Abstract
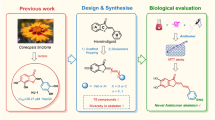
Aurora A is a cell cycle kinase linked to cancer. For the purpose of finding biologically active of novel compounds and providing new ideas for drug-design, we performed virtual screening in commercially available databases and got pyrazoleanthrone with promising inhibitory activity against Aurora A. Optimization of solvent accessible C7 position of pyrazoleanthrone made us get thirteen target compounds. These pyrazoleanthrone derivatives were evaluated by Aurora A inhibition assays in vitro. The results show that some target compounds could inhibit Aurora A kinase. Meanwhile, these title compounds were tested in vitro against hepatocellular carcinoma(HepG2) cells by the 3′-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide(MTT) method, showing that most of them had inhibitory potency. The inhibition rate of compound 6h was about 80% against HepG2 cells, and the IC50 value was 17.4 μmol/L, which would be considered for further study.
Similar content being viewed by others
References
Glover D. M., Leibowitz M. H., McLean D. A., Parry H., Cell, 1995, 81, 95
Andrews P. D., Knatko E., Moore W. J., Swedlow J. R., Curr. Opin. Cell Biol., 2003, 15, 672
Andrews P. D., Oncogene, 2005, 25, 5005
Qi W. Q., Liu X. B., Cooke L. S., Persky D. O., Miller T. P., Squires M., Mahadevan D., Int. J. Cancer, 2012, 130, 299
Marumoto T., Zhang D., Saya H., Nat. Rev. Cancer, 2005, 5, 42
Kollareddy M., Zheleva D., Dzubak P., Brahmkshatriya P. S., Lepsik M., Hajduch M., Invest. New Drugs, 2012, 30, 2411
Fujii S., Dotto G. P., Methods and Compositions for Reducing Skin Damage, WO 2007016419 A2, 2007-2-d8
Howard S., Berdini V., Boulstridge J. A., Carr M. G., Cross D. M., Curry J., Devine L. A., Early T. R., Fazal L., Gill A. L., Heathcote M., Maman S., Matthews J. E., McMenamin R. L., Navarro E. F., O’Brien M. A., O’Reilly M., Rees D. C., Reule M., Tisi D., Williams G., Vinković M., Wyatt P. G., J. Med. Chem., 2009, 52, 379
Aliagas-Martin I., Burdick D., Corson L., Dotson J., Drummond J., Fields C., Huang O. W., Hunsaker T., Kleinheinz T., Krueger E., Liang J., Moffat J., Phillips G., Pulk R., Rawson T. E., Ultsch M., Walker L., Wiesmann C., Zhang B., Zhu B. Y., Cochran A. G., J. Med. Chem., 2009, 52, 3300
Zhao B., Smallwood A., Yang J., Koretke K., Nurse K., Calamari A., Kirkpatrick R. B., Lai Z., Protein Sci., 2008, 17, 1791
van Donggen M., Weigelt J., Uppenberg J., Schultz J., Wikstrom M., Drug Discov. Today, 2002, 7, 471
Waszkowycz B., Curr. Opin. Drug Discov. Devel., 2002, 5, 407
Chopra D., Guru R. T. N. G., CrystEngComm, 2008, 10, 54
Kim M. F., Wiemer D., Tetrahedron Lett., 2004, 45, 4977
Cheetham G. M., Knegtel R. M., Coll J. T., Renwick S. B., Swenson L., Weber P., Lippke J. A., Austen D. A., J. Biol. Chem., 2002, 277, 42419
Bernal C., Mendez E., Terencio J., Boronat A., Imperial S., J. Anal. Biochem., 2005, 340, 245
Liu D., Zhu X. J., Jiang M., Chen H., Lan S. P., Chem. J. Chinese Universities, 2012, 33(10), 2249
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the Tianjin Small and Meclium-sized Enterprise Technology Innovation Fund, China(No.11ZXCXSY03500).
Rights and permissions
About this article
Cite this article
Sun, Xx., Sun, T., Wang, Ty. et al. Synthesis and characterization of pyrazoleanthrone derivatives as Aurora A kinase inhibitors. Chem. Res. Chin. Univ. 29, 1098–1103 (2013). https://doi.org/10.1007/s40242-013-3216-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-013-3216-2