Abstract
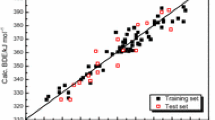
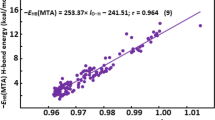
The enthalpies of formation of solid organic compounds containing carbon, nitrogen, oxygen, and hydrogen were estimated using two suggested descriptor sets, separately, by machine learning methods. The two descriptor sets are both composed of descriptors of Benson’s groups and corrected groups. The main differences between them are that one is generated based on atoms and the other is based on bonds. An in-house program was specially written in Java to extract all the descriptors with a function to ensure that each atom(or bond) of a molecule is represented by Benson’s groups once for an atom-based(or bond-based) descriptor set. Multiple linear regression and partial least squares were used, separately, to build models to predict the enthalpy of formation for two descriptor sets. The combination of the models constructed by two descriptor sets based on the atoms and the bonds achieved the best-predicted results in this paper, and the corresponding results of the test set are better than that in the literature, from which the original data were retrieved. Further, a small data set of fluorinated molecules was collected, and satisfactory results were also obtained for these molecules containing fluorine with the assistance of the former data set.

Similar content being viewed by others
References
Rosenholm J. B., Adv. Colloid Interface Sci., 2017, 247, 264
Argoub K., Benkouider A. M., Yahiaoui A., Kessas R., Guella S., Bagui F., Fluid Phase Equilibria, 2014, 380, 121
Liu Y. L., Zhao G., Tang Y. X., Zhang J. C., Hu L., Imler G. H., Parrish D. A., Shreeve J. M., Journal of Materials Chemistry A, 2019, 7, 7875
Gao H., Shreeve J. M., Chem. Rev., 2011, 111, 7377
Xiong H., Yang H., Lei C., Yang P., Hu W., Cheng G., Dalton Trans, 2019, 48, 14705
Chavez D. E., Hiskey M. A., Naud D. L., Parrish D., Angewandte Chemie, 2008, 120, 8431
Jafari M., Ghani K., Keshavarz M. H., Derikvandy F., Propellants, Explosives, Pyrotechnics, 2018, 43, 1236
Cawkwell M. J., Burch A. C., Ferreira S. R., Lease N., Manner V. W., J. Chem. Inf. Model, 2021, 61, 3337
Xiaohong L., Ruizhou Z., Xianzhou Z., Structural Chemistry, 2012, 24, 1193
Emel’yanenko V. N., Nagrimanov R. N., Verevkin S. P., Journal of Thermal Analysis and Calorimetry, 2017, 128, 1535
Zaitsau D. H., Verevkin S. P., Journal of Molecular Liquids, 2019, 287, 110959
Verevkin S. P., Emel’yanenko V. N., Zherikova K. V., Zelenina L. N., Zaitsau D. H., Pimerzin A. A., Chemical Physics Letters, 2020, 739, 136911
Zaitsau D. H., Yermalayeu A. V., Emel’yanenko V. N., Verevkin S. P., Chemical Engineering & Technology, 2018, 41, 1604
Konovalihin S. V., Chuev I. I., Guda S. A., Kovalev D. Y., Physics of Metals and Metallography, 2021, 121, 1188
Nagrimanov R. N., Ziganshin M. A., Solomonov B. N., Verevkin S. P., Structural Chemistry, 2018, 30, 247
Roknabadi A. G., Keshavarz M. H., Esmailpour K., Zamani M., Journal of the Iranian Chemical Society, 2016, 14, 57
Roknabadi A. G., Keshavarz M. H., Esmailpour K., Zamani M., Chemistry Select, 2016, 1, 6735
Zhang W., Zhang T., Guo W., Wang L., Li Z., Zhang J., Journal of Energetic Materials, 2019, 37, 433
Xu S., Wang Q. D., Sun M. M., Yin G., Liang J., RSC Adv, 2021, 11, 29690
Guan C., Lu M., Zeng W., Yang D., Han D., Industrial & Engineering Chemistry Research, 2020, 59, 4785
Yan X., Lan T., Jia Q., Yan F., Wang Q., Fluid Phase Equilibria, 2020, 507, 112437
Hayat S., Khan S., Journal of Molecular Structure, 2021, 1228, 129789
Wan Z., Journal of Molecular Structure, 2020, 1221, 128867
Sheibani N., Propellants Explosives Pyrotechnics, 2019, 44, 1254
Joudaki D., Shafiei F., Current Computer-Aided Drug Design, 2020, 16, 571
Wu T., Li W. L., Chen M. Y., Zhou Y. M., Zhang Q. Y., Chemical Papers, 2020, 75, 1619
Wu T., Li W. L., Chen M. Y., Zhou Y. M., Zhang Q. Y., Mol. Inform., 2020, 39, e2000102
Marrero J., Gani R., Fluid Phase Equilibria, 2001, 183/184, 183
Xiao K. X., Chen M. Y., Zhao T., Zhang Q. Y., Chemometrics and Intelligent Laboratory Systems, 2018, 178, 56
Xiao K. X., Chen M. Y., Zhao T., Zhou Y. M., Liu X., Zhang Q. Y., Xu L., Chemometrics and Intelligent Laboratory Systems, 2017, 169, 100
Zhang Q. Y., Wu C., Suo J., Zhou Y. M., Xu L., Journal of Chemometrics, 2016, 30, 70.
Wu T., Chen M. Y., Xiao K. X., Zhou Y. M., Zhang Q. Y., Chem. J. Chinese Universities, 2019, 40(6), 1158
National Institute of Standards and Technology, https://webbook.nist.gov/chemistry/NISTChemistryWebBook, 2022
Zhang G., Handbook of New Energetic Compound Data, Chemical Industry Press, Beijing, 2016
Zhang Q. Y., Xiao K. X., Chen M. Y., Xu L., Journal of Chemometrics, 2018, 32, 2928
Acknowledgements
This work was supported by the Open Research Fund Program of Science and Technology on Aerospace Chemical Power Laboratory, China(No.120201B01) and the National Natural Science Foundation of China(Nos.21875061, 21975066).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Conflicts of Interest
The authors declare no conflicts of interest.
Rights and permissions
About this article
Cite this article
Li, X., Luan, Y., Lu, Y. et al. Estimation of Enthalpy of Formation Using Benson’s Group Addition and Functional Group Correction. Chem. Res. Chin. Univ. 39, 296–304 (2023). https://doi.org/10.1007/s40242-022-2234-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-022-2234-3