicator of recurrence and a novel therapeutic target in breast cancer. Clin Cancer Res. 2010;16(7):2147–56.
Rose AA, Pepin F, Russo C, Abou Khalil JE, Hallett M, Siegel PM. Osteoactivin promotes breast cancer metastasis to bone. Mol Cancer Res. 2007;5(10):1001–14.
Okamoto I, Pirker C, Bilban M, Berger W, Losert D, Marosi C, et al. Seven novel and stable translocations associated with oncogenic gene expression in malignant melanoma. Neoplasia. 2005;7(4):303–11.
Tomihari M, Chung JS, Akiyoshi H, Cruz PD Jr, Ariizumi K. DC-HIL/glycoprotein Nmb promotes growth of melanoma in mice by inhibiting the activation of tumor-reactive T cells. Cancer Res. 2010;70(14):5778–87.
Tse KF, Jeffers M, Pollack VA, McCabe DA, Shadish ML, Khramtsov NV, et al. CR011, a fully human monoclonal antibody–auristatin E conjugate, for the treatment of melanoma. Clin Cancer Res. 2006;12(4):1373–82.
Williams MD, Esmaeli B, Soheili A, Simantov R, Gombos DS, Bedikian AY, et al. GPNMB expression in uveal melanoma: a potential for targeted therapy. Melanoma Res. 2010;20(3):184–90.
Onaga M, Ido A, Hasuike S, Uto H, Moriuchi A, Nagata K, et al. Osteoactivin expressed during cirrhosis development in rats fed a choline-deficient, L-amino acid-defined diet, accelerates motility of hepatoma cells. J Hepatol. 2003;39(5):779–85.
Kuan CT, Wakiya K, Dowell JM, Herndon JE 2nd, Reardon DA, Graner MW, et al. Glycoprotein nonmetastatic melanoma protein B, a potential molecular therapeutic target in patients with glioblastoma multiforme. Clin Cancer Res. 2006;12(7 Pt 1):1970–82.
Loging WT, Lal A, Siu IM, Loney TL, Wikstrand CJ, Marra MA, et al. Identifying potential tumor markers and antigens by database mining and rapid expression screening. Genome Res. 2000;10(9):1393–402.
Rich JN, Shi Q, Hjelmeland M, Cummings TJ, Kuan CT, Bigner DD, et al. Bone-related genes expressed in advanced malignancies induce invasion and metastasis in a genetically defined human cancer model. J Biol Chem. 2003;278(18):15951–7.
Li YN, Zhang L, Li XL, Cui DJ, Zheng HD, Yang SY, et al. Glycoprotein nonmetastatic B as a prognostic indicator in small cell lung cancer. APMIS. 2014;1228(2):140–6.
Gerber HP, Kung-Sutherland M, Stone I, Morris-Tilden C, Miyamoto J, McCormick R, et al. Potent antitumor activity of the anti-CD19 auristatin antibody drug conjugate hBU12-vcMMAE against rituximab-sensitive and -resistant lymphomas. Blood. 2009;113(18):4352–61.
Ingle GS, Chan P, Elliott JM, Chang WS, Koeppen H, Stephan JP, et al. High CD21 expression inhibits internalization of anti-CD19 antibodies and cytotoxicity of an anti-CD19-drug conjugate. Br J Haematol. 2008;140(1):46–58. doi:10.1111/j.1365-2141.2007.06883.x.
Advani A, Coiffier B, Czuczman MS, Dreyling M, Foran J, Gine E, et al. Safety, pharmacokinetics, and preliminary clinical activity of inotuzumab ozogamicin, a novel immunoconjugate for the treatment of B-cell non-Hodgkin’s lymphoma: results of a phase I study. J Clin Oncol. 2010;28(12):2085–93. doi:10.1200/JCO.2009.25.1900.
Dijoseph JF, Dougher MM, Armellino DC, Kalyandrug L, Kunz A, Boghaert ER, et al. CD20-specific antibody-targeted chemotherapy of non-Hodgkin’s B-cell lymphoma using calicheamicin-conjugated rituximab. Cancer Immunol. 2007;56(7):1107–17. doi:10.1007/s00262-006-0260-5.
Sjogren HO, Isaksson M, Willner D, Hellstrom I, Hellstrom KE, Trail PA. Antitumor activity of carcinoma-reactive BR96-doxorubicin conjugate against human carcinomas in athymic mice and rats and syngeneic rat carcinomas in immunocompetent rats. Cancer Res. 1997;57(20):4530–6.
Johnson DA, Baker AL, Laguzza BC, Fix DV, Gutowski MC. Antitumor activity of L/1C2-4-desacetylvinblastine-3-carboxhydrazide immunoconjugate in xenografts. Cancer Res. 1990;50(6):1790–4.
Elias DJ, Hirschowitz L, Kline LE, Kroener JF, Dillman RO, Walker LE, et al. Phase I clinical comparative study of monoclonal antibody KS1/4 and KS1/4-methotrexate immunconjugate in patients with non-small cell lung carcinoma. Cancer Res. 1990;50(13):4154–9.
Doronina SO, Toki BE, Torgov MY, Mendelsohn BA, Cerveny CG, Chace DF, et al. Development of potent monoclonal antibody auristatin conjugates for cancer therapy. Nat Biotechnol. 2003;21(7):778–84.
Francisco JA, Cerveny CG, Meyer DL, Mixan BJ, Klussman K, Chace DF, et al. cAC10-vcMMAE, an anti-CD30-monomethyl auristatin E conjugate with potent and selective antitumor activity. Blood. 2003;102(4):1458–65.
Erickson HK, Lewis Phillips GD, Leipold DD, Provenzano CA, Mai E, Johnson HA et al. The effect of different linkers on target cell catabolism and pharmacokinetics/pharmacodynamics of trastuzumab maytansinoid conjugates. Mol Cancer Ther. 2012;11(5):1133–42. doi:10.1158/1535-7163.MCT-11-0727.
Zhao RY, Wilhelm SD, Audette C, Jones G, Leece BA, Lazar AC, et al. Synthesis and evaluation of hydrophilic linkers for antibody–maytansinoid conjugates. J Med Chem. 2011;54(10):3606–23. doi:10.1021/jm2002958.
Lewis Phillips GD, Li G, Dugger DL, Crocker LM, Parsons KL, Mai E et al. Targeting HER2-positive breast cancer with trastuzumab-DM1, an antibody–cytotoxic drug conjugate. Cancer Res. 2008;68(22):9280–90. doi:10.1158/0008-5472.CAN-08-1776.
Junutula JR, Raab H, Clark S, Bhakta S, Leipold DD, Weir S, et al. Site-specific conjugation of a cytotoxic drug to an antibody improves the therapeutic index. Nat Biotechnol. 2008;26(8):925–32.
Junutula JR, Flagella KM, Graham RA, Parsons KL, Ha E, Raab H, et al. Engineered thio-trastuzumab-DM1 conjugate with an improved therapeutic index to target human epidermal growth factor receptor 2-positive breast cancer. Clin Cancer Res. 2010;16(19):4769–78. doi:10.1158/1078-0432.CCR-10-0987.
Hamblett KJ, Senter PD, Chace DF, Sun MM, Lenox J, Cerveny CG, et al. Effects of drug loading on the antitumor activity of a monoclonal antibody drug conjugate. Clin Cancer Res. 2004;10(20):7063–70. doi:10.1158/1078-0432.CCR-04-0789.
Oflazoglu E, Stone IJ, Gordon K, Wood CG, Repasky EA, Grewal IS, et al. Potent anticarcinoma activity of the humanized anti-CD70 antibody h1F6 conjugated to the tubulin inhibitor auristatin via an uncleavable linker. Clin Cancer Res. 2008;14(19):6171–80. doi:10.1158/1078-0432.CCR-08-0916.
Krop IE, Beeram M, Modi S, Jones SF, Holden SN, Yu W, et al. Phase I study of trastuzumab-DM1, an HER2 antibody–drug conjugate, given every 3 weeks to patients with HER2-positive metastatic breast cancer. J Clin Oncol. 2010;28(16):2698–704. doi:10.1200/JCO.2009.26.2071.
Vaklavas C, Forero-Torres A. Safety and efficacy of brentuximab vedotin in patients with Hodgkin lymphoma or systemic anaplastic large cell lymphoma. Ther Adv Hematol. 2012;3(4):209–25. doi:10.1177/2040620712443076.
Mendez MJ, Green LL, Corvalan JR, Jia XC, Maynard-Currie CE, Yang XD, et al. Functional transplant of megabase human immunoglobulin loci recapitulates human antibody response in mice. Nat Genet. 1997;15(2):146–56.
Yang XD, Jia XC, Corvalan JR, Wang P, Davis CG, Jakobovits A. Eradication of established tumors by a fully human monoclonal antibody to the epidermal growth factor receptor without concomitant chemotherapy. Cancer Res. 1999;59(6):1236–43.
Kito M, Itami S, Fukano Y, Yamana K, Shibui T. Construction of engineered CHO strains for high-level production of recombinant proteins. Appl Microb Biotechnol. 2002;60(4):442–8. doi:10.1007/s00253-002-1134-1.
Pettit GR, Kamano Y, Herald CL, Tuinman AA, Boettner FE, Kizu H, et al. The isolation and structure of a remarkable marine animal antineoplastic constituent: dolastatin 10. J Am Chem Soc. 1987;109:6883–5.
Madden T, Tran HT, Beck D, Huie R, Newman RA, Pusztai L, et al. Novel marine-derived anticancer agents: a phase I clinical, pharmacological, and pharmacodynamic study of dolastatin 10 (NSC 376128) in patients with advanced solid tumors. Clin Cancer Res. 2000;6(4):1293–301.
Vaishampayan U, Glode M, Du W, Kraft A, Hudes G, Wright J, et al. Phase II study of dolastatin-10 in patients with hormone-refractory metastatic prostate adenocarcinoma. Clin Cancer Res. 2000;6(11):4205–8.
Weterman MA, Ajubi N, van Dinter IM, Degen WG, van Muijen GN, Ruitter DJ, et al. nmb, a novel gene, is expressed in low-metastatic human melanoma cell lines and xenografts. Int J Cancer. 1995;60(1):73–81.
Hoashi T, Sato S, Yamaguchi Y, Passeron T, Tamaki K, Hearing VJ. Glycoprotein nonmetastatic melanoma protein b, a melanocytic cell marker, is a melanosome-specific and proteolytically released protein. FASEB J. 2010;24(5):1616–29.
Chung JS, Sato K, Dougherty II, Cruz PD Jr, Ariizumi K. DC-HIL is a negative regulator of T lymphocyte activation. Blood. 2007;109(10):4320–7.
Tomihari M, Hwang SH, Chung JS, Cruz PD Jr, Ariizumi K. Gpnmb is a melanosome-associated glycoprotein that contributes to melanocyte/keratinocyte adhesion in a RGD-dependent fashion. Exp Dermatol. 2009;18(7):586–95.
Shikano S, Bonkobara M, Zukas PK, Ariizumi K. Molecular cloning of a dendritic cell-associated transmembrane protein, DC-HIL, that promotes RGD-dependent adhesion of endothelial cells through recognition of heparan sulfate proteoglycans. J Biol Chem. 2001;276(11):8125–34.
Chung JS, Dougherty I, Cruz PD Jr, Ariizumi K. Syndecan-4 mediates the coinhibitory function of DC-HIL on T cell activation. J Immunol. 2007;179(9):5778–84.
Sheng MH, Wergedal JE, Mohan S, Lau KH. Osteoactivin is a novel osteoclastic protein and plays a key role in osteoclast differentiation and activity. FEBS Lett. 2008;582(10):1451–8.
Abdelmagid SM, Barbe MF, Arango-Hisijara I, Owen TA, Popoff SN, Safadi FF. Osteoactivin acts as downstream mediator of BMP-2 effects on osteoblast function. J Cell Physiol. 2007;210(1):26–37.
Abdelmagid SM, Barbe MF, Rico MC, Salihoglu S, Arango-Hisijara I, Selim AH, et al. Osteoactivin, an anabolic factor that regulates osteoblast differentiation and function. Exp Cell Res. 2008;314(13):2334–51.
Selim AA, Abdelmagid SM, Kanaan RA, Smock SL, Owen TA, Popoff SN, et al. Anti-osteoactivin antibody inhibits osteoblast differentiation and function in vitro. Crit Rev Eukaryot Gene Expr. 2003;13(2–4):265–75.
Ripoll VM, Meadows NA, Raggatt LJ, Chang MK, Pettit AR, Cassady AI, et al. Microphthalmia transcription factor regulates the expression of the novel osteoclast factor GPNMB. Gene. 2008;413(1–2):32–41.
Ripoll VM, Irvine KM, Ravasi T, Sweet MJ, Hume DA. Gpnmb is induced in macrophages by IFN-gamma and lipopolysaccharide and acts as a feedback regulator of proinflammatory responses. J Immunol. 2007;178(10):6557–66.
Chung JS, Bonkobara M, Tomihari M, Cruz PD Jr, Ariizumi K. The DC-HIL/syndecan-4 pathway inhibits human allogeneic T-cell responses. Eur J Immunol. 2009;39(4):965–74.
Lynch CC, Hikosaka A, Acuff HB, Martin MD, Kawai N, Singh RK, et al. MMP-7 promotes prostate cancer-induced osteolysis via the solubilization of RANKL. Cancer Cell. 2005;7(5):485–96.
clinicaltrials.gov. Study of CR011-vcMMAE to treat locally advanced or metastatic breast cancer; 2013. http://clinicaltrials.gov/ct2/show/NCT00704158.
Burris H, Saleh MN, Bendell J, Hart L, Rose AAN, Dong Z, et al. A phase I/II study of CR011-vcMMAE (CDX-011), an antibody–drug conjugate, in patients with locally advanced or metastatic breast cancer. Cancer Res. 2009;69(24 Supplement 3); Thirty-second annual CTRC-AACR San Antonio breast cancer symposium, San Antonio; 2009.
clinicaltrials.gov. A study of CDX-011 (CR011-vcMMAE) in patients with advanced GPNMB-expressing breast cancer (EMERGE); 2013. http://clinicaltrials.gov/ct2/show/NCT01156753.
Yardley DA, Weaver R, Melisko ME, Saleh MN, Arena FP, Forero A, et al. A randomized phase 2 study of the antibody–drug conjugate CDX-011 in advanced GPNMB-overexpressing breast cancer: the EMERGE study. Cancer Res. 2012;72(24, Supplement 3); Thirty-fifth annual CTRC-AACR San Antonio breast cancer symposium, San Antonio; 2012.
Hamid O, Sznol M, Pavlick AC, Kluger HM, Kim KB, Boasberg PD, et al. Frequent dosing and GPNMB expression with CDX-011 (CR011-vcMMAE), an antibody–drug conjugate (ADC), in patients with advanced melanoma. 2010 ASCO annual meeting Chicago. J Clin Oncol. 2010;28:15s (suppl; abstr 8525).
http://ctep.cancer.gov/protocoldevelopment/electronic_applications/docs/ctcaev3.pdf. Common Terminology Criteria for Adverse Events v3.0 (CTCAE). National Cancer Institute; 2006. http://ctep.cancer.gov/protocoldevelopment/electronic_applications/docs/ctcaev3.pdf. Accessed 31 Dec 2013.
Saleh MN, Bendell JC, Rose A, Siegel P, Hart LL, Sirpal S, et al. Correlation of GPNMB expression with outcome in breast cancer (BC) patients treated with the antibody–drug conjugate (ADC), CDX-011 (CR011-vcMMAE). J Clin Oncol. 2010;28:15s (suppl; abstr 1095).
Acknowledgments
CV and AF wrote the manuscript. CV is supported by a Young Investigator Award of the American Society of Clinical Oncology (ASCO). The authors have no potential conflicts of interest pertaining to this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vaklavas, C., Forero, A. Management of Metastatic Breast Cancer with Second-Generation Antibody–Drug Conjugates: Focus on Glembatumumab Vedotin (CDX-011, CR011-vcMMAE). BioDrugs 28, 253–263 (2014). https://doi.org/10.1007/s40259-014-0085-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40259-014-0085-2