Abstract
Background
A number of studies were carried out to assess the association of angiotensin I converting enzyme (ACE) I/D and plasminogen activator inhibitor-1 (PAI-1-1) 4G/5G polymorphisms with susceptibility to type 2 diabetes mellitus (T2DM). However, there are a few studies in Iranian patients with T2DM. Here, we tested for an association of ACE I/D and PAI-1 4G/5G polymorphisms with T2DM risk.
Methods
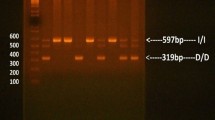
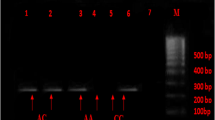
One hundred-eighteen patients with T2DM and 125 healthy subjects were participates in this study. The ACE I/D (rs4340) and PAI-1 4G/5G (rs1799889) polymorphisms was genotyped by conventional and PCR–RFLP assays, receptively. The associations was evaluated by calculating the odds ratio (OR) and 95% confidence interval (95% CI).
Results
The genotype distribution of ACE I/D and PAI-1 4G/5G polymorphisms were not deviated from the Hardy–Weinberg equilibrium in healthy controls. The ACE II, ID, and DD genotype frequencies were 18.6%, 48.3%, and 33.1% in the T2DM patients versus 24.0%, 45.6% and 30.4% in healthy subjects, respectively. The PAI-1 4G/4G, 4G/5G, and 5G/5G genotype frequencies were 16.9%, 51.7%, and 31.4% in cases versus 24.8%, 57.6% and 17.6% in controls, respectively. There is a significant distribution in genotype/allele of PAI-1 4G/4G between cases with T2DM and healthy control, but not for ACE I/D. Moreover, the 5G/5G genotype is significantly (OR = 2.139, CI 95% 1.171–3.907, p = 0.013) increased the risk of T2DM by two folds in the cases than healthy controls.
Conclusions
Our findings suggest that PAI-1 4G/5G may be likelihood risk factor for the development of T2DM in the Iranian patients. The higher frequency of PAI-1 5G/5G genotype in patients with T2DM revealed that individuals with the 5G allele may be at higher risk of T2DM development than those with 4G. However, there was no significant association between ACE I/D polymorphism and T2DM in our population. Future rigorous, well-designed studies with larger sample should replicate this study to confirm our findings in Iranian T2DM patients.
Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during this study are the corresponding author on reasonable request.
References
Dendup T, Feng X, Clingan S, Astell-Burt T. Environmental risk factors for developing type 2 diabetes mellitus: a systematic review. Int J Environ Res Public Health. 2018;15:78.
Roshanravan N, Alamdari NM, Jafarabadi MA, Mohammadi A, Shabestari BR, Nasirzadeh N, et al. Effects of oral butyrate and inulin supplementation on inflammation-induced pyroptosis pathway in type 2 diabetes: a randomized, double-blind, placebo-controlled trial. Cytokine. 2020;131:155101.
Li S, Wang J, Zhang B, Li X, Liu Y. Diabetes mellitus and cause-specific mortality: a population-based study. Diabetes Metab J. 2019;43:319–41.
Khan MAB, Hashim MJ, King JK, Govender RD, Mustafa H, Al-Kaabi J. Epidemiology of Type 2 diabetes - global burden of disease and forecasted trends. J Epidemiol Glob Health. 2020;10:107–11. Atlantis Press International.
Razmpoosh E, Safi S, Mazaheri M, Salehi-Abargouei A, Abdollahi N, Nazari M, et al. Effects of oral Nigella sativa oil on the expression levels and serum concentrations of adiponectin, PPAR-γ, and TNF-α in overweight and obese women: a study protocol for a crossover-designed, double-blind, placebo-controlled randomized clinical trial. Trials. 2019;20:512. BioMed Central Ltd.
Lin X, Xu Y, Pan X, Xu J, Ding Y, Sun X, et al. Global, regional, and national burden and trend of diabetes in 195 countries and territories: an analysis from 1990 to 2025. Scientific Reports. 2020;10:14790. Nature Research.
Murea M, Ma L, Freedman BI. Genetic and environmental factors associated with type 2 diabetes and diabetic vascular complications. Rev Diabet Stud. 2012;9:6–22.
Galicia-Garcia U, Benito-Vicente A, Jebari S, Larrea-Sebal A, Siddiqi H, Uribe KB, et al. Pathophysiology of type 2 diabetes mellitus. Int J Mol Sci. 2020;21:1–34. MDPI AG.
Xue A, Wu Y, Zhu Z, Zhang F, Kemper KE, Zheng Z, et al. Genome-wide association analyses identify 143 risk variants and putative regulatory mechanisms for type 2 diabetes. Nat Commun. 2018;9:2941. Nature Publishing Group.
Mirjalili H, Dastgheib SA, Shaker SH, Bahrami R, Mazaheri M, Sadr-Bafghi SMH, et al. Proportion and mortality of Iranian diabetes mellitus, chronic kidney disease, hypertension and cardiovascular disease patients with COVID-19: a meta-analysis. J Diabetes Metab Disord. 2021;20:1–13.
Hou G, Jin Y, Liu M, Wang C, Song G. UCP2–866G/A polymorphism is associated with prediabetes and type 2 diabetes. Arch Med Res. 2020;51:556–63. Elsevier Inc.
Ahmed SAH, Ansari SA, Mensah-Brown EPK, Emerald BS. The role of DNA methylation in the pathogenesis of type 2 diabetes mellitus. Clini Epigenetics. 2020;12:104. BioMed Central.
Mobasseri M, Ostadrahimi A, Tajaddini A, Asghari S, Barati M, Akbarzadeh M, et al. Effects of saffron supplementation on glycemia and inflammation in patients with type 2 diabetes mellitus: a randomized double-blind, placebo-controlled clinical trial study. Diabetes Metab Syndr Clin Res Rev. 2020;14:527–34. Elsevier Ltd.
Saleem S, Azam A, Maqsood SI, Muslim I, Bashir S, Fazal N, et al. Role of ACE and PAI-1 polymorphisms in the development and progression of diabetic retinopathy. Ljubimov A V., editor. PLOS ONE. 2015;10:e0144557.
Moghimi M, Kargar S, Jafari MA, Ahrar H, Jarahzadeh MH, Neamatzadeh H, et al. Angiotensin converting enzyme insertion/deletion polymorphism is associated with breast cancer risk: a meta-analysis. Asian Pac J Cancer Prev. 2018;19:3225–31.
Aslbahar F, Neamatzadeh H, Tabatabaiee R, Karimi-Zarchi M, Javaheri A, Mazaheri M, et al. Association of angiotensin-converting enzyme insertion/deletion polymorphism with recurrent pregnancy loss: a meta-analysis of 26 case-control studies. Rev Bras Ginecol Obstet. 2018;40:631–41.
Kamali M, Hantoushzadeh S, Borna S, Neamatzadeh H, Mazaheri M, Noori-Shadkam M, et al. Association between thrombophilic genes polymorphisms and recurrent pregnancy loss susceptibility in the Iranian population: a systematic review and meta-analysis. Iran Biomed J. 2018;22:78–89. Pasteur Institute of Iran.
Sobhan MR, Mahdinezhad-Yazdi M, Moghimi M, Aghili K, Jafari M, Zare-Shehneh M, et al. Plasminogen Activator Inhibitor-1 4G/5G polymorphism contributes to osteonecrosis of the femoral head susceptibility: evidence from a systematic review and meta-analysis. Arch Bone Jt Surg. 2018;6:468–77.
Al-Rubeaan K, Siddiqui K, Saeb ATM, Nazir N, Al-Naqeb D, Al-Qasim S. ACE I/D and MTHFR C677T polymorphisms are significantly associated with type 2 diabetes in Arab ethnicity: a meta-analysis. Gene Gene. 2013;520:166–77.
Gohari M, Dastgheib SA, Noorishadkam M, Lookzadeh MH, Mirjalili SR, Akbarian-Bafghi MJ, et al. Association of eNOS and ACE polymorphisms with retinopathy of prematurity: a systematic review and meta-analysis. Fetal Pediatr Pathol. 2020;39:334–45.
Luo S, Shi C, Wang F, Wu Z. Association between the angiotensin-converting enzyme (ACE) genetic polymorphism and diabetic retinopathy-a meta-analysis comprising 10,168 subjects. Int J Environ Res Public Health. 2016;13:1142.
Mahwish U, Ponnaluri K, Heera B, Alavala S, Devi KR, Raju S, et al. Link between ACE I/D gene polymorphism and dyslipidemia in diabetic nephropathy: a case-control study from Hyderabad, India. Indian J Nephrol. 2020;30:77.
Yildiz M, Karkucak M, Yakut T, Gorukmez O, Ozmen A. Lack of association of genetic polymorphisms of angiotensin-converting enzyme gene I/D and glutathione-S-transferase enzyme T1 and M1 with retinopathy of prematures. Genet Mol Res. 2010;9:2138–46.
Rouhi N, Nazm SA, Bonyadi M, JabbarpoorBonyadi MH, Soheilian M. Angiotensin-converting enzyme gene polymorphism in Behçet’s disease in Iranian population [Internet]. Ophthalmic Genet. 2019;40:388–9. Taylor and Francis Ltd.
Jhawat V, Gupta S, Agarwal BK, Roy P, Saini V. Angiotensin converting enzyme gene insertion/deletion polymorphism is not responsible for antihypertensive therapy induced new onset of type 2 diabetes in essential hypertension. Clin Med Insights: Endocrinol Diabetes. 2019;12:1179551418825037.
Khalaf FA, Ibrahim HR, Bedair HM, Allam MM, Elshormilisy AA, Ali ST, et al. Plasminogen activator inhibitor-1 gene polymorphism as a risk factor for vascular complications in type 2 diabetes mellitus. Egypt J Med Hum Genet. 2019;20:18. Springer.
Gao W, Guo Y, Bai Y, Ding X, Yan Y, Wu Z. Association between PAI-1 4G/5G polymorphism and diabetic nephropathy: a meta-analysis in the Chinese population. Int Urol Nephrol. 2016;48:1483–9.
Jafari-Nedooshan J, Dastgheib SA, Kargar S, Zare M, Raee-Ezzabadi A, Heiranizadeh N, et al. Association of IL-6 -174 G>C Polymorphism with susceptibility to colorectal cancer and gastric cancer: a systematic review and meta-analysis. Acta Med (Hradec Kralove). 2019;62:137–46. NLM (Medline).
Rani J, Mittal I, Pramanik A, Singh N, Dube N, Sharma S, et al. T2DiACoD: a gene atlas of type 2 diabetes mellitus associated complex disorders. Sci Rep. 2017;7:1–21. Nature Publishing Group.
Ingelsson E, McCarthy MI. Human genetics of obesity and type 2 diabetes mellitus: past, present, and future. Circ Genom Precis Med. 2018;11:e002090. NLM (Medline).
Nakhjavani M, Esfahanian F, Jahanshahi A, Esteghamati A, Nikzamir AR, Rashidi A, et al. The relationship between the insertion/deletion polymorphism of the ACE gene and hypertension in Iranian patients with type 2 diabetes. Nephrol Dial Transplant. 2007;22:2549–53.
Fathi M, Nikzamir AR, Esteghamati A, Nakhjavani M, Yekaninejad MS. Combination of angiotensin converting enzyme insertion/deletion (I/D) (rs4646994) and VEGF polymorphism (+405G/C; rs2010963) synergistically associated with the development, of albuminuria in iranian patients with type 2 diabetes. Iranian Red Crescent Med J. 2015;17:e19469. Kowsar Medical Publishing Company.
Moradzadegan A, Vaisi-Raygani A, Nikzamir A, Rahimi Z. Angiotensin converting enzyme insertion/deletion (I/D) (rs4646994) and Vegf polymorphism (+405G/C; Rs2010963) in type II diabetic patients: Association with the risk of coronary artery disease. J Renin-Angiotensin-Aldosterone Syst. 2015;16:672–80. SAGE Publications Ltd.
Dastgheib SA, Najafi F, Shajari A, Bahrami R, Asadian F, Sadeghizadeh-Yazdi J, et al. Association of plasminogen activator inhibitor-1 4G5G Polymorphism with risk of diabetic nephropathy and retinopathy: a systematic review and meta-analysis. J Diabetes Metab Disord {Internet]. 2020;19:2005–16. Available from.
Kee J-Y, Han Y-H, Kim D-S, Mun J-G, Park S-H, So H-S, et al. β-Lapachone suppresses the lung metastasis of melanoma via the MAPK signaling pathway. Ahmad A, editor. PLoS ONE. 2017;12:e0176937.
Grammer TB, Renner W, von Karger S, Boehm BO, Winkelmann BR, Maerz W. The angiotensin-I converting enzyme I/D polymorphism is not associated with type 2 diabetes in individuals undergoing coronary angiography. (The Ludwigshafen Risk and Cardiovascular Health Study). Mol Genet Metab. 2006;88:378–83. Available from.
Mazza A, Bossone E, Mazza F, Distante A. Reduced serum homocysteine levels in type 2 diabetes. Nutr Metab Cardiovasc Dis [Internet]. 2005;15:118–24. Available from.
Zhou D, Ruiter R, Zhang J, Zhou M, Liu H, Liu W, et al. Angiotensin-converting enzyme I/D polymorphism is not associated with type 2 diabetes in a Chinese population. J Renin-Angiotensin-Aldosterone Syst. 2012;13:372–8. SAGE Publications Ltd.
Feng Y, Niu T, Xu X, Chen C, Li Q, Qian R, et al. Insertion/deletion polymorphism of the ACE gene is associated with type 2 diabetes. Diabetes. 2002;51:1986–8. American Diabetes Association Inc.
Daimon M, Oizumi T, Saitoh T, Kameda W, Hirata A, Yamaguchi H, et al. The D allele of the angiotensin-converting enzyme insertion/deletion (I/D) polymorphism is a risk factor for type 2 diabetes in a population-based Japanese sample. Endocr J. 2003;50:393–8.
Xu F, Liu H, Sun Y. Association of plasminogen activator inhibitor-1 gene polymorphism and type 2 diabetic nephropathy. Renal Failure Taylor and Francis Ltd. 2016;38:157–62.
Ezzidi I, Mtiraoui N, Chaieb M, Kacem M, Mahjoub T, Almawi WY. Diabetic retinopathy, PAI-1 4G/5G and -844G/A polymorphisms, and changes in circulating PAI-1 levels in Tunisian type 2 diabetes patients. Diabetes Metab. 2009;35:214–9.
Li G, Liu Y, Li X, Ning Z, Sun Z, Zhang M, et al. Association of PAI-1 4G/5G polymorphism with ischemic stroke in Chinese patients with type 2 diabetes mellitus. Genet Test Mol Biomarkers. 2018;22:554–60. Mary Ann Liebert Inc.
Al-Hamodi Z, Saif-Ali R, Ismail IS, Ahmed KA, Muniandy S. Effect of plasminogen activator inhibitor-1 and tissue plasminogen activator polymorphisms on susceptibility to type 2 diabetes in Malaysian subjects. J Biomed Biotechnol. 2012;2012:234937.
Funding
This study was performed by individual funding grants.
Author information
Authors and Affiliations
Contributions
SM, MHS and SAD: Methodology, conceptualization.
SM, AS, and HN: investigation.
HN, SM, and SAD: Methodology, software.
MHS, HN, AS: Formal analysis, investigation.
SM, AS, HN and SAD: Writing, reviewing, editing.
Corresponding author
Ethics declarations
Ethics approval
This study was approved by Ashkezar Branch, Islamic Azad University, Ashkezar, Iran.
Consent to participate
A written informed consent was obtained from each participant.
Conflicts of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Miri, S., Sheikhha, M.H., Dastgheib, S.A. et al. Association of ACE I/D and PAI-1 4G/5G polymorphisms with susceptibility to type 2 diabetes mellitus. J Diabetes Metab Disord 20, 1191–1197 (2021). https://doi.org/10.1007/s40200-021-00839-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40200-021-00839-7