Abstract
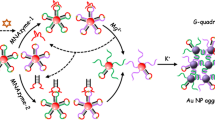
A label-free and turn-off fluorescent method for the quantitative detection of kanamycin based on a functional molecular beacon was developed. The molecular beacon consists of two hairpin structures with a split G-rich oligonucleotide in the middle. The kanamycin’s aptamer formed the loops portion for recognizing kanamycin, and the G-quadruplex bound by Thioflavin T(ThT) was employed as the reporter. In the absence of target, the molecular beacon folded into double stem-loops and the splited G-rich oligonucleotid came close to form a G-quadruplex. When ThT bound to the G-quadruplex, the fluorescence intensity of the solution increased. Upon the addition of kanamycin, the function between kanamycin and aptamer unfolded the hairpin and disassembled the G-quadraplex structure, resulting in a significant decrease in the fluorescence intensity. A good linear relationship ranging from 0.7 nmol/L to 10 nmol/L was achieved and the limit of detection was 0.37 nmol/L. Besides, it could efficiently recognize kanamycin in real samples.
Similar content being viewed by others
References
Megoulas N. C., Koupparis M. A., Anal. Chim. Acta, 2005, 547, 64
Cox L. A., Clin. Infect. Dis., 2006, 42, 1053
Hurd H. S., Enoe C., Sorensen L., Wachman H., Corns S. M., Bryden K. M., Grenier M., Risk Anal., 2008, 28, 695
Phillips I., Casewell M., Cox T., de Groot B., Friis C., Jones R., Nightingale C., Preston R., Waddell J., J. Antimicrob. Chemother., 2004, 54, 276
Phillips I., Casewell M., Cox T., de Groot B., Friis C., Jones R., Nightingale C., Preston R., Waddell J., J. Antimicrob. Chemother., 2004, 53, 28
Oertel R.Neumeister, Kirch W. V., J. Chromatogr. A, 2004, 1058, 197
Kitasato I., Yokota M., Inouye S., Igarashi M., Chemotherapy, 1990, 36, 155
Xing Y. P., Liu C., Zhou X. H., Sci. Rep.-UK, 2015, 5,8125
Chen S. H., Liang Y. C., Chou Y. W., J. Sep. Sci., 2006, 29, 607
Wang L. F., Peng J. D., Chromatographia, 2009, 69, 519
Blanchaert B., Jorge E. P., Jankovics P., Adams E., Ann V. S., Chro-matographia, 2013, 76, 1505
Long Y. H., Hernandez M., Kaale E., van Schepdael R. A., E., Bor-rull F., Calull M., Hoogmartens J., J. Chromatogr., 2003, 784, 255
Kaale E., Schepdael A.V., Roets E., Hoogmartens J., Electrophoresis, 2003, 24, 1119
Isoherranen N., Soback S., J. AOAC. Int., 1999, 82, 1017
Wang X. Y., Zou M. J., Xu X., Lei R., Li K., Li N., Anal. Bioanal. Chem., 2009, 395, 2397
Raz S. R., Bremer M. G. E. G., Haasnoot W., Norde W., Anal. Chem., 2009, 81, 7743
Frasconi M., Tel-Vered R., Riskin M., Willner I., Anal. Chem., 2010, 82, 2512
Althaus R., Berruga M. I., Montero A., Roca M., Molina M. P., Anal. Chim. Acta, 2009, 632, 159
Chen Y. Q., Wang Z. Q., Wang Z. H., Tang S. S., Zhu Y., Xiao X. L., J. Agric. Food Chem., 2008, 56, 2944
Loomans E. E. M. G., Wiltenburg J. V., Koets. M, Amerongen A. V., J. Agric., Food Chem., 2003, 51, 587
Tyagi S., Kramer N. F. R., Biotechnol., 1996, 14, 303
Morrison L. E., Halder T. C., Stols L. M., Anal. Biochem., 1989, 183, 231
Liu B., Yang X., Wang K., Chem. Res. Chinese Universities, 2012, 28(1), 37
Kostrikis L. G., Tyagi S., Mhlanga M. M., Ho D. D., Kramer F. R., Science, 1998, 279, 1228
Tyagi S., Bratu D. P., Kramer F. R., Nat. Biotechnol., 1998, 16, 49
Perlette J., Tan W., Anal. Chem., 2001, 73, 5544
Fang X., Li J. J., Tan W., Anal. Chem., 2000, 72, 3280
Li J., Tan W., Wang K., Xiao D., Yang X., He X., Tang Z., Anal. Sci., 2001, 17, 1149
Tang Z., Wang K., Tan W., Li J., Liu L., Guo Q., Meng X., Ma C., Huang S., Nucleic Acids Res., 2003, 31, e148
Li J. J., Geyer R., Tan W., Nucleic Acids Res., 2000, 28, e52
Bourdoncle A., Estevez T. A., Gosse C., Lacroix L., Vekhoff P., Le S. T., Jullien L., Mergny J. L., J. Am. Chem. Soc., 2006, 128, 11094
Williamson J. R., Annu. Rev. Biophys. Biomol. Struct., 1994, 23, 703
Sundquist W. I., Klug A., Nature, 1989, 342, 825
Song K. M., Cho M., Jo H., Min K., Jeon S. H., Kim T., Han M. S., Ku J. K., Ban C., Anal. Biochem., 2011, 415, 175
Chen T. X., Ning F., Liu H. S., Wu K. F., Li W., Chinese Chem. Lett., 2017, 28, 1380
Tong L. L., Li L., Chen Z., Biosens. Bioelectro., 2013, 49, 420
Wen Y., Wang L., Li L., Sensors, 2016, 16, 12
Stoltenburg R., Nikolaus N., Strehlitz B., J. Anal. Methods Chem., 2012, 14, 383
Bhasikuttan A. C., Mohanty J., Chem. Commun., 2015, 51, 7581
Tong L., Li L., Chen Z., Biosens. Bioelectron, 2013, 49, 420
Nikolaus N., Strehlitz B., Sensors, 2014, 14, 37
Amdursky N., Erez Y., Dan H. H., Acc. Chem. Res., 2012, 45, 1548
Mohanty J., Barooah N., Dhamodharan V., J. Am. Chem. Soc., 2013, 135, 367
Green J., Ying L., Klenerman D., Chem. Res. Chinese Universities, 2002, 18(1), 103
Author information
Authors and Affiliations
Corresponding authors
Additional information
Supported by the National Natural Science Foundation of China(No.21202010), the Natural Science Foundation of Hunan Province, China(No.2017JJ2275) and the Scientific Research Fund of Hunan Provincial Education Department, China (Nos.16B003, 17C0033).
Rights and permissions
About this article
Cite this article
Zhu, Y., Li, W., Tan, S. et al. A Label-free and Functional Fluorescent Oligonucleotide Probe Based on a G-Quadruplex Molecular Beacon for the Detection of Kanamycin. Chem. Res. Chin. Univ. 34, 541–545 (2018). https://doi.org/10.1007/s40242-018-7366-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-018-7366-0