Abstract
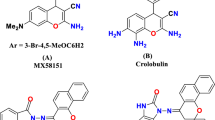
A novel series of first procaspase activating compound(PAC-1) analogues was designed, synthesized and evaluated for antitumor activity towards two cell lines[human promyelocytic leukemia cell line(HL60) and human embryonic lung fibroblast cell line(HLF)] by the MTT[3-(4,5)-dimethylthiahiazo(-z-y1)-3,5-di-phenytetrazo-liumromide]_method in vitro. The structures of all the compounds were confirmed by 1H NMR, MS and elemental analysis. Among the compounds synthesized,(E)-2-[(3-{[4-(tert-butyl)benzyl](methyl)amino}propyl)(methyl)amino]-N′-[4-(diethylamino)-2-hydroxybenzylidene]acetohydrazide(compound 6n) exhibits a good anti-proliferative activity to the majority of tumor cells tested, and selectively cleaves cancer cells. Thus, compound 6n was identified as promising lead compound for further structural modification.
Similar content being viewed by others
References
Igney F. H., Krammer P. H., Nature Rev. Cancer, 2002, 2, 277
Zornig M., Hueber A. O., Baum W., Evan G., Biochim. Biophys. Acta, 2001, 1551, F1
Su J. Q., Chi B. R., Li X., Liu L., Liu L. M., Qi Y. X., Wang Z. Y., Jin N. Y., Chem. Res. Chinese Universites, 2012, 28(3), 465
Hu W., Kavanagh J. J., Lancet Oncol., 2003, 4, 721
Hanahan D., Weinberg R., Cell, 2011, 144, 646
Earnshaw W. C., Martins L. M., Kaufmann S. H. Annu Rev Biochem., 1999, 68, 383
Hanahan D., Weinberg R., Cell, 2000, 100, 57
Fesik S. W., Nat. Rev. Cancer, 2005, 5, 876
Peterson Q. P., Goode D. R., West D. C. Ramsey K. N., Lee J. J. Y., Hergenrother P. J., J. Mol. Biol., 2009, 388, 144
Boatright K. M., Salvesen G. S., Curr. Opin. Cell Biol., 2003, 15, 725
Luthi A. U., Martin S. J., Cell Death Differ., 2007, 14, 641
Timmer J. C., Salvesen G. S., Cell Death Differ., 2007, 14, 66
Roy S., Bayly C. I., Gareau Y., Houtzager V. M., Kargman S., Keen S. L., Rowland K., Seiden I. M., Thornberry N. A., Nicholson D. W., Proc. Natl. Acad. Sci. USA, 2001, 98, 6132
Putt K. S., Chen G. W., Pearson J. M., Sandhorst J. S., Hoagland M. S., Kwon J. T., Hwang S. K., Jin H., Churchwell M. I., Cho M. H., Doerge D. R., Helferich W. G., Hergenrother P. J., Nature Chem. Biol., 2006, 2, 543
Peterson Q. P., Hsu D. C., Goode D. R., Novotny C. J., Totten R. K., Hergenrother P. J., J. Am. Chem. Soc., 2006, 128, 12424
Peterson Q. P., Hsu D. C., Novotny C. J., West D. C., Kim D., Schmit J. M., Dirikolu L., Hergenrother P. J., Fan T. M., Cancer Res., 2010, 70, 7232
Hsu D. C., Roth H. S., West D. C., Botham R. C., Novotny C. J. Schmid S. C., Hergenrother P. J., Comb. Sci., 2012, 14, 44
West D. C., Qin Y., Peterson Q. P., Thomas D. L., Palchaudhuri R., Morrison K. C., Lucas P. W., Palmer A. E., Fan T. M., Hergenrother P. J., Mol. Pharmaceutics, 2012, 9, 1425
Charkoudian L. K., Pham D. M., Franz K. J., J. Med. Chem., 2009, 52, 5721
Author information
Authors and Affiliations
Corresponding authors
Additional information
Supported by the National Natural Science Foundation of China(Nos.20771030, 20671025).
Rights and permissions
About this article
Cite this article
Luo, Hm., Yang, Cl., Zhang, Xy. et al. Design, synthesis and antitumor activity of a novel series of PAC-1 analogues. Chem. Res. Chin. Univ. 29, 906–910 (2013). https://doi.org/10.1007/s40242-013-3336-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-013-3336-8