Abstract
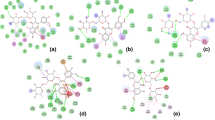
Traditionally, Tinospora cordifolia is commonly used in the treatment of diabetes and obesity; has been evaluated for their anti-diabetic and anti-obese potency in experimental animal models. However, the binding affinity of multiple bioactives with various proteins involved in the pathogenesis of diabetes and obesity has not been reported yet. Hence, the present study aimed to assess the binding affinity of multiple bioactives from T. cordifolia with various targets involved in the pathogenesis of diabetes and obesity. The ligands and targets were retrieved from the PubChem and Protein Data Bank respectively and docked using autodock4.0. Druglikeness and absorption, distribution, metabolism, excretion, and toxicity profile were predicted using Molsoft and admetSAR1 respectively. The multiple bioactives from T. cordifolia were identified to interact with multiple proteins involved in the pathogenesis of diabetes/obesity, i.e., isocolumbin (− 9 kcal/mol) with adiponectin (PDB: 4DOU), β-sitosterol (− 10.9 kcal/mol) with cholesteryl ester transfer protein (PDB: 2OBD), tinocordiside (− 6.9 kcal/mol) with lamin A/C (PDB: 3GEF), berberine (− 9.5 kcal/mol) with JNK1 (PDB:3ELJ), β-sitosterol & isocolumbin (− 10.1 kcal/mol) with peroxisome proliferator-activated receptor-γ (PDB:4CI5), berberine (− 7.5 kcal/mol) with suppressor of cytokine signaling 3 (PDB: 2BBU), isocolumbin (− 9.6 kcal/mol) with pancreatic α-amylase (PDB: 1B2Y), isocolumbin (− 9 kcal/mol) with α-glucosidase (PDB: 3TOP), and β-sitosterol (− 10.8 kcal/mol) with aldose reductase (PDB: 3RX2). Similarly, among the selected bioactives, tembetarine scored highest druglikeness score, i.e., 1.21. In contrast, isocolumbin scored lowest drug-likeness character i.e. − 0.52. The predicted result of phytochemicals from T. cordifolia for acute oral toxicity, rat acute toxicity, fish toxicity, drug-likeness score, and aqueous solubility showed the probability of lower side/adverse effects in human consumption. The study suggests processing for bioactives from T. cordifolia against diabetes and obesity via in-vitro and in-vivo approaches.



Similar content being viewed by others
Data availability
Data will be provided in case of a request.
References
Chatham JC, Blackband SJ (2001) Nuclear magnetic resonance spectroscopy and imaging in animal research. ILAR J 42(3):189–208. https://doi.org/10.1093/ilar.42.3.189
Dallinga-Thie GM, Dullaart RP, van Tol A (2007) Concerted actions of cholesteryl ester transfer protein and phospholipid transfer protein in type 2 diabetes: effects of apolipoproteins. Curr Opin Lipidol 18(3):251–257. https://doi.org/10.1097/MOL.0b013e3280e12685
de Souza PM, de Oliveira MP (2010) Application of microbial αamylase in industry—a review. Braz J Microbiol 41(4):850–861. https://doi.org/10.1590/S1517-83822010000400004
Duyu T, Khanal P, Khatib NA, Patil BM (2020) Mimosa pudica modulates neuroactive ligand-receptor interaction in Parkinson’s disease. Indian J Pharm Educ 54(3):732–739. https://doi.org/10.5530/ijper.54.3.124
Golay A, Ybarra J (2005) Link between obesity and type 2 diabetes. Best Pract Res Clin Endocrinol Metab 19(4):649–663. https://doi.org/10.1016/j.beem.2005.07.010
Government of India (2001) The Ayurvedic pharmacopoeia of India. Ministry Health Fam Welf 1:53
Halgren TA (1996) Merck molecular force field. I. Basis, form, scope, parameterization, and performance of MMFF94. J Comput Chem 17:490–519. https://doi.org/10.1002/(SICI)1096-987X(199604)17:5/6%3c490::AID-JCC1%3e3.0.CO;2-P
Kawano J, Arora R (2009) The role of adiponectin in obesity, diabetes, and cardiovascular disease. J Cardiometab Syndr 4(1):44–49. https://doi.org/10.1111/j.1559-4572.2008.00030.x
Kennedy T (1997) Managing the drug discovery/development interface. Drug Discov Today 2(10):436–444. https://doi.org/10.1016/S1359-6446(97)01099-4
Khanal P, Patil BM (2019) Gene set enrichment analysis of alpha-glucosidase inhibitors from Ficus benghalensis. Asian Pac J Trop Biomed 9(6):263–270. https://doi.org/10.4103/2221-1691.260399
Khanal P, Patil BM (2020a) Gene ontology enrichment analysis of α-amylase inhibitors from Duranta repens in diabetes mellitus. J Diabetes Metab Disord Available at: https://doi.org/10.1007/s40200-020-00554-9
Khanal P, Patil BM (2020b) α-Glucosidase inhibitors from Duranta repens modulate p53 signaling pathway in diabetes mellitus. Adv Tradit Med. https://doi.org/10.1007/s13596-020-00426-w
Khanal P, Mandar BK, Magadum P, Patil BM, Hullatti KK (2019a) In silico docking study of limonoids from Azadirachta indica with pfpk5: a novel target for Plasmodium falciparum. Indian J Pharm Sci 81(2):326–332
Khanal P, Mandar BK, Patil BM, Hullatti KK (2019b) In silico antidiabetic screening of borapetoside C, cordifolioside A and magnoflorine. Indian J Pharm Sci 81(3):550–555. https://doi.org/10.36468/pharmaceutical-sciences.543
Khanal P, Patil BM, Mandar BK, Dey YN, Duyu T (2019c) Network pharmacology-based assessment to elucidate the molecular mechanism ofanti-diabetic action of Tinospora cordifolia. Clin Phytosci 5:35. https://doi.org/10.1186/s40816-019-0131-1
Khanal P, Patil BM, Chand J, Naaz Y (2020) Anthraquinone derivatives as an immune booster and their therapeutic option against COVID-19. Nat Prod Bioprospect. https://doi.org/10.1007/s13659-020-00260-2
Kinoshita JH, Fukushi S, Kador P, Merola LO (1979) Aldose reductase in diabetic complications of the eye. Metabolism 28(4 Suppl 1):462–469. https://doi.org/10.1016/0026-0495(79)90057-x
Lipinski CA (2004) Lead- and drug-like compounds: the rule-of-five revolution. Drug Discov Today Technol 1(4):337–341. https://doi.org/10.1016/j.ddtec.2004.11.007
Mesa JL, Loos RJ, Franks PW et al (2007) Lamin A/C polymorphisms, type 2 diabetes, and the metabolic syndrome: case-control and quantitative trait studies. Diabetes 56(3):884–889. https://doi.org/10.2337/db06-1055
Morris GM, Huey R, Lindstrom W, Sanner MF, Belew RK, Goodsell DS, Olson AJ (2009) AutoDock4 and AutoDockTools4: automated docking with selective receptor flexibility. J Comput Chem 30:2785–2791. https://doi.org/10.1002/jcc.21256
Patel MB, Mishra S (2011) Hypoglycemic activity of alkaloidal fraction of Tinospora cordifolia. Phytomedicine 18(12):1045–1052. https://doi.org/10.1016/j.phymed.2011.05.006
Patil VS, Biradar PR, Attar V, Khanal P (2019) In silico docking analysis of active biomolecules from Cissus quadrangularis L. against PPAR-γ. Indian J Pharm Educ 53(3):S332–S3377. https://doi.org/10.5530/ijper.53.3s.103
Prabhu S, Vijayakumar S (2014) Antidiabetic, hypolipidemic and histopathological analysis of Gymnema sylvestre (R. Br) leaves extract on streptozotocin induced diabetic rats. Biomed Prev Nutr 4(3):425–430. https://doi.org/10.1016/j.bionut.2014.03.008
Puranik N, Kammar KF, Devi S (2010) Anti-diabetic activity of Tinospora cordifolia (Willd.) in streptozotocin diabetic rats; does it act like sulfonylureas? Turk J Med Sci 40(2):265–270. https://doi.org/10.3906/sag-0802-40
Ramírez NM, Toledo RCL, Moreira MEC et al (2017) Anti-obesity effects of tea from Mangifera indica L. leaves of the Ubá variety in high-fat diet-induced obese rats. Biomed Pharmacother 91:938–945. https://doi.org/10.1016/j.biopha.2017.05.015
Redinger RN (2007) The pathophysiology of obesity and its clinical manifestations. Gastroenterol Hepatol (N Y) 3(11):856–863. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3104148/pdf/GH-03-856.pdf
Reed AS, Unger EK, Olofsson LE, Piper ML, Myers MG Jr, Xu AW (2010) Functional role of suppressor of cytokine signaling 3 upregulation in hypothalamic leptin resistance and long-term energy homeostasis. Diabetes 59(4):894–906. https://doi.org/10.2337/db09-1024
Savjani KT, Gajjar AK, Savjani JK (2012) Drug solubility: importance and enhancement techniques. ISRN Pharm 2012:195727. https://doi.org/10.5402/2012/195727
Tyagi S, Gupta P, Saini AS, Kaushal C, Sharma S (2011) The peroxisome proliferator-activated receptor: a family of nuclear receptors role in various diseases. J Adv Pharm Technol Res 2(4):236–240. https://doi.org/10.4103/2231-4040.90879
WHO (1965) Diabetes mellitus. Report of a WHO expert committee. World Health Organization technical report series, pp 1–44. https://apps.who.int/iris/bitstream/handle/10665/38442/WHO_TRS_310.pdf;jsessionid=2C4D0847578CDB3E30D98B6B54F12241?sequence=1
Yang R, Trevillyan JM (2008) c-Jun N-terminal kinase pathways in diabetes. Int J Biochem Cell Biol 40(12):2702–2706. https://doi.org/10.1016/j.biocel.2008.06.012
Yano J, Yachandra VK (2009) X-ray absorption spectroscopy. Photosynth Res 102(2–3):241–254. https://doi.org/10.1007/s11120-009-9473-8
Zhang J, Onakpoya IJ, Posadzki P, Eddouks M (2015) The safety of herbal medicine: from prejudice to evidence. Evid Based Complement Altern Med 2015:316706. https://doi.org/10.1155/2015/316706
Acknowledgements
The authors are thankful to Principal KLE College of Pharmacy, Belagavi for providing necessary facilities and Head of Department of Pharmacology and Toxicology, KLE College of Pharmacy, Belagavi, for supporting to complete the work.
Funding
This work has not received any funds from national and international agencies.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
There are no conflicts of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mandar, B.K., Khanal, P., Patil, B.M. et al. In silico analysis of phytoconstituents from Tinospora cordifolia with targets related to diabetes and obesity. In Silico Pharmacol. 9, 3 (2021). https://doi.org/10.1007/s40203-020-00063-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s40203-020-00063-w