Abstract
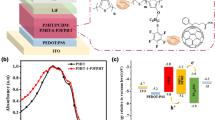
A series of tri(alkoxyl)benzene-fullerene dyads(PCBB-Cn, n=4, 6, 8, 10, 12) with varied tri(alkoxyl) chain lengths was designed, synthesized and used as acceptor materials in polymer solar cells(PSCs). The five fullerene dyads possess similar absorption spectra in dilute solution, decreased glass-transition temperature(T g ) and gradually elevated lowest unoccupied molecular orbital(LUMO) energy levels from–3.87 eV to–3.73 eV with the increase of the alkoxy chain length. In the fabrication of PSCs with poly(3-hexylthiophene)(P3HT) as donor and the fullerene dyads as acceptor, PCBB-Cn with longer tri(alkoxyl) chains and lower Tg can induce crystalline structure of P3HT during spin-coating the photoactive layer at room temperature and form nanoscale phase separated interpenetrating network of P3HT:PCBB-C n blend films, which results in the improvement of photovoltaic performance of PSCs. A power conversion efficiency of 3.03% for the PSCs based on P3HT:PCBB-C10 was obtained without thermal annealing or solvent annealing. The thermal and solvent annealing-free fabrication using the fullerene dyads as acceptor is very important for the roll to roll production of PSCs with flexible large area.
Similar content being viewed by others
References
Li G., Zhu R., Yang Y., Nat. Photon., 2012, 6(3), 153
Janssen R. A. J., Nelson J., Adv. Mater., 2013, 25(13), 1847
Zhang Z. G., Li Y. F., Sci. China Chem., 2015, 58(2), 192
Zhang S. Q., Ye L., Zhao W. C., Yang B., Wang Q., Hou J. H., Sci. China Chem., 2015, 58(2), 248
Yu G., Gao J., Hummelen J. C., Wudl F., Heeger A. J., Science, 1995, 270(5243), 1789
Halls J. J. M., Walsh C. A., Greenham N. C., Marseglia E. A., Friend R. H., Moratti S. C., Holmes A. B., Nature, 1995, 376(6540), 498
Schilinsky P., Waldauf C., Brabec C. J., Appl. Phys. Lett., 2002, 81(20), 3885
Li G., Shrotriya V., Huang J., Yao Y., Moriarty T., Emery K., Yang Y., Nat. Mater., 2005, 4(11), 864
Zhang M., Guo X., Ma W., Ade H., Hou J., Adv. Mater., 2014, 26(33), 5880
Cui C., Wong W. Y., Li Y., Energy Environ. Sci., 2014, 7(7), 2276
He Z., Xiao B., Liu F., Wu H., Yang Y., Xiao S., Wang C., Russell T. P., Cao Y., Nat. Photon., 2015, 9, 174
Liu Y., Zhao J., Li Z., Mu C., Ma W., Hu H., Jiang K., Lin H., Ade H., Yan H., Nat. Commun., 2014, 5, 5293
Etxebarria I., Guerrero A., Albero J., Garcia-Belmonte G., Palomares E., Pacios R., Org. Electron., 2014, 15(11), 2756
You J., Dou L., Yoshimura K., Kato T., Ohya K., Moriarty T., Emery K., Chen C. C., Gao J., Li G., Yang Y., Nat. Commun., 2013, 4, 1446
Bao Z., Dodabalapur A., Lovinger A. J., Appl. Phys. Lett., 1996, 69(26), 4108
Espinosa N., Hosel M., Angmo D., Krebs F. C., Energy Environ. Sci., 2012, 5(1), 5117
Choi S., Zhou Y., Haske W., Shim J. W., Fuentes-Hernandez C., Kippelen B., Org. Electron., 2015, 17, 349
Seidler N., Lazzerini G. M., Destri G. L., Marletta G., Cacialli F., J. Mater. Chem. C, 2013, 1(46), 7748
Chen D., Liu F., Wang C., Nakahara A., Russell T. P., Nano Lett., 2011, 11(5), 2071
Guo X., Cui C., Zhang M., Huo L., Huang Y., Hou J., Li Y., Energy Environ. Sci., 2012, 5(7), 7943
Zhang P., Li C., Li Y., Yang X., Chen L., Xu B., Tian W., Tu Y., Chem. Commun., 2013, 49(43), 4917
Li Y., Mao L., Gao Y., Zhang P., Li C., Ma C., Tu Y., Cui Z., Chen L., Sol. Energy Mater. Sol. Cells, 2013, 113, 85
Nakanishi T., Shen Y., Wang J., Li H., Fernandes P., Yoshida K., Yagai S., Takeuchi M., Ariga K., Kurth D. G., Mohwald H., J. Mater. Chem., 2010, 20(7), 1253
Cowie J. M. G., Reid V. M. C., McEwen I. J., British Poly. J., 1990, 23(4), 353
Reimschuessel H. K., J. Poly. Sci.: Polym. Chem. Edition, 1979, 17(8), 2447
Lee J., Han A. R., Yu H., Shin T. J., Yang C., Oh J. H., J. Am. Chem. Soc., 2013, 135(25), 9540
Jo J., Kim S. S., Na S. I., Yu B. K., Kim D. Y., Adv. Func. Mater., 2009, 19(6), 866
Ma W., Yang C., Gong X., Lee K., Heeger A. J., Adv. Func. Mater., 2005, 15(10), 1617
Kim Y., Cook S., Tuladhar S. M., Choulis S. A., Nelson J., Durrant J. R., Bradley D. D. C., Giles M., McCulloch I., Ha C. S., Ree M., Nat. Mater., 2006, 5(3), 197
Thompson B. C., Fréchet J. M. J., Angew. Chem. Int. Edit., 2008, 47(1), 58
Beaujuge P. M., Fréchet J. M. J., J. Am. Chem. Soc., 2011, 133(50), 20009
Xin H., Guo X., Ren G., Watson M. D., Jenekhe S. A., Adv. Energy Mater., 2012, 2(5), 575
Collins B. A., Li Z., Tumbleston J. R., Gann E., McNeill C. R., Ade H., Adv. Energy Mater., 2013, 3(1), 65
Lu X., Hlaing H., Germack D. S., Peet J., Jo W. H., Andrienko D., Kremer K., Ocko B. M., Nat. Commun., 2012, 3, 795
Qian D., Ma W., Li Z., Guo X., Zhang S., Ye L., Ade H., Tan Z., Hou J., J. Am. Chem. Soc., 2013, 135(23), 8464
Liu C., Hu X., Zhong C., Huang M., Wang K., Zhang Z., Gong X., Cao Y., Heeger A. J., Nanoscale, 2014, 6(23), 14297
Nakanishi T., Chem. Commun., 2010, 46(20), 3425
Chen L. M., Hong Z. R., Li G., Yang Y., Adv. Mater., 2009, 21(14/15), 1434
Vandewal K., Ma Z., Bergqvist J., Tang Z., Wang E., Henriksson P., Tvingstedt K., Andersson M. R., Zhang F., Inganäs O., Adv. Func. Mater., 2012, 22(16), 3480
Vandewal K., Tvingstedt K., Gadisa A., Inganäs O., Manca J. V., Phys. Rev. B, 2010, 81(12), 125204
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the National Natural Science Foundation of China(Nos.21204057, 91333204), the Natural Science Foundation of Jiangsu Province, China(No.BK2012213), the Priority Academic Development Program of Jiangsu Higher Education Institutions, China and the Soochow University Undergraduate Training Program for Innovation and Entrepreneurship, China( No.2013XjOZO).
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Xue, R., Zhao, Y., Xu, G. et al. Impact of alkoxyl tail of fullerene dyad acceptor on crystalline microstructure for efficient external treatment-free polymer solar cells with poly(3-hexylthiophene) as donor. Chem. Res. Chin. Univ. 31, 865–872 (2015). https://doi.org/10.1007/s40242-015-5184-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-015-5184-1