Abstract
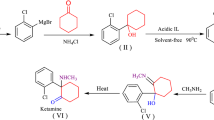
A series of new hydantoin compounds was synthesized with 3-bromo phenylacetic acid and 3,4-dimethylbromobenzene as starting material, 7-bromo-2-tetralone and 2-bromo-5,6,8,9-tetrahydro-7-benzocyclo-heptenone as intermediate and Ullmann reaction, Suzuki reaction and Bucherer-Berg’s reaction as key steps. The structures of the key intermediate and target compounds were confirmed by 1H NMR, 13C NMR, IR and MS. Bioactivity research showed that target compounds 6a, 6c, 6d, 6e and 15i had significant antitussive effect on the ammonia-induced cough of mice.
Similar content being viewed by others
References
Liu J. M., Zhao Y., Chinese Journal of Disinfection, 2001, 18, 218
Chen Q., Li Q. L., Journal of Fujian Agriculture and Forestry University (Natural Science Edition), 2003, 32, 225
Chen R. F., Xue G. B., Gu C. Y., Li H., Chinese Journal of Disease Control & Prevention, 2001, 5, 15
Zhang S. L., Xu Z. X., Huang J. X., Chen Z. X., Journal of Hubei University(Natural Science Edition), 2002, 24, 63
Li Q. L., Lin X. H., Journal of Fujian Medical University, 2001, 35, 176
Launay M., Potin D., Maillet M. J. B., Nicolai E. A., Dhar T. G. M., Iwanowicz E. J., Hydantoin Compounds Useful as Anti-inflammatory Agents, US 2002/0143035 A1, 2002
Murali D. T. G., Potin D., Maillet M. J. B., Launay M., Nicolai E. A., Iwanowicz E. J., Spiro-Hydantoin Compounds Useful as Anti-inflammatory Agents, US 2004/0009998 A1, 2004
Wang Y. S., 1-Methyl-Hydantoin Preparation in Application of Relieving Cough, Asthma and Phlegm Drugs. CN 1813717A, 2006
Kurono M., Unno R., Kimura H., Tomiya N., Sawai K., Miura K., Usui T., Kondo Y., Tanaka Y., Nakamura S., Suzuki T., Hayashi M., Hydantoin Derivatives for Treating Complications of Diabetes and Circulatory Diseases, US 5164391, 1992
Goodnow JR. R. A., Le K., Hydantoin-containing Glucokinase Activators, US 2003/0225286 A1, 2003
Sun Q., Kyle D. J., Aryl Substituted Hydantoin Compounds and Their Use as Sodium Channel Blockers, US 2004/0097569 A1, 2004
Mikami H., Ienage K., Method for Preventing or Treating Renal Failure, US 5084473, 1992
Yang B., Liu D., Li C. Z., Liu F. Y., Peng Y. M., Jiang Y. S., Renal Failure, 2007, 29, 1025
Chen D. G., Adjustment Androgen Acceptor Qctive Hydantoin Derivative Composition and Application, CN 200610067719.2, 2007
Liang T., Lu H. B., Xu Z. B., Mi H. Y., Wu P. F., Cui L., Wang E. S., Journal of Jilin University(Science Edition), 2008, 46, 139
Xu Z. B., Lu H. B., Liang Y. T., Liang T., Wang E. S., Journal of Jilin University(Science Edition), 2008, 46, 351
Wang E. S., Liang T., Lu H. B., Mi H. Y., Synthesis and Immunosuppressive Activity of New Amino Alcohol Derivatives, CN 101768086, 2010
Cui L. L., Zhang Q. L., Mi H. Y., Wang E. S., Journal of Jilin University( Science Edition), 2010, 48, 860
Mi H. Y., Cui L. L., Zhang Q. L., Li F., Jiang T., Liang Y. T., Wang E. S., Chem. Res. Chinese Universities, 2011, 27(4), 614
Cui L. L., Mi H. Y., Zhang Q. L., Wang W. W., Yang L. J., Liang Y. T., Wang E. S., Chem. Res. Chinese Universities, 2011, 27(5), 808
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Zhang, Ql., Song, Lj. & Wang, Es. Synthesis and antitussive effect of new hydantoin compounds. Chem. Res. Chin. Univ. 29, 76–81 (2013). https://doi.org/10.1007/s40242-012-2064-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-012-2064-9