Abstract
Introduction
Circulating levels of C1q/TNF-α‐related protein 6 (CTRP6) is an adipokine that is involved in regulation of glucose and lipid metabolism, inflammation, and insulin sensitivity. However, the exact role of CTRP6 in metabolic processes remains unclear due to conflicting findings. To address current gap, we aimed to investigate the serum levels of CTRP6 in patients with coronary artery disease (CAD) and its association with inflammatory cytokines.
Method
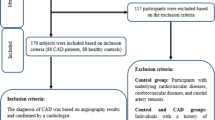
In this case-control study, the serum levels of CTRP6, interlukin-6 (IL-6), tumor necrosis factor- α (TNF-α), adiponectin, and fasting insulin were measured using enzyme-linked immunosorbent assay (ELISA) kits in a total of 176 participants, consisting of 88 CAD patients and 88 control subjects. Additionally, various anthropometric and biochemical measurements were measured and compared between cases and controls.
Results
The present study found that serum levels of CTRP6 were significantly higher in the CAD group (561.3 ± 15.14) compared to the control group (429.3 ± 12.85, p < 0.001). After adjusting for age, sex, and body mass index (BMI), CTRP6 levels were found to be positively associated with the risk of CAD (p < 0.001). Correlation analysis in CAD subjects revealed a positive correlation between CTRP6 levels and BMI, systolic blood pressure (SBP), malondialdehyde (MDA), TNF-α, and IL-6, as well as a negative correlation with creatinine and total anti-oxidant capacity.
Conclusion
The findings of this study provide novel evidence that elevated serum levels of CTRP6 are significantly associated with an increased risk of developing CAD. Moreover, our results indicate a correlation between CTRP6 and various risk factors for atherosclerosis.



Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study available from the corresponding author on reasonable request.
References
Liu L, Shi Z, Ji X, Zhang W, Luan J, Zahr T, et al. Adipokines, adiposity, and atherosclerosis. Cell Mol Life Sci. 2022;79(5):272. https://doi.org/10.1007/s00018-022-04286-2.
Bentzon JF, Otsuka F, Virmani R, Falk E. Mechanisms of plaque formation and rupture. Circ Res. 2014;114(12):1852–66. https://doi.org/10.1161/circresaha.114.302721.
Montarello NJ, Nguyen MT, Wong DTL, Nicholls SJ, Psaltis PJ. Inflammation in coronary atherosclerosis and its therapeutic implications. Cardiovasc Drugs Ther. 2022;36(2):347–62. https://doi.org/10.1007/s10557-020-07106-6.
Bai Y, Sun Q. Macrophage recruitment in obese adipose tissue. Obes Rev. 2015;16(2):127–36. https://doi.org/10.1111/obr.12242.
Maddux BA, See W, Lawrence JC Jr., Goldfine AL, Goldfine ID, Evans JL. Protection against oxidative stress-induced insulin resistance in rat L6 muscle cells by mircomolar concentrations of alpha-lipoic acid. Diabetes. 2001;50(2):404–10. https://doi.org/10.2337/diabetes.50.2.404.
Ceriello A, Motz E. Is oxidative stress the pathogenic mechanism underlying insulin resistance, diabetes, and cardiovascular disease? The common soil hypothesis revisited. Arterioscler Thromb Vasc Biol. 2004;24(5):816–23. https://doi.org/10.1161/01.Atv.0000122852.22604.78.
Zhou Y, Wei Y, Wang L, Wang X, Du X, Sun Z, et al. Decreased adiponectin and increased inflammation expression in epicardial adipose tissue in coronary artery disease. Cardiovasc Diabetol. 2011;10:2. https://doi.org/10.1186/1475-2840-10-2.
Kawano J, Arora R. The role of adiponectin in obesity, diabetes, and cardiovascular disease. J Cardiometab Syndr. 2009;4(1):44–9. https://doi.org/10.1111/j.1559-4572.2008.00030.x.
Zaidi H, Byrkjeland R, Njerve IU, Åkra S, Solheim S, Arnesen H, et al. Adiponectin in relation to exercise and physical performance in patients with type 2 diabetes and coronary artery disease. Adipocyte. 2021;10(1):612–20. https://doi.org/10.1080/21623945.2021.1996699.
Fadaei R, Moradi N, Kazemi T, Chamani E, Azdaki N, Moezibady SA, et al. Decreased serum levels of CTRP12/adipolin in patients with coronary artery disease in relation to inflammatory cytokines and insulin resistance. Cytokine. 2019;113:326–31. https://doi.org/10.1016/j.cyto.2018.09.019.
Schäffler A, Buechler C. CTRP family: linking immunity to metabolism. Trends Endocrinol Metab. 2012;23(4):194–204. https://doi.org/10.1016/j.tem.2011.12.003.
Lei X, Seldin MM, Little HC, Choy N, Klonisch T, Wong GW. C1q/TNF-related protein 6 (CTRP6) links obesity to adipose tissue inflammation and insulin resistance. J Biol Chem. 2017;292(36):14836–50. https://doi.org/10.1074/jbc.M116.766808.
Wang M, Tang X, Li L, Liu D, Liu H, Zheng H, et al. C1q/TNF-related protein-6 is associated with insulin resistance and the development of diabetes in Chinese population. Acta Diabetol. 2018;55(12):1221–9. https://doi.org/10.1007/s00592-018-1203-2.
Zheng WF, Zhang SY, Ma HF, Chang XW, Wang H. C1qTNF-related protein-6 protects against doxorubicin-induced cardiac injury. J Cell Biochem. 2019;120(6):10748–55. https://doi.org/10.1002/jcb.28366.
Murayama MA, Kakuta S, Inoue A, Umeda N, Yonezawa T, Maruhashi T, et al. CTRP6 is an endogenous complement regulator that can effectively treat induced arthritis. Nat Commun. 2015;6(1):1–12.
Kim MJ, Lee W, Park EJ, Park SY. C1qTNF-related protein-6 increases the expression of interleukin-10 in macrophages. Mol Cells. 2010;30(1):59–64. https://doi.org/10.1007/s10059-010-0088-x.
Lei H, Wu D, Wang J-Y, Li L, Zhang C-L, Feng H, et al. C1q/tumor necrosis factor-related protein-6 attenuates post-infarct cardiac fibrosis by targeting RhoA/MRTF-A pathway and inhibiting myofibroblast differentiation. Basic Res Cardiol. 2015;110(4):35. https://doi.org/10.1007/s00395-015-0492-7.
Wei C, Liu Y, Xing E, Ding Z, Tian Y, Zhao Z, et al. Association between Novel Pro- and Anti- Inflammatory adipocytokines in patients with Acute Coronary Syndrome. Clin Appl Thromb Hemost. 2022;28:10760296221128021. https://doi.org/10.1177/10760296221128021.
Emamgholipour S, Moradi N, Beigy M, Shabani P, Fadaei R, Poustchi H, et al. The association of circulating levels of complement-C1q TNF-related protein 5 (CTRP5) with nonalcoholic fatty liver disease and type 2 diabetes: a case-control study. Diabetol Metab Syndr. 2015;7:108. https://doi.org/10.1186/s13098-015-0099-z.
Fadaei R, Moradi N, Baratchian M, Aghajani H, Malek M, Fazaeli AA, et al. Association of C1q/TNF-Related Protein-3 (CTRP3) and CTRP13 serum levels with coronary artery disease in subjects with and without type 2 diabetes Mellitus. PLoS ONE. 2016;11(12):e0168773. https://doi.org/10.1371/journal.pone.0168773.
Bai B, Ban B, Liu Z, Zhang MM, Tan BK, Chen J. Circulating C1q complement/TNF-related protein (CTRP) 1, CTRP9, CTRP12 and CTRP13 concentrations in type 2 diabetes mellitus: in vivo regulation by glucose. PLoS ONE. 2017;12(2):e0172271.
Seldin MM, Tan SY, Wong GW. Metabolic function of the CTRP family of hormones. Reviews Endocr Metabolic Disorders. 2014;15(2):111–23. https://doi.org/10.1007/s11154-013-9255-7.
Lu L, Zhang RY, Wang XQ, Liu ZH, Shen Y, Ding FH, et al. C1q/TNF-related protein-1: an adipokine marking and promoting atherosclerosis. Eur Heart J. 2016;37(22):1762–71.
Moradi N, Fadaei R, Emamgholipour S, Kazemian E, Panahi G, Vahedi S, et al. Association of circulating CTRP9 with soluble adhesion molecules and inflammatory markers in patients with type 2 diabetes mellitus and coronary artery disease. PLoS ONE. 2018;13(1):e0192159.
Wong GW, Krawczyk SA, Kitidis-Mitrokostas C, Revett T, Gimeno R, Lodish HF. Molecular, biochemical and functional characterizations of C1q/TNF family members: adipose-tissue-selective expression patterns, regulation by PPAR-gamma agonist, cysteine-mediated oligomerizations, combinatorial associations and metabolic functions. Biochem J. 2008;416(2):161–77. https://doi.org/10.1042/bj20081240.
Xu E, Yin C, Yi X, Liu Y. Knockdown of CTRP6 inhibits high glucose-induced oxidative stress, inflammation and extracellular matrix accumulation in mesangial cells through regulating the Akt/NF‐κB pathway. Clin Exp Pharmacol Physiol. 2020;47(7):1203–11.
Lee W, Kim MJ, Park EJ, Choi YJ, Park SY. C1qTNF-related protein-6 mediates fatty acid oxidation via the activation of the AMP-activated protein kinase. FEBS Lett. 2010;584(5):968–72. https://doi.org/10.1016/j.febslet.2010.01.040.
Sadeghi A, Fadaei R, Moradi N, Fouani FZ, Roozbehkia M, Zandieh Z et al. Circulating levels of C1q/TNF-α-related protein 6 (CTRP6) in polycystic ovary syndrome. 2020;72(7):1449–59; https://doi.org/10.1002/iub.2272.
Liao X, Liu S, Tang X, Yang D, Liu H, Gao L, et al. Circulating CTRP6 levels are increased in overweight or obese Chinese individuals and Associated with insulin resistance parameters: a pilot study. Experimental and clinical endocrinology & diabetes: official journal. German Soc Endocrinol [and] German Diabetes Association. 2021;129(7):535–41. https://doi.org/10.1055/a-0929-6072.
Wu WJ, Mo DL, Zhao CZ, Zhao C, Chen YS, Pang WJ, et al. Knockdown of CTRP6 inhibits adipogenesis via lipogenic marker genes and Erk1/2 signalling pathway. Cell Biol Int. 2015;39(5):554–62. https://doi.org/10.1002/cbin.10422.
Wu W, Sun Y, Zhao C, Zhao C, Chen X, Wang G, et al. Lipogenesis in myoblasts and its regulation of CTRP6 by AdipoR1/Erk/PPARγ signaling pathway. Acta Biochim Biophys Sin. 2016;48(6):509–19.
Li Y, Sun J, Gu L, Gao X. Protective effect of CTRP6 on cerebral ischemia/reperfusion injury by attenuating inflammation, oxidative stress and apoptosis in PC12 cells. Mol Med Rep. 2020;22(1):344–52. https://doi.org/10.3892/mmr.2020.11108.
Xu E, Yin C, Yi X, Liu Y. Knockdown of CTRP6 inhibits high glucose-induced oxidative stress, inflammation and extracellular matrix accumulation in mesangial cells through regulating the Akt/NF-κB pathway. Clin Exp Pharmacol Physiol. 2020;47(7):1203–11. https://doi.org/10.1111/1440-1681.13289.
Aronson D, Edelman ER. Coronary artery disease and diabetes mellitus. Cardiol Clin. 2014;32(3):439–55.
Xu J, Zou MH. Molecular insights and therapeutic targets for diabetic endothelial dysfunction. Circulation. 2009;120(13):1266–86. https://doi.org/10.1161/circulationaha.108.835223.
Chi L, Hu X, Zhang W, Bai T, Zhang L, Zeng H, et al. Adipokine CTRP6 improves PPARγ activation to alleviate angiotensin II-induced hypertension and vascular endothelial dysfunction in spontaneously hypertensive rats. Biochem Biophys Res Commun. 2017;482(4):727–34. https://doi.org/10.1016/j.bbrc.2016.11.102.
Acknowledgements
Tehran University of Medical Sciences (TUMS), Tehran, Iran, (grant number: 59099) supported this project. We thank all the patients and individuals participating in the study.
Funding
Tehran University of Medical Sciences (TUMS), Tehran, Iran, (grant number: 59099) supported this project.
Author information
Authors and Affiliations
Contributions
Design: Nariman Moradi, Reza Fadaei. Data curation: Ali Abbasi, Seyed abdolhossein Tabatabaei. Formal analysis and Validation: Reza Fadaei. Investigation: Nariman Moradi. Frood Malekshahi, Arash Falahat. Methodology: Nariman Moradi, Akram Vatannejad, Maryam Mirahmad. Project administration: Ali Abbasi, Seyed abdolhosein Tabatabaei. Supervision and Resources: Ali Abbasi. Writing? original draft: Vida Farrokhi, Maryam Mirahmad, Reza Afrisham, Writing? review & editing: Reza Afrisham, Reza Fadaei, Ali Abbasi, Nariman Moradi. Frood Malekshahi, Arash Falahat.
Corresponding author
Ethics declarations
Ethical approval and consent to participate
This investigation was performed according to the Helsinki Declaration as well as was approved by the Ethics Committee of Tehran University of Medical Sciences (IR.TUMS.SHARIATI.REC.1401.017). All study contributors signed forms of written and informed consent.
Consent for publication
Not applicable.
Competing interests
The authors announce that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tabatabaei, S.a., Fadaei, R., Moradi, N. et al. Circulating levels of C1q/TNF-α‐related protein 6 (CTRP6) in coronary artery disease and its correlation with inflammatory markers. J Diabetes Metab Disord (2024). https://doi.org/10.1007/s40200-024-01415-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s40200-024-01415-5