Abstract
Purpose
Emerging of miRNAs have illustrated the new mechanistic layer to regulate type 2 diabetes process and suggests a possible role of these RNAs in this defect. Thus, we designed this study to improve our understanding of salivary miRNA-126 and 135a expression utility as an easy of collection and non-invasive way in diabetic patients instead of blood sample.
Methods
This case-control study was done on T2D (n = 40) and healthy individuals (n = 40). The level of biochemical parameters were determined by enzymatic methods as well as glycosylated hemoglobin (HbA1c) was measured by immunoturbidimetry. We used the pooled whole stimulated saliva sample from cases and controls to assess the differentiation expression of miRNA 126 and 135-a with quantitative RT-PCR method. Unpaired Student’s t test, Pearson’s correlation coefficient and Receiver Operating Characteristic (ROC) analysis were used.
Results
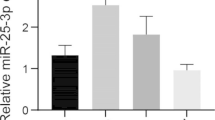
A correlation was observed between the level of HbA1c, glucose and lipid profiles (TG, TC, and LDL) in serum and whole stimulated saliva samples in T2D patients compared to control (p < 0.001). miR-135a expression was considerably higher by 4.7-fold in T2D compared to the control group (1.8-fold) (p < 0.001) while the miR126 expression was significantly decreased by 3.9-fold in T2D compared to the controls (6.3-fold) (p < 0.001).
Conclusions
The results of this case and control study showed that miR-135a and miR126 expression in saliva fluid as a reliable biomarkers and non-invasive approach in combination by change of lipid profiles, glucose and HbA1c may be used to monitor diabetic and non-diabetic patients, while further research is needed to investigate the relationship of these salivary miRNAs (miR135a, miR126) levels change on shifting the levels of clinical laboratory outcomes.
Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Franklin O, et al. Plasma micro-RNA alterations appear late in pancreatic cancer. Ann Surg. 2018;267(4):775.
Fehlmann T, et al. Distribution of microRNA biomarker candidates in solid tissues and body fluids. RNA Biol. 2016;13(11):1084–8.
Agha-Hosseini F, et al. Mucin 5B in saliva and serum of patients with oral lichen planus. Sci Rep. 2017;7(1):1–6.
Hsu Y-L, et al. Bone-marrow-derived cell-released extracellular vesicle miR-92a regulates hepatic pre-metastatic niche in lung cancer. Oncogene. 2020;39(4):739–53.
Kumar D, et al. Circulatory miR-133b and miR-21 as novel biomarkers in early prediction and diagnosis of coronary artery disease. Genes. 2020;11(2):164.
Mei J, et al. Long non-coding RNA NNT-AS1 regulates proliferation, apoptosis, inflammation and airway remodeling of chronic obstructive pulmonary disease via targeting miR-582-5p/FBXO11 axis. Biomed Pharmacother. 2020;129:110326.
Yang LG, et al. LncRNA XIST modulates HIF-1A/AXL signaling pathway by inhibiting miR-93-5p in colorectal cancer. Mol Genet Genom Med. 2020;8(4):e1112.
Alberti K, Zimmet P. New diagnostic criteria and classification of diabetes—again? 1998, Wiley Online Library.
Al-Muhtaresh HA, Al-Kafaji G. Evaluation of two-diabetes related microRNAs suitability as earlier blood biomarkers for detecting prediabetes and type 2 diabetes mellitus. J Clin Med. 2018;7(2):12.
Nielsen LB, et al. Circulating levels of microRNA from children with newly diagnosed type 1 diabetes and healthy controls: evidence that miR-25 associates to residual beta-cell function and glycaemic control during disease progression. Exp Diabetes Res. 2012;2012.
Poy MN, et al. A pancreatic islet-specific microRNA regulates insulin secretion. Nature. 2004;432(7014):226–30.
Zampetaki A, Mayr M. MicroRNAs in vascular and metabolic disease. Circ Res. 2012;110(3):508–22.
Dahlmans D, et al. Evaluation of muscle microRNA expression in relation to human peripheral insulin sensitivity: a cross-sectional study in metabolically distinct subject groups. Front Physiol. 2017;8:711.
Agha-Hosseini F, Mirzaii-Dizgah I, Mirjalili N. Relationship of unstimulated saliva cortisol level with severity of oral dryness feeling in menopausal women. Aust Dent J. 2011 Jun;56(2):171–4. https://doi.org/10.1111/j.1834-7819.2011.01320.x.
Agha-Hosseini F, Mirzaii-Dizgah I, Mirjalili N. Relationship of stimulated whole saliva cortisol level with the severity of a feeling of dry mouth in menopausal women. Gerodontology. 2012 Mar;29(1):43–7. https://doi.org/10.1111/j.1741-2358.2010.00403.x.
Di Pietro V, et al. Salivary MicroRNAs: diagnostic markers of mild traumatic brain injury in contact-sport. Front Mol Neurosci. 2018;11:290.
Humeau M, et al. Salivary microRNA in pancreatic cancer patients. PLoS One. 2015;10(6):e0130996.
Li F, et al. Discovery and validation of salivary extracellular RNA biomarkers for noninvasive detection of gastric cancer. Clin Chem. 2018;64(10):1513–21.
Hicks SD, et al. Overlapping microRNA expression in saliva and cerebrospinal fluid accurately identifies pediatric traumatic brain injury. J Neurotrauma. 2018;35(1):64–72.
Zhang X, et al. Alterations in miRNA processing and expression in pleomorphic adenomas of the salivary gland. Int J Cancer. 2009;124(12):2855–63.
Wiegand C, et al. Stress-associated changes in salivary microRNAs can be detected in response to the Trier social stress test: an exploratory study. Sci Rep. 2018;8(1):1–13.
Agarwal P, et al. miR-135a targets IRS2 and regulates insulin signaling and glucose uptake in the diabetic gastrocnemius skeletal muscle. Biochimica et Biophysica Acta (BBA)-Molecular Basis of Disease. 2013;1832(8):1294–303.
Amr K, et al. Potential value of circulating microRNA-126 and microRNA-210 as biomarkers for type 2 diabetes with coronary artery disease. Br J Biomed Sci. 2018;75(2):82–7.
He F, et al. MiR-135a promotes renal fibrosis in diabetic nephropathy by regulating TRPC1. Diabetologia. 2014;57(8):1726–36.
Honardoost M, et al. Development of insulin resistance through induction of miRNA-135 in C2C12 cells. Cell Journal (Yakhteh). 2016;18(3):353.
Honardoost M, et al. Expression change of miR-214 and miR-135 during muscle differentiation. Cell Journal (Yakhteh). 2015;17(3):461.
Liu Y, et al. The role of circulating microRNA-126 (miR-126): a novel biomarker for screening prediabetes and newly diagnosed type 2 diabetes mellitus. Int J Mol Sci. 2014;15(6):10567–77.
Sarookhani MR, Honardoost M, Foroughi F. Plasma miR-135a; a potential biomarker for diagnosis of new type 2 diabetes (T2DM). Bali Medical Journal. 2018.
Alberti KGMM, Zimmet PZ. Definition, diagnosis and classification of diabetes mellitus and its complications. Part 1: diagnosis and classification of diabetes mellitus. Provisional report of a WHO consultation. Diabet Med. 1998;15(7):539–53.
Kumar A, et al. Approach to sample size calculation in medical research. Curr Med Res Pract. 2014;4(2):87–92.
Li-Hui W, et al. Gender differences in the saliva of young healthy subjects before and after citric acid stimulation. Clin Chim Acta. 2016;460:142–5. https://doi.org/10.1016/j.cca.2016.06.040.
Gupta S, et al. Comparison of salivary and serum glucose levels in diabetic patients. J Diabetes Sci Technol. 2014;9(1):91–6.
Puttaswamy KA, Puttabudhi JH, Raju S. Correlation between salivary glucose and blood glucose and the implications of salivary factors on the oral health status in type 2 diabetes mellitus patients. Journal of International Society of Preventive & Community Dentistry. 2017;7(1):28.
Machida T, et al. miR-1246 and miR-4644 in salivary exosome as potential biomarkers for pancreatobiliary tract cancer. Oncol Rep. 2016;36(4):2375–81.
Fichtlscherer S, Zeiher AM, Dimmeler S. Circulating microRNAs: biomarkers or mediators of cardiovascular diseases? Arterioscler Thromb Vasc Biol. 2011;31(11):2383–90.
Zernecke A, et al. Delivery of microRNA-126 by apoptotic bodies induces CXCL12-dependent vascular protection. Science Signaling. 2009;2(100):ra81.
Qian Z-K, et al. Alisma orientalis (Sam.) juzep polysaccharide-regulated glucose-lipid metabolism in experimental rats and cell model of diabetes mellitus with regulation of miR-126. Pharmacognosy Magazine. 2019;15(65):652.
Al-Kafaji G, et al. Circulating endothelium-enriched microRNA-126 as a potential biomarker for coronary artery disease in type 2 diabetes mellitus patients. Biomarkers. 2017;22(3–4):268–78.
Al-Kafaji G, et al. Decreased expression of circulating microRNA-126 in patients with type 2 diabetic nephropathy: a potential blood-based biomarker. Exp Ther Med. 2016;12(2):815–22.
Bijkerk R, et al. Circulating microRNAs associate with diabetic nephropathy and systemic microvascular damage and normalize after simultaneous pancreas–kidney transplantation. Am J Transplant. 2015;15(4):1081–90.
Liu Y, et al. Stability of miR-126 in urine and its potential as a biomarker for renal endothelial injury with diabetic nephropathy. Int J Endocrinol. 2014;2014.
Ye P, et al. Hypoxia-induced deregulation of miR-126 and its regulative effect on VEGF and MMP-9 expression. Int J Med Sci. 2014;11(1):17.
Bagi Z, et al. Type 2 diabetic mice have increased arteriolar tone and blood pressure: enhanced release of COX-2–derived constrictor prostaglandins. Arterioscler Thromb Vasc Biol. 2005;25(8):1610–6.
Lu X, et al. MiR-135a promotes inflammatory responses of vascular smooth muscle cells from db/db mice via downregulation of FOXO1. Int Heart J. 2018;59(1):170–9.
Guo Z, et al. COX-2 up-regulation and vascular smooth muscle contractile hyperreactivity in spontaneous diabetic db/db mice. Cardiovasc Res. 2005;67(4):723–35.
Farnós O, et al. Rapid high-yield production of functional sars-cov-2 receptor binding domain by viral and non-viral transient expression for pre-clinical evaluation. Vaccines. 2020;8(4):1–20.
Al-Rawi NH, et al. Salivary microRNA 155, 146a/b and 203: a pilot study for potentially non-invasive diagnostic biomarkers of periodontitis and diabetes mellitus. PLoS One. 2020;15(8):e0237004.
Sun Y, et al. Comparative proteomic analysis of exosomes and microvesicles in human saliva for lung cancer. J Proteome Res. 2018;17(3):1101–7.
Acknowledgements
We would like to thank the clinical staff of pathobiology laboratory in Pasteur Hospital (Qazvin province, Iran) for their technical assistance.
Funding
This study was funded by Aja University of Medical Sciences and Qazvin University of Medical Sciences.
Author information
Authors and Affiliations
Contributions
Conceptualization and methodology: Y.K.M., I.M.D., and MR.M.D. Material preparation: E.K., and MR.S; Investigation: Y.K.M., I.M.D. and S.H..; Data collection: E.K., Y.K.M., and S.H.; Writing—original draft preparation: Y.K.M., I.M.D., Writing—review and editing: Y.K.M., I.M.D., MR.M.D. And MR.S.
Corresponding author
Ethics declarations
Conflict of interest
Authors declare that they have no conflict of interest.
Ethical approval
The current study was carried out according to Helsinki Declaration and was approved by the ethics committee of AJA University of Medical Sciences (IR.AJAUMS.REC.1397.056).
Informed consent
Informed consent was obtained from all individual participants included in the study.
Study limitations
The most important limitation in writing our manuscript was that we could not find reports in regard to the assessment of the expression level of Micro RNAs in saliva samples as biomarkers in diabetes.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Monfared, Y.K., Mirzaii-Dizgah, MR., Khodabandehloo, E. et al. Salivary microRNA-126 and 135a: a potentially non-invasive diagnostic biomarkers of type- 2 diabetes. J Diabetes Metab Disord 20, 1631–1638 (2021). https://doi.org/10.1007/s40200-021-00914-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40200-021-00914-z