Abstract
Background
Probiotic supplementation in early life may be effective in preventing atopic dermatitis (AD); however, results regarding efficacy have been controversial.
Objective
The aim of our study was to investigate the effect of probiotic supplementation on the risk of AD.
Methods
We systematically searched PubMed, EBSCO, Embase and Web of Science databases up to 8 March 2018 for potentially relevant studies regarding probiotic supplementation and AD. Included infants and children were those with probiotic exposure in utero and/or after birth who were not previously diagnosed with AD. We calculated the odds ratios (ORs) and 95% confidence intervals (CIs) and used the Jadad and Newcastle–Ottawa scales to assess methodologic quality.
Results
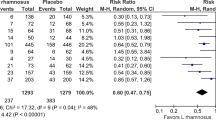
A total of 28 studies met the inclusion criteria. Compared with controls, probiotic treatment was associated with a reduced risk of AD (OR 0.69; 95% CI 0.58–0.82, P < 0.0001). The use of probiotics during both the prenatal and the postnatal period significantly reduced the incidence of AD (OR 0.67; 95% CI 0.54–0.82); however, analysis of studies of probiotics given prenatally only or postnatally only did not reach statistical significance.
Conclusions
Our meta-analysis showed that probiotic supplementation during both the prenatal and the postnatal period reduced the incidence of AD in infants and children. Our findings suggest that starting probiotic treatment during gestation and continuing through the first 6 months of the infant’s life may be of benefit in the prevention of AD.



Similar content being viewed by others
References
Legatzki A, Roler B, von Mutius ME. Microbiome diversity and asthma and allergy risk. Curr Allergy Asthma Rep. 2014;14:466.
Leung DY, Bieber T. Atopic dermatitis. Lancet. 2003;361:151–60.
Ege MJ, Bieli C, Frei R, van Strien RT, Riedler J, Ublagger E, et al. Prenatal farm exposure is related to the expression of receptors of the innate immunity and to atopic sensitization in school-age children. J Allergy Clin Immunol. 2006;117:817–23.
Kalliomaki M, Salminen S, Arvilommi H, Kero P, Koskinen P, Isolauri E. Probiotics in primary prevention of atopic disease: a randomised placebo-controlled trial. Lancet. 2001;357:1076–9.
Abrahamsson TR, Jakobsson T, Bottcher MF, et al. Probiotics in prevention of IgE-associated eczema: a doubleblind, randomized, placebo-controlled trial. J Allergy Clin Immunol. 2007;119:1174–80.
Kuitunen M, Kukkonen K, Juntunen-Backman K, Korpela R, Poussa T, Tuure T, et al. Probiotics prevent IgE-associated allergy until age 5 years in cesarean-delivered children but not in the total cohort. J Allergy Clin Immunol. 2009;123:335–41.
Huurre A, Laitinen K, Rautava S, Korkeamaki M, Isolauri E. Impact of maternal atopy and probiotic supplementation during pregnancy on infant sensitization: a double blind placebo-controlled study. Clin Exp Allergy. 2008;38:1342–8.
Isolauri E, Arvola T, Sütas Y, Moilanen E, Salminen S. Probiotics in the management of atopic eczema. Clin Exp Allergy. 2000;30:1604–10.
Cabana MD, McKean M, Caughey AB, Fong L, Lynch S, Wong A, et al. Early probiotic supplementation for eczema and asthma prevention: a randomized controlled trial. Pediatrics. 2017;140(3):e20163000.
Allen SJ, Jordan S, Storey M, Thornton CA, Gravenor MB, Garaiova I, et al. Probiotics in the prevention of eczema: a randomised controlled trial. Arch Dis Child. 2014;99(11):1014–9.
Mansfield JA, Bergin SW, Cooper JR, Olsen CH. Comparative probiotic strain efficacy in the prevention of eczema in infants and children: a systematic review and meta-analysis. Mil Med. 2014;179(6):580–92.
Jadad AR, Moore RA, Carroll D, Jenkinson C, Reynolds DJ, Gavaghan DJ, et al. Assessing the quality of reports of randomized clinical trials: is blinding necessary? Control Clin Trials. 1996;17(1):1–12.
Stroup DF, Berlin JA, Morton SC, Olkin I, Williamson GD, Rennie D, et al. Meta-analysis of observational studies in epidemiology: a proposal for reporting-Meta-analysis Of Observational Studies in Epidemiology (MOOSE) group. JAMA. 2000;283:2008–12.
Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ (Clin Res Ed). 2003;327:557–60.
Mantel N, Haenszel W. Statistical aspects of the analysis of data from retrospective studies of disease. J Natl Cancer Inst. 1959;22:719–48.
DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials. 1986;7:177–88.
Munafo MR, Clark TG, Flint J. Assessing publication bias in genetic association studies: evidence from a recent meta-analysis. Psychiatry Res. 2004;129:39–44. https://doi.org/10.1016/j.psychres.2004.06.011.
Begg CB, Mazumdar M. Operating characteristics of a rank correlation test for publication bias. Biometrics. 1994;50:1088–101.
Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997;315(7109):629–34.
Enomoto T, Sowa M, Nishimori K, Shimazu S, Yoshida A, Yamada K, et al. Effects of bifidobacterial supplementation to pregnant women and infants in the prevention of allergy development in infants and on fecal microbiota. Allergol Int. 2014;63(4):575–85.
Loo EX, Llanora GV, Lu Q, Aw MM, Lee BW, Shek LP. Supplementation with probiotics in the first 6 months of life did not protect against eczema and allergy in at-risk Asian infants: a 5-year follow-up. Int Arch Allergy Immunol. 2014;163(1):25–8.
Abrahamsson TR, Jakobsson T, Björkstén B, Oldaeus G, Jenmalm MC. No effect of probiotics on respiratory allergies: a seven-year follow-up of a randomized controlled trial in infancy. Pediatr Allergy Immunol. 2013;24(6):556–61.
West CE, Hammarström ML, Hernell O. Probiotics in primary prevention of allergic disease—follow-up at 8–9 years of age. Allergy. 2013;68(8):1015–20.
Rautava S, Kainonen E, Salminen S, Isolauri E. Maternal probiotic supplementation during pregnancy and breast-feeding reduces the risk of eczema in the infant. J Allergy Clin Immunol. 2012;130(6):1355–60.
Jensen MP, Meldrum S, Taylor AL, Dunstan JA, Prescott SL. Early probiotic supplementation for allergy prevention: long-term outcomes. J Allergy Clin Immunol. 2012;130(5):1209–11.
Wickens K, Black P, Stanley TV, Mitchell E, Barthow C, Fitzharris P, et al. A protective effect of Lactobacillus rhamnosus HN001 against eczema in the first 2 years of life persists to age 4 years. Clin Exp Allergy. 2012;42(7):1071–9.
Boyle RJ, Ismail IH, Kivivuori S, Licciardi PV, Robins-Browne RM, Mah LJ, et al. Lactobacillus GG treatment during pregnancy for the prevention of eczema: a randomized controlled trial. Allergy. 2011;66(4):509–16.
Kim JY, Kwon JH, Ahn SH, Lee SI, Han YS, Choi YO, et al. Effect of probiotic mix (Bifidobacterium bifidum, Bifidobacterium lactis, Lactobacillus acidophilus) in the primary prevention of eczema: a double-blind, randomized, placebo-controlled trial. Pediatr Allergy Immunol. 2010;21:e386–93.
Niers L, Martín R, Rijkers G, Sengers F, Timmerman H, van Uden N, et al. The effects of selected probiotic strains on the development of eczema (the PandA study). Allergy. 2009;64(9):1349–58.
West CE, Hammarström ML, Hernell O. Probiotics during weaning reduce the incidence of eczema. Pediatr Allergy Immunol. 2009;20(5):430–7.
Soh SE, Aw M, Gerez I, Chong YS, Rauff M, Ng YP, et al. Probiotic supplementation in the first 6 months of life in at risk Asian infants—effects on eczema and atopic sensitization at the age of 1 year. Clin Exp Allergy. 2009;39(4):571–8.
Wickens K, Black PN, Stanley TV, Mitchell E, Fitzharris P, Tannock GW, et al. A differential effect of 2 probiotics in the prevention of eczema and atopy: a double-blind, randomized, placebo-controlled trial. J Allergy Clin Immunol. 2008;122(4):788–94.
Kalliomäki M, Salminen S, Poussa T, Isolauri E. Probiotics during the first 7 years of life: a cumulative risk reduction of eczema in a randomized, placebo-controlled trial. J Allergy Clin Immunol. 2007;119(4):1019–21.
Kukkonen K, Savilahti E, Haahtela T, Juntunen-Backman K, Korpela R, Poussa T, et al. Probiotics and prebiotic galacto-oligosaccharides in the prevention of allergic diseases: a randomized, double-blind, placebo-controlled trial. J Allergy Clin Immunol. 2007;119(1):192–8.
Dotterud CK, Storro O, Johnsen R, Oien T. Probiotics in pregnant women to prevent allergic disease: a randomized, double-blind trial. Br J Dermatol. 2010;163:616–23.
Kopp MV, Hennemuth I, Heinzmann A, Urbanek R. Randomized, double-blind, placebo-controlled trial of probiotics for primary prevention: no clinical effects of Lactobacillus GG supplementation. Pediatrics. 2008;121:e850–6.
Allen SJ, Jordan S, Storey M, et al. Probiotics and atopic ECZEMA: a double-blind randomised controlled trial. Arch Dis Child. 2012;97:A2.
Rozé JC, Barbarot S, Butel MJ, Kapel N, Waligora-Dupriet AJ, De Montgolfier I, et al. An (alpha)-lactalbuminenriched and symbiotic-supplemented v a standard infant formula: a multicentre, double-blind, randomised trial. Br J Nutr. 2012;107:1616–22.
Kukkonen AK, Kuitunen M, Savilahti E, Pelkonen A, Malmberg P, Mäkelä M. Airway inflammation in probiotic-treated children at 5 years. Pediatr Allergy Immunol. 2011;22:249–51.
Ou CY, Kuo HC, Wang L, Hsu TY, Chuang H, Liu CA, et al. Prenatal and postnatal probiotics reduces maternal but not childhood allergic diseases: a randomized, double-blind, placebo-controlled trial. Clin Exp Allergy. 2012;42:1386–96.
Rautava S, Arvilommi H, Isolauri E. Specific probiotics in enhancing maturation of IgA responses in formula-fed infants. Pediatr Res. 2006;60(2):221–4.
Williams HC, Burney PG, Pembroke AC, Hay RJ. The U.K. Working Party’s diagnostic criteria for atopic dermatitis III Independent hospital validation. Br J Dermatol. 1994;131:406–16.
Hanifin JM, Rajka G. Diagnostic features of atopic dermatitis. Acta Derm Venerol (Stockh). 1980;92:44–7.
Novak N, Yu CF, Bussmann C, Maintz L, Peng WM, Hart J, et al. Putative association of a TLR9 promoter polymorphism with atopic eczema. Allergy. 2007;62:766–72.
He F, Ouwehand AC, Isolauri E, Hashimoto H, Benno Y, Salminen S. Comparison of mucosal adhesion and species identification of Bifidobacteria isolated from healthy and allergic infants. FEMS Immunol Med Microbiol. 2001;30:43–7.
Prescott SL, Wickens K, Westcott L, Jung W, Currie H, Black PN, et al. Supplementation with Lactobacillus rhamnosus or Bifidobacterium lactis probiotics in pregnancy increases cord blood interferon-gamma and breast milk transforming growth factor-beta and immunoglobin A detection. Clin Exp Allergy. 2008;38:1606–14.
Acknowledgements
The authors acknowledge the authors of the studies that made up the database for this meta-analysis.
Author information
Authors and Affiliations
Contributions
Lin Li conceived and designed the paper. Lin Li, Zhen Han and Xiaoping Niu extracted the data. Yuliang Jia analyzed the data. Guozheng Zhang and Shunguo Zhang contributed materials/analysis tools. Lin Li and Zhen Han contributed to the writing of the manuscript. Chiyi He proofread the manuscript.
Corresponding author
Ethics declarations
Funding
No sources of funding were used to conduct this study or prepare this manuscript.
Conflict of interest
Dr. Lin Li, Dr. Zhen Han, Dr. Xiaoping Niu, Dr. Guozheng Zhang, Dr. Yuliang Jia, Dr. Shunguo Zhang and Dr. Chiyi He have no conflicts of interest that are directly relevant to the content of this article.
Rights and permissions
About this article
Cite this article
Li, L., Han, Z., Niu, X. et al. Probiotic Supplementation for Prevention of Atopic Dermatitis in Infants and Children: A Systematic Review and Meta-analysis. Am J Clin Dermatol 20, 367–377 (2019). https://doi.org/10.1007/s40257-018-0404-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40257-018-0404-3