Abstract
Introduction
Agricultural commodities contaminated by molds and mycotoxins can be considered as public health problems in less developed countries, particularly in Iran. Hence the main purpose of this study was to identify mold fungi and molecular analysis of the most important species of aflatoxin-B1-producing Aspergillus species in some dried nuts and grains in local markets in Tehran.
Materials and methods
Two hundred fifty samples of wheat, rice, corn, pistachios, and peanuts were collected from the five different locations of Tehran between January 2018 and January 2019. The samples were analyzed by using direct seed inoculation method and grain crushing method. Fungal strains were identified as Aspergillus spp. on the basis of morphological characters and further confirmed by using of β-tubulin gene sequencing. To differentiate between aflatoxigenic and non-aflatoxigenic Aspergillus spp., the isolates were screened for the presence of aflatoxigenic genes (nor-1, ver-1, omtA, and aflR).
Results
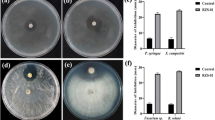
One-handed forty-eight aflatoxigenic Aspergillus isolates (144 A. flavus and 4 A. parasiticus) were identified and aflR gene was the most frequent gene in these species. Five isolates (4 A. flavus, 1 A. parasiticus) had quadruplet pattern, 64 isolates (63 A. flavus, 1 A. parasiticus) had more than 1 gene and 39 isolates (38 A. flavus,1 A. parasiticus) did not have any genes.
Conclusion
According to the contamination of dried nuts and grains by some aflatoxigenic fungi, an extensive surveillance is necessary to provide a wider view on these products. Moreover, effective and efficient aflatoxin control program requires identifying and managing key elements that are effective in reducing mycotoxin production at farm level or in storage conditions.


Similar content being viewed by others
Change history
19 October 2021
A Correction to this paper has been published: https://doi.org/10.1007/s40201-021-00745-3
References
Gholami-Shabani M, Shams-Ghahfarokhi M, Razzaghi-Abyaneh M. Aflatoxins and aflatoxigenic fungi in Iran: A systematic review of the past, present, and future. Mycol Iran. 2017;4(2):65–84.
Shanakht H, Ali Shahid A, Waseem Ali S. Characterization of fungal microbiota on rice grains from local markets of Lahore. J Hyg Eng Des. 2014;9:35–40.
Gherbawy YA, Elhariry HM, Bahobial AAS. Mycobiota and mycotoxins (aflatoxins and ochratoxin) associated with some Saudi date palm fruits. Foodborne Pathog Dis. 2012;9(6):561–7.
Schmidt-Heydt M, Rüfer CE, Abdel-Hadi A, Magan N, Geisen R. The production of aflatoxin B1 or G1 by Aspergillus parasiticus at various combinations of temperature and water activity is related to the ratio of aflS to aflR expression. Mycotoxin Res. 2010;26(4):241–6.
Iram W, Anjum T, Iqbal M, Ghaffar A, Abbas M. Structural eluci-dation and toxicity assessment of degraded products of aflatoxin B1and B2 by aqueous extracts of Trachyspermum ammi. Front Microbiol. 2016;7:346.
Akinola SA, Ateba CN, Mwanza M. Polyphasic assessment of aflatoxin production potential in selected aspergilli. Toxins (Basel). 2019;11(12):692.
Hamzeh Pour S, Mahmoudi S, Masoumi S, Rezaie S, Barac A, Ranjbaran M, et al. Aflatoxin M1 contamination level in Iranian milk and dairy products: A systematic review and meta-analysis. World Mycotoxin J. 2020;13;67–82. Wageningen Academic Publishers.
Sardiñas N, Vázquez C, Gil-Serna J, González-Jaén MT, Patiño B. Specific detection and quantification of Aspergillus flavus and Aspergillus parasiticus in wheat flour by SYBR® Green quantitative PCR. Int J Food Microbiol. 2011;145(1):121–5.
Chebon S, Wanyoike W, Bii C, Gathumbi J, Ogoyi D. Prevalence of aflatoxin biosynthesis genes according to aflatoxin levels in maize of different varieties in Kenya. Biotechnol J Int. 2017;19(2):1–21.
Hedayati MT, Khodavaisy S, Alialy M, Omran SM, Habibi MR. Invasive aspergillosis in intensive care unit patients in Iran. Acta Medica (Hradec Kralove). 2013;56(2):52-6
Yelton MM, Hamer JE, Timberlake WE. Transformation of Aspergillus nidulans by using a trpC plasmid. Proc Natl Acad Sci U S A. 1984;81:1470–1474.
Hussain A, Afzal A, Irfan M, Malik KA. Molecular detection of aflatoxin producing strains of aspergillus flavus from peanut (Arachis Hypogaea). Turkish J Agric - Food Sci Technol. 2015;3(5):335.
Khodavaisy S, Badali H, Hashemi S.J, et al. In vitro activities of five antifungal agents against 199 clinical and environmental isolates of Aspergillus flavus, an opportunistic fungal pathogen. Journal de Mycologie Médicale. 2016;26:116–21.
Degola F, Berni E, Dall’Asta C, et al. A multiplex RT-PCR approach to detect aflatoxigenic strains of Aspergillus flavus. J Appl Microbiol. 2007;103(2):409–17.
Gherbawy YA, Shebany YM, Hussein MA, Maghraby TA. Molecular detection of mycobiota and aflatoxin contamination of chili. Arch Biol Sci. 2015;67(1):223–34.
Manonmani HK, Anand S, Chandrashekar A, Rati ER. Detection of aflatoxigenic fungi in selected food commodities by PCR. 2005;40:2859–64.
Price MS, Yu J, Nierman WC, Kim HS, Pritchard B, Jacobus CA, et al. The aflatoxin pathway regulator AflR induces gene transcription inside and outside of the aflatoxin biosynthetic cluster. FEMS Microbiol Lett. 2006;255(2):275-9.
Lutfullah G, Hussain A. Studies on contamination level of aflatoxins in some cereals and beans of Pakistan. Food Control [Internet]. 2012;23(1):32–6. Available from: https://doi.org/10.1016/j.foodcont.2011.06.004.
Hedayati MT, Kaboli S, Mayahi S. Mycoflora of pistachio and peanut kernels from sari, iran. Jundishapur J Microbiol. 2010;3:114–20.
Khodavaisy S, Maleki A, Hossainzade B, Rezai S, Ahmadi F. Occurrence of fungal contamination in pistachio and peanut samples from retail shops in Sanandaj province, 2012;6(39):6781–4.
Rahimi P, Sharifnabi B, Bahar M. Detection of aflatoxin in Aspergillus species isolated from pistachio in Iran. J Phytopathol. 2008;156(1):15–20.
Furlung EB, Soares LM, Lasca CC, Kohara EY. Mycotoxins and fungi in wheat harvested during 1990 in test plots in the state of Sao Paulo, Brazil. Mycopathologia. 1995;131(3):185–90.
Criseo G, Bagnara A, Bisignano G. Differentiation of aflatoxin-producing and non-producing strains of Aspergillus flavus group. Lett Appl Microbiol. 2001;33(4):291–5.
Criseo G, Racco C, Romeo O. High genetic variability in non-aflatoxigenic A. flavus strains by using Quadruplex PCR-based assay. Int J Food Microbiol. 2008;125(3):341–3.
Okoth S, De Boevre M, Vidal A, Di Mavungu JD, Landschoot S, Kyallo M, et al. Genetic and toxigenic variability within Aspergillus flavus population isolated from maize in two diverse environments in Kenya. Front Microbiol. 2018;9(JAN):57.
Acknowledgements
This study was part of a M.S. thesis supported by Tehran University of Medical Sciences (grant No. 9411399003). We are grateful to the colleagues of Molecular Biology Laboratory of Medical Mycology, Tehran University of Medical Sciences (Mohammad Reza Safari and Azar Berahmeh) for their assistance in conducting this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There is no conflict of interest between authors in this study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised: "The original article has been updated"
Rights and permissions
About this article
Cite this article
Rahimi, A., Sasani, E., Rezaie, S. et al. Molecular identification of aflatoxigenic Aspergillus species in dried nuts and grains collected from Tehran, Iran. J Environ Health Sci Engineer 19, 1795–1799 (2021). https://doi.org/10.1007/s40201-021-00734-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40201-021-00734-6