Abstract
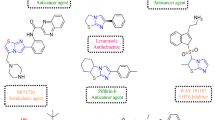
4-Amino-3-chloro-1H-pyrrole-2,5-dione derivatives were designed and synthesized as potential tyrosine kinase inhibitors. One of them has been shown to inhibit growth of cancer cell lines and in vivo tumors. To determine the impact of side groups on biological activity the ability of different 4-amino-3-chloro-1H-pyrrole-2,5-diones to interact with ATP-binding domains of growth factor receptors and with model cell membranes were aimed to be discovered. The methods of molecular docking, short-molecular dynamics (in silico) and non-steady cyclic current–voltage characteristics (in vitro) were used. Five 4-amino-3-chloro-1H-pyrrole-2,5-diones were synthesized from 3,4-dichloro-1H-pyrrole-2,5-diones. All of them demonstrated the potential ability to form complexes with ATP-binding domains of EGFR and VEGFR2. These complexes were more stable compared to those with ANP. 4-Amino-3-chloro-1H-pyrrole-2,5-diones while interact with different bilayer lipid membranes caused an increase of their specific conductance and electric capacity, demonstrating the certain disturbance in lipid packing. Obtained data allowed us to suggest that proposed chemicals can interact with the surface of lipid bilayer, do likely intercalate into the membrane and form stable complexes with EGFR and VEGFR2. So, the prospect of developed chemicals to be effective EGFR and VEGFR2 inhibitors and therefore realize antitumor activity was concluded.




Similar content being viewed by others
References
Arakelyan VB, Torosyan AL, Ghazaryan RK (2014) Cyclic current-voltage characteristics of a bilayer lipid membrane in the presence of porphyrin. Proc YSU Phys Mathcs 2:60–63
Bond SD, Leimkuhler BJ, Laird BB (1999) The Nosé-Poincaré method for constant temperature molecular dynamics. J Comput Phys 151:114–134
Broekman F, Giovannetti E, Peters GJ (2011) Tyrosine kinase inhibitors: multi-targeted or single-targeted? World J Clin Oncol 2:80–93
Bychko A, Artemenko A, Byelinska I, Gurnyak O, Rybalchenko V (2014) Membranotropic and cellular effects of maleimide derivatives as potential antitumor compounds. Visnyk of the Lviv University. Ser Biol 68:79–90
Di Maio M, Basch E, Bryce J, Perrone F (2016) Patient-reported outcomes in the evaluation of toxicity of anticancer treatments. Nat Rev Clin Oncol 13:319–325
Dubinina GG, Golovach SM, Kozlovsky VO, Tolmachov AO, Volovenko YuM (2007) Antiproliferative action of the new derivatives of l-(4-R-benzyl)-3-R1-4-(R2-phenylamino)-1H-pyrrol-2,5-dione. Zh Org Farm Khim 5:39–49
Elez E, Macarulla T, Tabernero J (2008) Handling side-effects of targeted therapies: safety of targeted therapies in solid tumours. Ann Oncol 19:vii146–vii152
Garcia-Celma JJ, Hatahet L, Kunz W, Fendler K (2007) Specific anion and cation binding to lipid membranes investigated on a solid supported membrane. Langmuir 23:10074–10080
Garmanchuk LV, Denis EO, Nikulina VV, Dzhus OI, Skachkova OV, Ribalchenko VK, Ostapchenko LI (2013a) MI1-derivative of maleimide inhibits progression of tumor cells of epithelial origin. Biopolym Cell 29:70–74
Garmanchuk LV, Linchak OV, Denis EO, Nikulina VV, Dzhus OI, Khranovskaya NN, Nikolaenko TV, Babuta EN, Rybalchenko VK (2013b) Potential cytostatic effect of the maleimide derivative 1-(4-Cl-benzyl)-3-Cl-4-(CF3-phenylamino)-1H-pyrrol-2,5-dione. Eksp Klin Farmakol 76:39–42
Gerber DE (2008) Targeted therapies: a new generation of cancer treatments. Am Fam Physician 77:311–319
Jorgensen WL, Chandrasekhar J, Madura JD, Impey RW, Klein ML (1983) Comparison of simple potential functions for simulating liquid water. J Chem Phys 79:926–935
Koper I, Stefan M, Schiller SM, Giess F, Naumann R, Knoll W (2006) Functional tethered bimolecular lipid membranes (tBLMs). In: Leitmannova Liu A (ed) Advances in planar lipid bilayers and liposomes. Elsevier, Amsterdam, pp 37–53
Kroschinsky F, Stölzel F, von Bonin S, Beutel G, Kochanek M, Kiehl M, Schellongowski P, Intensive Care in Hematological and Oncological Patients (iCHOP) Collaborative Group (2017) New drugs, new toxicities: severe side effects of modern targeted and immunotherapy of cancer and their management. Crit Care 14:1–11
Kurilovich SA, Kruchinina MV, Generalov VM, Bakirov TS, Rikhter VA, Semenov DV (2009) Electrical parameters and structure of membranes of erythrocytes at diffuse liver diseases. Russ J Gastroenterol Hepatol Coloproctol 19:30–36
Kuznietsova HM, Lynchak OV, Danylov MO, Kotlyar IP, Rybalchenko VK (2013) Effects of dihydropyrrol and maleimide derivatives on state of liver and colon in normal rats and in setting of colorectal carcinogenesis. Ukr Biochem J 85:62–72
Kuznietsova HM, Luzhenetska VK, Kotliar IP, Rybalchenko VK (2015) Effects of 5-Amyno-4-(1,3-benzothyazol-2-yn)-1-(3-methoxyphenyl)-1,2-dihydro-3H-pyrrol-3-one intake on digestive system in a rat model of colon cancer. Sci World J. https://doi.org/10.1155/2015/376576
Kuznietsova HM, Yena MS, Kotliar IP, Rybalchenko VK (2016) Antiinflammatory effects of protein kinase inhibitor pyrrol derivate. Sci World J. https://doi.org/10.1155/2016/2145753
Liu W, Beck J, Schmidt LC, Roolf C, Pews-Davtyan A, Rütgen BC, Hammer S, Willenbrock S, Sekora A, Rolfs A, Beller M, Brenig B, Nolte I, Junghanss C, Schütz E, Murua Escobar H (2016) Characterization of the novel indolylmaleimides’ PDA-66 and PDA-377 effect on canine lymphoma cells. Oncotarget 7:35379–35389
McMartin C, Bohacek RS (1997) QXP: powerful, rapid computer algorithms for structure-based drug design. J Comput Aided Mol Des 11:333–344
Nesterenko AM, Ermakov YuA (2012) Molecular-dynamic simulation of phospholipid bilayers: ion distribution at the surface of neutral and charged bilayer in the liquid crystalline state. Biochem Moscow Suppl Ser A 6:320–328
Ostrovska GV, Nizheradze KA, Dubinina GG, Rybalchenko VK (2007) Cytostatic action of maleimide derivatives on HEK-293 cell line. In: II Ukrainian cell biology society meeting (Kyiv), p 126
Pajak B, Orzechowska S, Gajkowska B, Orzechowski A (2008) Bisindolylmaleimides in anti-cancer therapy—more than PKC inhibitors. Adv Med Sci 53:21–31
Pestalozzi BC, Knuth A (2004) New antibodies in cancer treatment. Praxis 93:1589–1592
Relles MH (1973) Dichloromaleimide Chemistry. II. A thionyl chloride-pyridine method for the conversion of maleimides to dichloromaleimides. J Org Chem 37:3630–3637
Roskoski R (2019) Properties of FDA-approved small molecule protein kinase inhibitors. Pharmacol Res 144:19–50
Stewart BW, Wild CP (2014) World cancer report. International Agency for Research on Cancer, Lyon
Sturgeon JB, Laird BB (2000) Symplectic algorithm for constant-pressure molecular-dynamics using a Nose-Poincare thermostat. J Chem Phys 112:3474–3482
van Eis MJ, Evenou J, Schuler W, Zenke G, Vangrevelinghe E, Wagner J, von Matt P (2017) Indolyl-naphthyl-maleimides as potent and selective inhibitors of protein kinase C-α/β. Bioorg Med Chem Lett 27:781–786
Warren GL, Andrews CW, Capelli AM, Clarke B, LaLonde J, Lambert MH, Lindvall M, Nevins N, Semus SF, Senger S, Tedesco G, Wall ID, Woolven JM, Peishoff CE, Head MS (2006) A critical assessment of docking programs and scoring functions. J Med Chem 49:5912–5931
Acknowledgements
The research was carried out in the frame of the Fundamental research grants of Ministry of Education and Science of Ukraine No. 0118U000244 and No. 19BP07-03.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest regarding the publication of this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kuznietsova, H.M., Hurmach, V.V., Bychko, A.V. et al. Synthesis and biological activity of 4-amino-3-chloro-1H-pyrrole-2,5-diones. In Silico Pharmacol. 7, 2 (2019). https://doi.org/10.1007/s40203-019-0051-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s40203-019-0051-2