Abstract
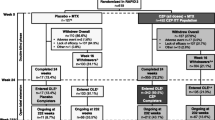
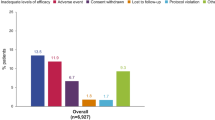
Certolizumab pegol, a pegylated Fab′ anti-tumour necrosis factor (TNF)-α agent, has shown efficacy in patients with rheumatoid arthritis (RA) unresponsive to previous treatment. In key randomised controlled trials involving patients with moderate to severe RA and an inadequate response to methotrexate or one or more disease-modifying antirheumatic drug (DMARD), the efficacy of certolizumab pegol, as monotherapy or with methotrexate, was similar to that reported in other anti-TNF clinical studies, with 60 % or fewer of patients achieving American College of Rheumatology 20 % improvement in RA. Rapid clinical response was also seen, with significant differences evident at week 1, and efficacy maintained at study end and in open-label extensions. Adding certolizumab pegol to non-biological DMARDs is efficacious in other RA populations. In the CERTAIN study, certolizumab increased remission rates, prevented disease worsening and improved work productivity and daily activity in patients with low to moderate RA. In the REALISTIC study, rapid and consistent clinical responses were observed in a diverse group of anti-TNF-eligible RA patients representing those seen in clinical practice. In the RAPID studies, rapid and sustained reduction in RA signs and symptoms, inhibition of structural joint damage progression, and improved physical function were seen with certolizumab pegol plus methotrexate versus methotrexate alone in RA patients with an incomplete response to methotrexate. Certolizumab pegol was generally well-tolerated in clinical trials, although long-term observational data are not yet available. Current data suggest that certolizumab pegol suits a ‘treat to target’ approach, providing rapid and sustained improvements in RA signs and symptoms, and beneficial effects on workplace and home productivity in patients with RA.


Similar content being viewed by others
References
Firestein GS. Evolving concepts of rheumatoid arthritis. Nature. 2003;423(6937):356–61.
Symmons D, Turner G, Webb R, Asten P, Barrett E, Lunt M, et al. The prevalence of rheumatoid arthritis in the United Kingdom: new estimates for a new century. Rheumatology. 2002;41(7):793–800.
McInnes IB, Schett G. Cytokines in the pathogenesis of rheumatoid arthritis. Nature Rev Immunol. 2007;7(6):429–42.
European Medicines Agency. Assessment report for Cimzia. London: European Medicines Agency; 2009. http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Public_assessment_report/human/001037/WC500069735.pdf. Accessed 28 Oct 2011.
UCB Pharma SA. Cimzia 200 mg solution for injection (certolizumab pegol). Summary of product characteristics. Braine l’Alleud: UCB Pharma SA; 2012. http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Product_Information/human/001037/WC500069763.pdf. Accessed 5 Sep 2012.
Fleischmann R, Vencovsky J, van Vollenhoven RF, Borenstein D, Box J, Coteur G, et al. Efficacy and safety of certolizumab pegol monotherapy every 4 weeks in patients with rheumatoid arthritis failing previous disease-modifying antirheumatic therapy: the FAST4WARD study. Ann Rheum Dis. 2009;68(6):805–11.
Keystone E, Heijde D, Mason D Jr, Landewe R, Vollenhoven RV, Combe B, et al. Certolizumab pegol plus methotrexate is significantly more effective than placebo plus methotrexate in active rheumatoid arthritis: findings of a fifty-two-week, phase III, multicenter, randomized, double-blind, placebo-controlled, parallel-group study. Arthritis Rheum. 2008;58(11):3319–29.
Smolen JS, Emery P, Ferraccioli GF, Samborski W, Berenbaum F, Davies O, et al. Efficacy and safety of certolizumab pegol after incomplete response to DMARDs in RA patients with low moderate disease activity: results from CERTAIN, a phase IIIb study [abstract no. THU0244]. Ann Rheum Dis. 2011;70(Suppl. 3):259.
Smolen J, Landewe RB, Mease P, Brzezicki J, Mason D, Luijtens K, et al. Efficacy and safety of certolizumab pegol plus methotrexate in active rheumatoid arthritis: the RAPID 2 study. A randomised controlled trial. Ann Rheum Dis. 2009;68(6):797–804.
Choy E, McKenna F, Vencovsky J, Valente R, Goel N, Vanlunen B, et al. Certolizumab pegol plus MTX administered every 4 weeks is effective in patients with RA who are partial responders to MTX. Rheumatology. 2012;51(7):1226–34.
Weinblatt ME, Fleischmann R, Huizinga TW, Emery P, Pope J, Massarotti E, et al. Efficacy and safety of certolizumab pegol in a broad population of patients with active rheumatoid arthritis: results from the REALISTIC phase IIIb study. Rheumatology. 2012;51:2204–14.
Felson DT, Anderson JJ, Boers M, Bombardier C, Furst D, Goldsmith C, et al. American College of Rheumatology. Preliminary definition of improvement in rheumatoid arthritis. Arthritis Rheum. 1995;38(6):727–35.
Aletaha D, Nell VP, Stamm T, Uffmann M, Pflugbeil S, Machold K, et al. Acute phase reactants add little to composite disease activity indices for rheumatoid arthritis: validation of a clinical activity score. Arthritis Res Ther. 2005;7(4):R796–806.
Horton S, Walsh C, Emery P. Certolizumab pegol for the treatment of rheumatoid arthritis. Expert Opin Biol Ther. 2012;12(2):235–49.
van der Heijde DM. Radiographic imaging: the ‘gold standard’ for assessment of disease progression in rheumatoid arthritis. Rheumatology. 2000;39(Suppl. 1):9–16.
Keystone EC, Curtis JR, Fleischmann RM, Furst DE, Khanna D, Smolen JS, et al. Rapid improvement in the signs and symptoms of rheumatoid arthritis following certolizumab pegol treatment predicts better longterm outcomes: post-hoc analysis of a randomized controlled trial. J Rheumatol. 2011;38(6):990–6.
Schiff M, Keystone E, Kvien TK, Curtis JR, Emery P, Luijtens K, et al. DAS28(ESR) response at week 12 is predictive of long-term disease activity in rheumatoid arthritis patients treated with certolizumab pegol. Ann Rheum Dis. 2009;2009(68):543.
Van Der Heijde D, Keystone EC, Curtis JR, Landewe RB, Schiff MH, Khanna D, et al. Timing and magnitude of initial change in disease activity score 28 predicts the likelihood of achieving low disease activity at 1 year in rheumatoid arthritis patients treated with certolizumab pegol: a post-hoc analysis of the RAPID 1 Trial. J Rheumatol. 2012;39(7):1326–33.
Strand V, Smolen JS, van Vollenhoven RF, Mease P, Burmester GR, Hiepe F, et al. Certolizumab pegol plus methotrexate provides broad relief from the burden of rheumatoid arthritis: analysis of patient-reported outcomes from the RAPID 2 trial. Ann Rheum Dis. 2011;70(6):996–1002.
Strand V, Mease P, Burmester GR, Nikai E, Coteur G, van Vollenhoven R, et al. Rapid and sustained improvements in health-related quality of life, fatigue, and other patient-reported outcomes in rheumatoid arthritis patients treated with certolizumab pegol plus methotrexate over 1 year: results from the RAPID 1 randomized controlled trial. Arthritis Res Ther. 2009;11(6):R170.
Tack BB. Self-reported fatigue in rheumatoid arthritis. A pilot study. Arthritis Care Res. 1990;3(3):154–7.
Ware JE Jr. SF-36 Health Survey Update; 2012. http://www.sf-36.org/tools/sf36.shtml. Accessed 25 Jan 2012.
Kavanaugh A, Smolen JS, Emery P, Purcaru O, Keystone E, Richard L, et al. Effect of certolizumab pegol with methotrexate on home and work place productivity and social activities in patients with active rheumatoid arthritis. Arthritis Rheum. 2009;61(11):1592–600.
Osterhaus JT, Purcaru O, Richard L. Discriminant validity, responsiveness and reliability of the rheumatoid arthritis-specific work productivity survey (WPS-RA). Arthritis Res Ther. 2009;11(3):R73.
Keystone EC, Combe B, Smolen J, Strand V, Goel N, van Vollenhoven R, et al. Sustained efficacy of certolizumab pegol added to methotrexate in the treatment of rheumatoid arthritis: 2-year results from the RAPID 1 trial. Rheumatology. 2012;51(9):1628–38.
Smolen JS, van Vollenhoven RF, Kavanaugh A, Goel N, Fichtner A, Strand V, et al. Efficacy and safety of certolimab pegol plus methotrexate in patients with rheumatoid arthritis: 3-year data from the RAPID 2 study [abstract]. Arthritis Rheum. 2010;62(Suppl. 10):1806.
Fleischmann R, Choy EH, Van Vollenhoven R, et al. Safety, efficacy and sustained improvements in household productivity and daily activities with certolizumab pegol (CZP) monotherapy over two years in patients with active rheumatoid arthritis [abstract]. Arthritis Rheum. 2010;62(Suppl. 10):S765.
Yamamoto K, Takeuchi T, Yamanak H, Ishiguro N, Tanaka Y, Eguchi K, et al. Efficacy and safety of certolizumab pegol without methotrexate co-administration in Japanese patients with active rheumatoid arthritis [abstract]. Arthritis Rheum. 2011;63(Suppl. 10):1220.
Sokka T, Pincus T. Most patients receiving routine care for rheumatoid arthritis in 2001 did not meet inclusion criteria for most recent clinical trials or American College of Rheumatology criteria for remission. J Rheumatol. 2003;30(6):1138–46.
Pope J, Fleischmann R, Dougados M, Bingham CO, Massarotti E, Wollenhaupt J, et al. Rapid reductions in fatigue and sleep problems and correlation with improvements in patient-reported outcomes in patients with active RA treated with certolizumab pegol in the REALISTIC 12-week phase IIIb randomised controlled study [abstract no. 4491]. Value Health. 2011;14(7):A313.
Genovese MC, Rubbert-Roth A, Smolen JS, Kremer J, Khraishi M, Gomez-Reino J, et al. Longterm safety and efficacy of tocilizumab in patients with rheumatoid arthritis: a cumulative analysis of up to 4.6 years of exposure. J Rheumatol. 2013;40(4):768–80.
Singh JA, Wells GA, Christensen R, Tanjong Ghogomu E, Maxwell L, Macdonald JK, et al. Adverse effects of biologics: a network meta-analysis and Cochrane overview. Cochrane Database Syst Rev. 2011(2):CD008794.
Canadian Agency for Drugs and Technologies in Health. CADTH therapeutic review: clinical and economic overview: biological response modifier agents for adults with rheumatoid arthritis; 2011. http://www.cadth.ca/media/pdf/TR_RA_Clinical_and_Economic_Overview_e.pdf. Accessed 5 Sep 2012.
Mariette X, Bertin P, Arendt C, Terpstra I, VanLunen B, de Longueville M. Pooled analysis of the risk of serious infections and opportunistic infections in clinical trials of certolizumab pegol for rheumatoid arthritis [poster no. 213]. Rheumatology 2012;51(suppl 3):iii132
Lopez-Olivo MA, Tayar JH, Martinez-Lopez JA, Pollono EN, Cueto JP, Gonzales-Crespo MR, et al. Risk of malignancies in patients with rheumatoid arthritis treated with biologic therapy: a meta-analysis. JAMA. 2012;308(9):898–908.
Vassilopoulos D, Apostolopoulou A, Hadziyannis E, Papatheodoridis GV, Manolakopoulos S, Koskinas J, et al. Long-term safety of anti-TNF treatment in patients with rheumatic diseases and chronic or resolved hepatitis B virus infection. Ann Rheum Dis. 2010;69(7):1352–5.
Ramos-Casals M. Therapy: are TNF blockers safe for patients with hepatitis B virus infection? Nat Rev Rheumatol. 2010;6(11):618–20.
Schoels M, Knevel R, Aletaha D, Bijlsma JW, Breedveld FC, Boumpas DT, et al. Evidence for treating rheumatoid arthritis to target: results of a systematic literature search. Ann Rheum Dis. 2010;69(4):638–43.
Goekoop-Ruiterman YP, de Vries-Bouwstra JK, Allaart CF, van Zeben D, Kerstens PJ, Hazes JM, et al. Comparison of treatment strategies in early rheumatoid arthritis: a randomized trial. Ann Intern Med. 2007;146(6):406–15.
Grigor C, Capell H, Stirling A, McMahon AD, Lock P, Vallance R, et al. Effect of a treatment strategy of tight control for rheumatoid arthritis (the TICORA study): a single-blind randomised controlled trial. Lancet. 2004;364(9430):263–9.
Smolen JS, Landewe R, Breedveld FC, Dougados M, Emery P, Gaujoux-Viala C, et al. EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs. Ann Rheum Dis. 2010;69(6):964–75.
Goekoop-Ruiterman YP, de Vries-Bouwstra JK, Kerstens PJ, Nielen MM, Vos K, van Schaardenburg D, et al. DAS-driven therapy versus routine care in patients with recent-onset active rheumatoid arthritis. Ann Rheum Dis. 2010;69(1):65–9.
Smolen JS, Han C, van der Heijde DM, Emery P, Bathon JM, Keystone E, et al. Radiographic changes in rheumatoid arthritis patients attaining different disease activity states with methotrexate monotherapy and infliximab plus methotrexate: the impacts of remission and tumour necrosis factor blockade. Ann Rheum Dis. 2009;68(6):823–7.
Verstappen SM, Jacobs JW, van der Veen MJ, Heurkens AH, Schenk Y, ter Borg EJ, et al. Intensive treatment with methotrexate in early rheumatoid arthritis: aiming for remission. Computer Assisted Management in Early Rheumatoid Arthritis (CAMERA, an open-label strategy trial). Ann Rheum Dis. 2007;66(11):1443–9.
Aletaha D, Funovits J, Keystone EC, Smolen JS. Disease activity early in the course of treatment predicts response to therapy after one year in rheumatoid arthritis patients. Arthritis Rheum. 2007;56(10):3226–35.
Gulfe A, Kristensen LE, Geborek P. Six and 12 weeks treatment response predicts continuation of tumor necrosis factor blockade in rheumatoid arthritis: an observational cohort study from southern Sweden. J Rheumatol. 2009;36(3):517–21.
Lacroix BD, Lovern MR, Stockis A, Sargentini-Maier ML, Karlsson MO, Friberg LE. A pharmacodynamic Markov mixed-effects model for determining the effect of exposure to certolizumab pegol on the ACR20 score in patients with rheumatoid arthritis. Clin Pharmacol Ther. 2009;86(4):387–95.
Curtis JR, Luijtens K, Kavanaugh A. Predicting future response to certolizumab pegol in rheumatoid arthritis patients: features at 12 weeks associated with low disease activity at 1 year. Arthritis Care Res. 2012;64(5):658–67.
Scott DL, Wolfe F, Huizinga TW. Rheumatoid arthritis. Lancet. 2010;376(9746):1094–108.
Breedveld FC, Combe B. Understanding emerging treatment paradigms in rheumatoid arthritis. Arthritis Res Ther. 2011;13(Suppl. 1):S3.
Buch MH, Bingham SJ, Bryer D, Emery P. Long-term infliximab treatment in rheumatoid arthritis: subsequent outcome of initial responders. Rheumatology. 2007;46(7):1153–6.
Kavanaugh A. Rheumatoid arthritis: guidelines for RA therapy-avoiding hamartia. Nat Rev Rheumatol. 2010;6(9):505–6.
Acknowledgments
The authors acknowledge Jane Caple, David Murdoch and Mary Hines of inScience Communications, Springer Healthcare, for medical writing and editorial assistance. Assistance with post-submission requirements was provided by Sheridan Henness, PhD, inScience Communications, Springer Healthcare. This assistance was funded by UCB Pharma, Milan, Italy. This article was published in a supplement sponsored by UCB Pharma SpA, Italy. The supplement was guest edited by Daniel Aletaha and peer reviewed by Leonard H. Calabrese who both received a small honorarium from Springer Healthcare to cover out-of-pocket expenses. D.A. has received honoraria and research grants from UCB, and honoraria from Abbvie, Grünenthal, Janssen, Merck, Medac, Mitsubishi Tanabe, Pfizer, AstraZeneca, Eli Lilly, Novo Nordisk, and Sanofi/Regeneron. L.H.C. has consulted for UCB, Roche, Janssen, Pfizer and BMS.
Conflict of interest
The authors have no conflict of interest to declare.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Punzi, L., Lapadula, G. & Mathieu, A. Efficacy and Safety of Certolizumab Pegol in Rheumatoid Arthritis: Meeting Rheumatologists’ Requirements in Routine Clinical Practice. BioDrugs 28 (Suppl 1), 25–37 (2014). https://doi.org/10.1007/s40259-013-0065-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40259-013-0065-y