Abstract
Background
Cost-effectiveness analyses (CEAs) have been performed for oral non-disease-altering osteoarthritis (OA) treatments for well over a decade. During that period the methods for performing these analyses have evolved as pharmacoeconomic methods have advanced, new treatments have been introduced, and the knowledge of associated adverse events (AEs) has improved.
Objective
The objective of this systematic review was to trace the development of CEAs for oral non-disease-altering treatments in OA.
Methods
A systematic search for CEAs of OA oral treatments was performed of the English-language medical literature using the following databases: PubMed, EMBASE, MEDLINE In-Process, EconLit, and Cochrane. Key requirements for inclusion were that the population described patients with OA or arthritis and that the analysis reported at least one incremental cost-effectiveness ratio. Each identified publication was assessed for inclusion. Thirteen characteristics and all AEs appearing in each included CEA were extracted and organized. Reference lists from these CEAs were also searched. A chronology of key CEAs in the field was compiled, noting the characteristics that advanced the state of the art in modeling oral OA treatments.
Results
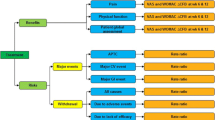
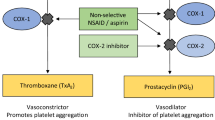
Thirty publications of 28 CEAs were identified and evaluated. Developments in CEAs included an expanded set of comparators that broadened from non-steroidal anti-inflammatory drugs (NSAIDs) only to NSAIDs plus gastroprotective agents, cyclooxygenase-2 inhibitors, and opioids. In turn, AEs expanded from gastrointestinal (GI) events to also include cardiovascular (CV) and neurological events. Efficacy, which initially was presumed to be equivalent for all treatments, evolved to treatment-specific efficacies. Decision-tree analyses were generally replaced by Markov models or, occasionally, stochastic or discrete event simulation. Finally, outcomes have progressed from GI-centric measures to also include quality-adjusted life-years.
Conclusion
Methods used by CEAs of oral non-disease-altering OA treatments have evolved in response to changing treatments with different safety profiles and efficacies as well as technical advances in the application of decision science to health care.

Similar content being viewed by others
References
Centers for Disease Control and Prevention. Prevalence of disabilities and associated health conditions among adults—United States, 2005. MMWR. 2009;58(16):421–6.
Centers for Disease Control and Prevention. Prevalence of doctor-diagnosed arthritis and arthritis-attributable activity limitation—United States, 2007–2009. MMWR. 2010;59(39):1261–5.
Centers for Disease Control and Prevention. National and state medical expenditures and lost earnings attributable to arthritis and other rheumatic conditions—United States, 2003. MMWR. 2007;56(1):4–7.
Hootman JM, Helmick CG. Projections of US prevalence of arthritis and associated activity limitations. Arthritis Rheum. 2006;54(1):226–9.
Murphy L, Helmick CG. The impact of osteoarthritis in the United States: a population-health perspective. Am J Nurs. 2012;112(3 Suppl 1):S13–9.
Caruso I, Bianchi PG. Gastroscopic evaluation of anti-inflammatory agents. Br Med J. 1980;280(6207):75–8.
Dajani EZ, Nissen CH. Gastrointestinal cytoprotective effects of misoprostol: clinical efficacy overview. Dig Dis Sci. 1985;30(11 Suppl):194S–200S.
Carrin GJ, Torfs KE. Economic evaluation of prophylactic treatment with misoprostol in osteoarthritic patients treated with NSAIDs. The case of Belgium. Rev Epidemiol Sante Publique. 1990;38(3):187–99.
Wielage RC, Bansal M, Andrews JS, et al. Cost-utility analysis of duloxetine in osteoarthritis: a US private payer perspective. Appl Health Econ Health Policy. 2013;11(3):219–36.
Rothfuss J, Mau W, Zeidler H, et al. Socioeconomic evaluation of rheumatoid arthritis and osteoarthritis: a literature review. Semin Arthritis Rheum. 1997;26(5):771–9.
Ruof J, Hülsemann JL, Stucki G. Evaluation of costs in rheumatic diseases: a literature review. Curr Opin Rheumatol. 1999;11(2):104–9.
Stucki G, Johannesson M, Liang MH. Is misoprostol cost-effective in the prevention of nonsteroidal anti-inflammatory drug-induced gastropathy in patients with chronic arthritis? A review of conflicting economic evaluations. Arch Intern Med. 1994;154(18):2020–5.
Chen Y-F, Jobanputra P, Barton P, et al. Cyclooxygenase-2 selective non-steroidal anti-inflammatory drugs (etodolac, meloxicam, celecoxib, rofecoxib, etoricoxib, valdecoxib and lumiracoxib) for osteoarthritis and rheumatoid arthritis: a systematic review and economic evaluation. Health Technol Assess. 2008;12(11):1–278, iii.
Brown TJ, Hooper L, Elliott RA, et al. A comparison of the cost-effectiveness of five strategies for the prevention of non-steroidal anti-inflammatory drug-induced gastrointestinal toxicity: a systematic review with economic modelling. Health Technol Assess. 2006;10(38):iii–iv, xi–xiii, 1–183.
Drummond M, Maetzel A, Gabriel S, et al. Towards a reference case for use in future economic evaluations of interventions in osteoarthritis. J Rheumatol. 2003;30(Suppl 68):26–30.
Drummond MF. Health economic models: a question of balance—summary of an open discussion on the pharmacoeconomic evaluation of non-steroidal anti-inflammatory drugs. Rheumatology (Oxford). 2000;39(Suppl 2):29–32.
Maetzel A, Krahn M, Naglie G. The cost-effectiveness of celecoxib and rofecoxib in patients with osteoarthritis or rheumatoid arthritis. Ottawa: Canadian Coordinating Office for Health Technology Assessment; 2001.
Maetzel A, Krahn M, Naglie G. The cost effectiveness of rofecoxib and celecoxib in patients with osteoarthritis or rheumatoid arthritis. Arthritis Rheum. 2003;49(3):283–92.
Latimer N, Lord J, Grant RL, et al. Cost effectiveness of COX 2 selective inhibitors and traditional NSAIDs alone or in combination with a proton pump inhibitor for people with osteoarthritis. BMJ. 2009;339:b2538.
National Collaborating Centre for Chronic Conditions. Osteoarthritis: national clinical guideline for care and management in adults. Appendix D. London: Royal College of Physicians; 2008.
Brixner DI. A decision analysis model in the evaluation of NSAIDs in a managed care setting: a case study. Med Interface. 1994;7(11):145.
Rafter N, Milne R, Jackson R. PHARMAC Technology Assessment Report No. 55—listing rofecoxib and celecoxib in the Pharmaceutical Schedule. 2003.
McCabe CJ, Akehurst RL, Kirsch J, et al. Choice of NSAID and management strategy in rheumatoid arthritis and osteoarthritis: the impact on costs and outcomes in the UK. Pharmacoeconomics. 1998;14(2):191–9.
Haglund U, Svarvar P. The Swedish ACCES model: predicting the health economic impact of celecoxib in patients with osteoarthritis or rheumatoid arthritis. Rheumatology (Oxford). 2000;39(Suppl 2):51–6.
Pettitt D, Goldstein JL, McGuire A, et al. Overview of the Arthritis Cost Consequence Evaluation System (ACCES): a pharmacoeconomic model for celecoxib. Rheumatology (Oxford). 2000;39(Suppl 2):33–42; discussion 57–9
Svarvar P, Aly A. Use of the ACCES model to predict the health economic impact of celecoxib in patients with osteoarthritis or rheumatoid arthritis in Norway. Rheumatology (Oxford). 2000;39(Suppl 2):43–50.
Chancellor JVM, Hunsche E, de Cruz E, et al. Economic evaluation of celecoxib, a new cyclo-oxygenase 2 specific inhibitor, in Switzerland. Pharmacoeconomics. 2001;19(Suppl 1):59–75.
Marshall JK, Pellissier JM, Attard CL, et al. Incremental cost-effectiveness analysis comparing rofecoxib with nonselective NSAIDs in osteoarthritis: Ontario Ministry of Health perspective. Pharmacoeconomics. 2001;19(10):1039–49.
Moore AR, Phillips CJ, Pellissier JM, et al. Health economic comparisons of rofecoxib versus conventional nonsteroidal antiinflammatory drugs for osteoarthritis in the United Kingdom. J Med Econ. 2001;4(1–4):1–17.
Pellissier JM, Straus WL, Watson DJ, et al. Economic evaluation of rofecoxib versus nonselective nonsteroidal anti-inflammatory drugs for the treatment of osteoarthritis. Clin Ther. 2001;23(7):1061–79.
El-Serag HB, Graham DY, Richardson P, et al. Prevention of complicated ulcer disease among chronic users of nonsteroidal anti-inflammatory drugs: the use of a nomogram in cost-effectiveness analysis. Arch Intern Med. 2002;162(18):2105–10.
Kamath CC, Kremers HM, Vanness DJ, et al. The cost-effectiveness of acetaminophen, NSAIDs, and selective COX-2 inhibitors in the treatment of symptomatic knee osteoarthritis. Value Health. 2003;6(2):144–57.
Spiegel BMR, Targownik L, Dulai GS, et al. The cost-effectiveness of cyclooxygenase-2 selective inhibitors in the management of chronic arthritis. Ann Intern Med. 2003;138(10):795–806.
Moore A, Phillips C, Hunsche E, et al. Economic evaluation of etoricoxib versus non-selective NSAIDs in the treatment of osteoarthritis and rheumatoid arthritis patients in the UK. Pharmacoeconomics. 2004;22(10):643–60.
Yen Z-S, Lai M-S, Wang C-T, et al. Cost-effectiveness of treatment strategies for osteoarthritis of the knee in Taiwan. J Rheumatol. 2004;31(9):1797–803.
Schaefer M, DeLattre M, Gao X, et al. Assessing the cost-effectiveness of COX-2 specific inhibitors for arthritis in the Veterans Health Administration. Curr Med Res Opin. 2005;21(1):47–60.
Spiegel B, Chiou C, Ofman J. Minimizing complications from nonsteroidal antiinflammatory drugs: cost effectiveness of competing strategies in varying risk groups. Arthritis Rheum. 2005;53(2):185–97.
Marshall D, Strauss M, Pericak D, et al. Economic evaluation of controlled-release oxycodone vs oxycodone–acetaminophen for osteoarthritis pain of the hip or knee. Am J Manag Care. 2006;12(4):205–14.
Loyd M, Rublee D, Jacobs P. An economic model of long-term use of celecoxib in patients with osteoarthritis. BMC Gastroenterol. 2007;7:25.
Patkar A, Langley P, Janagap C, et al. Cost-effectiveness of extended-release and immediate-release tramadol for the treatment of chronic osteoarthritis pain. Value Health. 2007;10(3):A118.
Ward A, Bozkaya D, Fleischmann J, et al. Modeling the economic and health consequences of managing chronic osteoarthritis pain with opioids in Germany: comparison of extended-release oxycodone and OROS hydromorphone. Curr Med Res Opin. 2007;23(10):2333–45.
Al MJ, Maniadakis N, Grijseels EWM, et al. Costs and effects of various analgesic treatments for patients with rheumatoid arthritis and osteoarthritis in the Netherlands. Value Health. 2008;11(4):589–99.
Contreras-Hernández I, Mould-Quevedo JF, Torres-González R, et al. Cost-effectiveness analysis for joint pain treatment in patients with osteoarthritis treated at the Instituto Mexicano del Seguro Social (IMSS): comparison of nonsteroidal anti-inflammatory drugs (NSAIDs) vs. cyclooxygenase-2 selective inhibitors. Cost Eff Resour Alloc. 2008;6:21.
Bessette L, Risebrough N, Mittmann N, et al. Cost-utility of celecoxib use in different treatment strategies for osteoarthritis and rheumatoid arthritis from the Quebec healthcare system perspective. J Med Econ. 2009;12(3):246–58.
Brereton N, Winn B, Akehurst R. The cost-effectiveness of celecoxib vs diclofenac in the treatment of osteoarthritis in the UK: an update to the NICE model using data from the CONDOR trial. J Med Econ. 2012;15(3):465–72.
Day RO, Graham GG, Williams KM, et al. Variability in response to NSAIDs: fact or fiction? Drugs. 1988;36(6):643–51.
Burke TA, Zabinski RA, Pettitt D, et al. A framework for evaluating the clinical consequences of initial therapy with NSAIDs, NSAIDs plus gastroprotective agents, or celecoxib in the treatment of arthritis. Pharmacoeconomics. 2001;19(Suppl 1):33–47.
Lister BJ, Poland M, DeLapp RE. Efficacy of nabumetone versus diclofenac, naproxen, ibuprofen and piroxicam in osteoarthritis and rheumatoid arthritis. Am J Med. 1993;95(Suppl 2A):2S–9S.
Hawkey C, Kahan A, Steinbrück K, et al. Gastrointestinal tolerability of meloxicam compared to diclofenac in osteoarthritis patients. International MELISSA Study Group. Meloxicam Large-Scale International Study Safety Assessment. Br J Rheumatol. 1998;37(9):937–45.
Fries JF, Williams CA, Bloch DA, et al. Nonsteroidal anti-inflammatory drug-associated gastropathy: incidence and risk factor models. Am J Med. 1991;91(3):213–22.
MacDonald TM, Morant SV, Robinson GC, et al. Association of upper gastrointestinal toxicity of non-steroidal anti-inflammatory drugs with continued exposure: cohort study. BMJ. 1997;315(7119):1333–7.
Silverstein FE, Graham DY, Senior JR, et al. Misoprostol reduces serious gastrointestinal complications in patients with rheumatoid arthritis receiving nonsteroidal anti-inflammatory drugs: a randomized, double-blind, placebo-controlled trial. Ann Intern Med. 1995;123(4):241–9.
Food and Drug Administration. Celebrex Advisory Committee Briefing Document. 1998. Available from: http://www.fda.gov/ohrms/dockets/ac/98/transcpt/3480t1.rtf. Accessed 8 Jul 2000.
Langman MJ, Jensen DM, Watson DJ, et al. Adverse upper gastrointestinal effects of rofecoxib compared with NSAIDs. JAMA. 1999;282(20):1929–33.
Gabriel SE, Jaakkimainen RL, Bombardier C. The cost-effectiveness of misoprostol for nonsteroidal antiinflammatory drug-associated adverse gastrointestinal events. Arthritis Rheum. 1993;36(4):447–59.
Laine L. Approaches to nonsteroidal anti-inflammatory drug use in the high-risk patient. Gastroenterology. 2001;120:594–606.
Network PDR. Red Book: pharmacy’s fundamental reference. Montvale: PDR Network, LLC.; 2011.
Food and Drug Administration. Vioxx (Rofecoxib) tablets review. 1999.
Food and Drug Administration. Celebrex (celecoxib) capsules review: osteoarthritis statistical review. 1998.
Bensen WG, Fiechtner JJ, McMillen JI, et al. Treatment of osteoarthritis with celecoxib, a cyclooxygenase-2 inhibitor: a randomized controlled trial. Mayo Clin Proc. 1999;74(11):1095–105.
Geba GP, Weaver AL, Polis AB, et al. Efficacy of rofecoxib, celecoxib, and acetaminophen in osteoarthritis of the knee: a randomized trial. JAMA. 2002;287(1):64–71.
Pincus T, Koch GG, Sokka T, et al. A randomized, double-blind, crossover clinical trial of diclofenac plus misoprostol versus acetaminophen in patients with osteoarthritis of the hip or knee. Arthritis Rheum. 2001;44(7):1587–98.
Day R, Morrison B, Luza A, et al. A randomized trial of the efficacy and tolerability of the COX-2 inhibitor rofecoxib vs ibuprofen in patients with osteoarthritis. Arch Intern Med. 2000;160(12):1781–7.
Silverstein FE, Faich G, Goldstein JL, et al. Gastrointestinal toxicity with celecoxib vs nonsteroidal anti-inflammatory drugs for osteoarthritis and rheumatoid arthritis: the CLASS study: a randomized controlled trial. Celecoxib Long-Term Arthritis Safety Study. JAMA. 2000;284(10):1247–55.
Food and Drug Administration. Celebrex capsules (celecoxib) medical officer review NDA 20-998/S-009. 2000.
Food and Drug Administration. VIOXX tablets: VIOXX oral suspension (rofecoxib). VIOXX Gastrointestinal Outcomes Research Study (VIGOR) briefing information. NDA 21-052. 2001.
Williams HJ, Ward JR, Egger MJ, et al. Comparison of naproxen and acetaminophen in a two-year study of treatment of osteoarthritis of the knee. Arthritis Rheum. 1993;36(9):1196–206.
Bradley JD, Brandt KD, Katz BP, et al. Comparison of an antiinflammatory dose of ibuprofen, an analgesic dose of ibuprofen, and acetaminophen in the treatment of patients with osteoarthritis of the knee. N Engl J Med. 1991;325(2):87–91.
Food and Drug Administration. Medical officer’s advisory committee GI briefing document. Division of Anti-inflammatory, Analgesic and Ophthalmologic Drug Products: HFD-550. NDA 21-042. 2001.
Ofman JJ, Maclean CH, Straus WL, et al. Meta-analysis of dyspepsia and nonsteroidal antiinflammatory drugs. Arthritis Rheum. 2003;49(4):508–18.
Watson D, Yu C, Bolognese J, et al. Improved upper-GI safety with etoricoxib compared with non selective cyclooxygenase inhibitors (NSAIDs) [abstract]. Arthritis Rheum. 2003;48(Suppl 9):S72.
Maetzel A, Ferraz MB, Bombardier C. The cost-effectiveness of misoprostol in preventing serious gastrointestinal events associated with the use of nonsteroidal antiinflammatory drugs. Arthritis Rheum. 1998;41(1):16–25.
Singh G, Fort JG, Goldstein JL, et al. Celecoxib versus naproxen and diclofenac in osteoarthritis patients: SUCCESS-I study. Am J Med. 2006;119(3):255–66.
Bensen WG, Zhao SZ, Burke TA, et al. Upper gastrointestinal tolerability of celecoxib, a COX-2 specific inhibitor, compared to naproxen and placebo. J Rheumatol. 2000;27(8):1876–83.
Goldstein JL, Eisen GM, Burke TA, et al. Dyspepsia tolerability from the patients’ perspective: a comparison of celecoxib with diclofenac. Aliment Pharmacol Ther. 2002;16(4):819–27.
Simon LS, White WB, MacDonald TM, et al. Cardiovascular safety of celecoxib: a meta-analysis of 41 clinical studies in 44,300 patients. Arthritis Rheum. 2005;52(Suppl 9):S406–7.
Wallace M, Thipphawong J. Clinical trial results with OROS® hydromorphone. J Pain Symptom Manage. 2007;33(Suppl 2):S25–32.
Kosinski M, Schein J, Kavanagh S. Health-related quality-of life (HRQoL) outcomes for patients with chronic osteoarthritis (OA) pain of the hip or knee treated with once-daily OROS hydromorphone versus ER oxycodone [poster 818]. In: 25th Annual Scientific Meeting of the American Pain Society, San Antonio, TX; 2006.
Furlan A, Sandoval J, Mailis-Gagnon A, et al. Opioids for chronic noncancer pain: a meta-analysis of effectiveness and side effects. CMAJ. 2006;174(11):1589–94.
Ta LE, Dionne RA. Treatment of painful temporomandibular joints with a cyclooxygenase-2 inhibitor: a randomized placebo-controlled comparison of celecoxib to naproxen. Pain. 2004;111(1–2):13–21.
Kivitz AJ, Moskowitz RW, Woods E, et al. Comparative efficacy and safety of celecoxib and naproxen in the treatment of osteoarthritis of the hip. J Int Med Res. 2001;29(6):467–79.
Pincus T, Koch G, Lei H, et al. Patient Preference for Placebo, Acetaminophen (paracetamol) or Celecoxib Efficacy Studies (PACES): two randomised, double blind, placebo controlled, crossover clinical trials in patients with knee or hip osteoarthritis. Ann Rheum Dis. 2004;63(8):931–9.
Lehmann R, Brzosko M, Kopsa P, et al. Efficacy and tolerability of lumiracoxib 100 mg once daily in knee osteoarthritis: a 13-week, randomized, double-blind study vs. placebo and celecoxib. Curr Med Res Opin. 2005;21(4):517–26.
Sheldon E, Beaulieu A, Paster Z, et al. Efficacy and tolerability of lumiracoxib in the treatment of osteoarthritis of the knee: a 13-week, randomized, double-blind comparison with celecoxib and placebo. Clin Ther. 2005;27(1):64–77.
Farkouh ME, Kirshner H, Harrington RA, et al. Comparison of lumiracoxib with naproxen and ibuprofen in the Therapeutic Arthritis Research and Gastrointestinal Event Trial (TARGET), cardiovascular outcomes: randomised controlled trial. Lancet. 2004;364(9435):675–84.
Schnitzer TJ, Burmester GR, Mysler E, et al. Comparison of lumiracoxib with naproxen and ibuprofen in the Therapeutic Arthritis Research and Gastrointestinal Event Trial (TARGET), reduction in ulcer complications: randomised controlled trial. Lancet. 2004;364(9435):665–74.
Bjordal JM, Ljunggren AE, Klovning A, et al. Non-steroidal anti-inflammatory drugs, including cyclo-oxygenase-2 inhibitors, in osteoarthritic knee pain: meta-analysis of randomised placebo controlled trials. BMJ. 2004;329(7478):1317.
Deeks JJ, Smith LA, Bradley MD. Efficacy, tolerability, and upper gastrointestinal safety of celecoxib for treatment of osteoarthritis and rheumatoid arthritis: systematic review of randomised controlled trials. BMJ. 2002;325(7365):619.
Zhang W, Jones A, Doherty M. Does paracetamol (acetaminophen) reduce the pain of osteoarthritis? A meta-analysis of randomised controlled trials. Ann Rheum Dis. 2004;63(8):901.
White WB, West CR, Borer JS, et al. Risk of cardiovascular events in patients receiving celecoxib: a meta-analysis of randomized clinical trials. Am J Cardiol. 2007;99(1):91–8.
Laine L, Curtis SP, Cryer B, et al. Assessment of upper gastrointestinal safety of etoricoxib and diclofenac in patients with osteoarthritis and rheumatoid arthritis in the Multinational Etoricoxib and Diclofenac Arthritis Long-term (MEDAL) programme: a randomised comparison. Lancet. 2007;369(9560):465–73.
Chan FKL, Lanas A, Scheiman J, et al. Celecoxib versus omeprazole and diclofenac in patients with osteoarthritis and rheumatoid arthritis (CONDOR): a randomised trial. Lancet. 2010;376(9736):173–9.
Agency for Healthcare Research and Quality. HCUPnet: a tool for identifying, tracking, and analyzing national hospital statistics. Available from: http://hcupnet.ahrq.gov/. Accessed 5 Oct 2012.
Wielage RC, Samsa GP, Klein TM, et al. A maximum likelihood simulation technique for estimating adverse event rates from published trials PRM145. In: International Society for Pharmacoeconomics and Outcomes Research 15th Annual European Congress. Berlin; 2012. Available from: http://www.ispor.org/research_pdfs/42/pdffiles/PRM145.pdf. Accessed 8 Apr 2013.
Bucher HC, Guyatt GH, Griffith LE, et al. The results of direct and indirect treatment comparisons in meta-analysis of randomized controlled trials. J Clin Epidemiol. 1997;50(6):683–91.
Tavakoli M. Modelling therapeutic strategies in the treatment of osteoarthritis: an economic evaluation of meloxicam versus diclofenac and piroxicam. Pharmacoeconomics. 2003;21(6):443–54.
Bentkover JD, Baker AM, Kaplan H. Nabumetone in elderly patients with osteoarthritis: economic benefits versus ibuprofen alone or ibuprofen plus misoprostol. Pharmacoeconomics. 1994;5(4):335–42.
You JHS, Lee KKC, Chan TYK, et al. Arthritis treatment in Hong Kong—cost analysis of celecoxib versus conventional NSAIDS, with or without gastroprotective agents. Aliment Pharmacol Ther. 2002;16(12):2089–96.
Knill-Jones R, Drummond M, Kohli H, et al. Economic evaluation of gastric ulcer prophylaxis in patients with arthritis receiving non-steroidal anti-inflammatory drugs. Postgrad Med J. 1990;66(778):639–46.
Knill-Jones RP, McGhee SM, MacDonald FC. An economic evaluation of enteric-coated and standard formulations of naproxen from a gastrointestinal perspective. Br J Med Econ. 1994;7:185–96.
Jansen RB, Capri S, Nuijten MJC, et al. Economic evaluation of meloxicam (7.5 mg) versus sustained release diclofenac (100 mg) treatment for osteoarthritis: a cross-national assessment for France, Italy and the UK. Br J Med Econ. 1997;11:9–22.
Liaropoulos L, Spinthouri M, Ignatiades T, et al. Economic evaluation of nimesulide versus diclofenac in the treatment of osteoarthritis in Greece. Pharmacoeconomics. 1998;14(5):575–88.
Tarricone R, Martelli E, Parazzini F, et al. Economic evaluation of nimesulide versus diclofenac in the treatment of osteoarthritis in France, Italy and Spain. Clin Drug Investig. 2001;21(7):453–64.
Walton SM, Schumock GT, McLain DA. Cost analysis of flavocoxid compared to naproxen for management of mild to moderate OA. Curr Med Res Opin. 2010;26(9):2253–61.
Liedgens H, Nuijten MJC, Nautrup BP. Economic evaluation of tramadol/paracetamol combination tablets for osteoarthritis pain in the Netherlands. Clin Drug Investig. 2005;25(12):785–802.
Vidal J, Brosa M, Benito P, et al. Economic evaluation of tramadol/paracetamol combination tablets vs NSAIDs for osteoarthritis pain in Spain: PMS53. Value Health. 2009;12(7):A443.
Becker RV, Burke TA, McCoy MA, et al. A model analysis of costs of blood pressure destabilization and edema associated with rofecoxib and celecoxib among older patients with osteoarthritis and hypertension in a Medicare Choice population. Clin Ther. 2003;25(2):647–62.
Zabinski RA, Burke TA, Johnson J, et al. An economic model for determining the costs and consequences of using various treatment alternatives for the management of arthritis in Canada. Pharmacoeconomics. 2001;19(Suppl 1):49–58.
Kristiansen IS, Kvien TK. Cost-effectiveness of replacing NSAIDs with coxibs: diclofenac and celecoxib in rheumatoid arthritis. Expert Rev Pharmacoecon Outcomes Res. 2002;2(3):229–41.
Yun HR, Bae S-C. Cost-effectiveness analysis of NSAIDs, NSAIDs with concomitant therapy to prevent gastrointestinal toxicity, and COX-2 specific inhibitors in the treatment of rheumatoid arthritis. Rheumatol Int. 2003;25(1):9–14.
Bae S-C, Corzillius M, Kuntz KM, et al. Cost-effectiveness of low dose corticosteroids versus non-steroidal anti-inflammatory drugs and COX-2 specific inhibitors in the long-term treatment of rheumatoid arthritis. Rheumatology. 2003;42(1):46–53.
Fendrick AM, Bandekar RR, Chernew ME, et al. Role of initial NSAID choice and patient risk factors in the prevention of NSAID gastropathy: a decision analysis. Arthritis Rheum. 2002;47(1):36–43.
Ko C, Deyo R. Cost-effectiveness of strategies for primary prevention of nonsteroidal anti-inflammatory drug-induced peptic ulcer disease. J Gen Intern Med. 2000;15:400–10.
Acknowledgments
Funding for this research was received from Eli Lilly and Company, Indianapolis, IN, USA. None of the authors have any conflicts of interest. Ankur J. Patel, an employee of Medical Decision Modeling, made substantial contributions to the work but does not meet the criteria for authorship. The article also relies on work performed by Frank Cinfio, an employee of Medical Decision Modeling.
Mr. Wielage is an employee of Medical Decision Modeling, a contracted research organization and vendor to Eli Lilly and Company. Ms. Myers is an employee of Medical Decision Modeling. Mr. Klein is an employee and principal of Medical Decision Modeling. Dr. Happich is an employee of Lilly Deutschland GmbH.
Mr. Wielage was the primary researcher, writer and compiler of this work. He is the guarantor for the overall content. Ms. Myers designed and performed the literature search. Mr. Klein edited and contributed to the conceptual framework of the manuscript. Mr. Happich initiated the project and edited the manuscript. All authors approved the final manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wielage, R.C., Myers, J.A., Klein, R.W. et al. Cost-Effectiveness Analyses of Osteoarthritis Oral Therapies: a Systematic Review. Appl Health Econ Health Policy 11, 593–618 (2013). https://doi.org/10.1007/s40258-013-0061-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40258-013-0061-x