Abstract
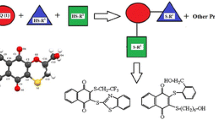
Structural modification of native compounds is an effective way to develop new drugs with increased pharmacological activities and improved material characteristics. In this study, caffeic acid sulphonamide deriva-tives(CSs) were synthesised by conjugating sulphonamides to the backbone of caffeic acid. The structures of these derivatives were elucidated by means of Fourier transform infrared spectroscopy(FTIR), 1H NMR, 13C NMR, and electrospray ionization mass spectroscopy(ESI-MS). A content determination method was established by ultraviolet detection. The lipophilicity, 2,2-diphenyl-1-picrylhydrazy free radical(DPPH) scavenging capacity, anti-coagulant effects, anti-bacterial activity, cytotoxicity, in vitro anti-pro liferative activity against tumour cells, and the ability of the compounds to promote the proliferation of chondrocytes were evaluated. The results indicate that CSs exhibit strong DPPH scavenging activity, high lipophilicity, good anti-coagulant activities, wide anti-bacterial activity range, low cytotoxicity, and an excellent ability to promote chondrocyte proliferation.
Similar content being viewed by others
References
Huang L., Lei T., Lin C., Kuang X., Chen H., Zhou H., Fitoterapia, 2010, 81(5), 389
Luo X., Du C., Cheng H., Chen J. H., Lin C., Molecules, 2017, 22(12), 2047
Vinayagam R., Jayachandran M., Xu B., Phytother. Res., 2016, 30(2), 184
Khadem S., Maries R. J. Molecules, 2010, 75(11), 7985
Samec D., Valek-Zulj L., Martinez S., Griiz I., Piljac A., Piljac-Zegarac J., Ind. Crops andProd., 2016, 84, 104
Reis F. S., Sousa D., Barros L., Martins A., Morales P., Ferreira I. C., Vasconcelos M. H., Pood Chem. Toxicol, 2016, 90, 45
Marino A., Zengin G., Nostro A., Ginestra G., Dugo P., Cacciola F., Miceli N., Taviano M. F., Filocamo A., Bisignano G., Aktumsek A., Nat. Prod. Res., 2016, 30(22), 2620
Zhang Y. B., Wu P., Zhang X. L., Xia C., Li G. Q., Ye W. C., Wang G. C., Li Y. L., Molecules, 2015, 20(11), 19947
Kerns E. H., Di L., Drug-like Properties: Concepts, Structure Design and Methods, Elsevier Press, Oxford, 2008
Son S., Lewis B. A., J. Agric. Pood. Chem., 2002, 50(3), 468
Wei Q., Jiang H., Zhang J., Guo P., Wang H., Med. Chem. Res., 2012, 27(8), 1905
Jiang R. W., Lau K. M., Hon P. M., Mak T. C., Woo K. S., Fung K. P., Curr. Med. Chem., 2005, 72(2), 237
Murthy H. N., Kim Y. S., Park S. Y., Paek K. Y., Appl. Microbiol. Biotechnol., 2014, 98(19), 7707
Wei J., Liu K., Lin F., He C., Luo X., Zou J., He W., Nong W., Lin C., Appl. Biol. Chem., 2016, 59(2), 271
Wei S., Lu Z., Zou Y., Lin X., Lin C., Liu B., Zheng L., Zhao J., Plos One, 2015, 70(6), e0125930
Liu Q., Li M. Y., Lin X., Lin C. W, Liu B. M., Zheng L., Zhao J. M., Chem. Biol. Interact, 2014, 227, 127
Huang L., Lin C., Li A., Wei B., Teng J., Li L., Nat. Prod. Commun., 2010, 5(8), 1263
Kato Y., Subcell. Biochem., 2014, 77, 21
Chu Y., Gao J., Niu J., Huang Y. F., Chen M., Wang M. Z., Shang Q., Lu W. Q., Peng L. H., Jiang Z. H., Biomed. Pharmacother, 2018, 98, 157
Liu W., Han L. Y., Bi Y. L., J. Henan Univ. Tech., 2014, 35(3), 98
Chen Y. J., Shiao M. S., Hsu M. L., Tsai T. H., Wang S.Y, J. Agric. Food Chem., 2001, 49(11), 5615
Burhop A., Week R., Atzrodt J., Derdau V., Eur. J. Org. Chem., 2017, 2077(11), 1418
Koksal Z., Kalin R., Camadan Y., Usanmaz H., Almaz Z., Gulcin I., Gokcen T., Goren A. C., Ozdemir H., Molecules, 2017, 22(6), 793
Liu L., Liu Q., Lin X., Wei Q. J., Zheng L., In vitro Cell. Dev. Biol. Anim., 2014, 50(10), 982
Abraham D. J., Burger’s Medicinal Chemistry and Drug Discovery, Volume 2, Drug Discovery and Drug Development, 6th Edition, John Wiley and Sons Inc. Press, California, 2003
Brusač E., Jeličič M., Klarič D., Mornar A., Acta. Pharm., 2019, 69(4), 649
Atrián-Blasco E., Gascón S., RodríGuez-Yoldi M. J., Laguna M., Cerrada E., Inorg Chem., 2017, 56(14), 8562
Smith M. A., Rottkamp C. A., Nunomura A., Raina A. K., Perry G., Biochim. Biophys. Acta, 2000, 7502(1), 139
Li Y. Q., Song Q. L., Chen P. P., Zhao W. J., Wang S. S., Chem. Res. Chinese Universities, 2006, 22(6), 742
Wen Y., Killalea S., Mcgettigan R., Peely J. Ir. J. Med. Sci., 1996, 765(3), 210
Chen X., Touyz R. M., Park J. B., Schiffrin E. L., Hypertension, 2001, 38(3), 606
Alaa B., Biologies, 2011, 5, 7
Nunez-Navarro N. E., Santana F. M., Parra L. P., Zacconi F. C., Curr. Med. Chem., 2019, 26(17), 3175
Li C., Hu M., Jiang S., Liang Z., Wang J., Liu Z., Wang H., Kang W., Molecules, 2020, 25(1), 177
Liu Z., Wang X., Mao B., Xie X., West China J. Pharm. Sci., 2016, 2, 157
Banerjee T., Vliet A. V. D., Ziboh V. A., Prostaglandins Leukot Essent Patty Acids, 2002, 66(5), 485
Li Y., Yang M., Meng T., Niu Y., Dai Y., Zhang L., Zheng X., Jalava P., Dong G., Gao W., Zheng Y., Sci. Total Environ., 2020, 709(3), 135802
Yagawa Y., Tanigawa K., Kobayashi Y., Yamamoto M. J., Cancer Metast. Treat, 2017, 3(10), 218
Niehoff A., Kersting U. G., Helling S., Dargel J., Maurer J., Thevis M., Braggemann G. P., Eur. J. Appl. Physiol, 2010, 770(3), 651
Lu Z., Wei S., Wu H., Xiao L., Lin C., Liu B., Li Z., Zhao J., Bioorg Chem., 2014, 57, 99
Yammani R. R., Carlson C. S., Bresnick A. R., Loeser R. F., Arthritis Rheum., 2010, 54(9), 2901
Jiang P., Wei P., Zhao M. C., Chen Q., Wang Z., Chinese J. Tissue Eng Res., 2014, 18(30), 4845
Author information
Authors and Affiliations
Corresponding authors
Additional information
Supported by the Guangxi Natural Science Foundation, China(No.2016GXNSFBA380053), the Fund of the Guangxi Key Laboratory of Traditional Chinese Medicine Quality Standards, China(No.Guizhongzhongkai201505).
Rights and permissions
About this article
Cite this article
Peng, X., Zhao, A., Huang, K. et al. Synthesis of Caffeic Acid Sulphonamide Derivatives and Preliminary Exploration of Their Biological Applications. Chem. Res. Chin. Univ. 36, 795–803 (2020). https://doi.org/10.1007/s40242-020-0014-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-020-0014-5