Abstract
Purpose
Spexin, a novel 14-amino acid peptide, has multiple physiological functions. The purpose of this paper was to systematically evaluate the current literature on the role of Spexin neuropeptide in obesity and its related comorbidities, food intake and overall metabolic status in human, animal and in vitro studies.
Methods
Multiple databases, including PubMed, EMBASE, ProQuest, Scopus and Google Scholar were searched for English-language papers published since inception until December 2018, that investigated Spexin levels in relation to chronic metabolic diseases, overall metabolism control and feeding-related behaviors.The quality of the included observational studies was assessed by a version of the Newcastle Ottawa Scale (NOS) designed for non-randomized studies and SYRCLE’s assessment tool for animal models.
Results
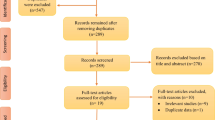
Out of 224 records screened, search results led to a total of 24 related studies (12 human studies (ten cross-sectional studies, one cohort study, and one longitudinal study) and 12 studies in either animals or in vitro).Nine of the included cross-sectional studies and one Longitudinal study had moderate to good study quality, and one cross-sectional and one cohort study had high-quality (or low risk of bias).
Conclusion
It appears that Spexin has a positive impact on overall metabolic status. As a novel appetite-regulating peptide, Spexin can act as an anorexigenic factor. Information about Spexin is very limited, and well-designed randomized controlled clinical trials are warranted for replicating, validating, and extending the current findings.
PROSPERO registration number
CRD42018117198).


Similar content being viewed by others
References
Mirabeau O, Perlas E, Severini C, Audero E, Gascuel O, Possenti R, et al. Identification of novel peptide hormones in the human proteome by hidden Markov model screening. Genome Res. 2007;17(3):320–7.
Wan B, Wang XR, Zhou YB, Zhang X, Huo K, Han ZG. C12ORF39, a novel secreted protein with a typical amidation processing signal. Biosci Rep. 2009;30(1):1–10.
Wong MK, et al. Goldfish spexin: solution structure and novel function as a satiety factor in feeding control. Am J Physiol Endocrinol Metab. 2013;305(3):E348–66.
Porzionato A, Rucinski M, Macchi V, Stecco C, Malendowicz LK, de Caro R. Spexin expression in normal rat tissues. J Histochem Cytochem. 2010;58(9):825–37.
Ma A, Bai J, He M, Wong AOL. Spexin as a neuroendocrine signal with emerging functions. Gen Comp Endocrinol. 2018;265:90–6.
Kumar S, Hossain J, Nader N, Aguirre R, Sriram S, Balagopal PB. Decreased circulating levels of Spexin in obese children. J Clin Endocrinol Metab. 2016;101(7):2931–6.
Walewski JL, Ge F, Lobdell H IV, Levin N, Schwartz GJ, Vasselli JR, et al. Spexin is a novel human peptide that reduces adipocyte uptake of long chain fatty acids and causes weight loss in rodents with diet-induced obesity. Obesity (Silver Spring). 2014;22(7):1643–52.
Kolodziejski PA, et al. Serum levels of spexin and kisspeptin negatively correlate with obesity and insulin resistance in women. Physiol Res. 2018;67(1):45–56.
Kumar S, Hossain MJ, Javed A, Kullo IJ, Balagopal PB. Relationship of circulating spexin with markers of cardiovascular disease: a pilot study in adolescents with obesity. Pediatr Obes. 2018;13(6):374–80.
Li S, Liu Q, Xiao L, Chen H, Li G, Zhang Y, et al. Molecular cloning and functional characterization of spexin in orange-spotted grouper (Epinephelus coioides). Comp Biochem Physiol B Biochem Mol Biol. 2016;196-197:85–91.
Karaca A, Bakar-Ates F, Ersoz Gulcelik N. Decreased Spexin levels in patients with type 1 and type 2 diabetes. Med Princ Pract. 2018;27:549–54.
Al-Daghri NM, et al. Spexin levels are associated with metabolic syndrome components. Dis Markers. 2018;2018:1679690.
Agha M, Agha R. The rising prevalence of obesity: part A: impact on public health. Int J Surg Oncol (N Y). 2017;2(7):e17.
Gu L, Ma Y, Gu M, Zhang Y, Yan S, Li N, et al. Spexin peptide is expressed in human endocrine and epithelial tissues and reduced after glucose load in type 2 diabetes. Peptides. 2015;71:232–9.
Ge JF, et al. Regulation of hepatocellular fatty acid uptake in mouse models of fatty liver disease with and without functional Leptin signaling: roles of NfKB and SREBP-1C and the effects of Spexin. Semin Liver Dis. 2016;36(4):360–72.
Hodges SK, Teague AM, Dasari PS, Short KR. Effect of obesity and type 2 diabetes, and glucose ingestion on circulating spexin concentration in adolescents. Pediatr Diabetes. 2018;19(2):212–6.
Canas JA, Sweeten S, Balagopal PB. Biomarkers for cardiovascular risk in children. Curr Opin Cardiol. 2013;28(2):103–14.
Zieske AW, Tracy RP, McMahan CA, Herderick EE, Homma S, Malcom GT, et al. Elevated serum C-reactive protein levels and advanced atherosclerosis in youth. Arterioscler Thromb Vasc Biol. 2005;25(6):1237–43.
Al-Daghri NM, et al. Associations of Spexin and cardiometabolic parameters among women with and without gestational diabetes mellitus. Saudi J Biol Sci. 2018;25(4):710–4.
Stroup DF, Berlin JA, Morton SC, Olkin I, Williamson GD, Rennie D, et al. Meta-analysis of observational studies in epidemiology: a proposal for reporting. Meta-analysis of observational studies in epidemiology (MOOSE) group. Jama. 2000;283(15):2008–12.
Moher D, Liberati A, Tetzlaff J, Altman DG, PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Ann Intern Med. 2009;151(4):264–9.
Wells G, et al.. The Newcastle-Ottawa Scale (NOS) for assessing the quality if nonrandomized studies in meta-analyses. 2009. Epub Available from: URL: http://www.ohri. ca/programs/clinical_epidemiology/oxford. htm [cited 2009 Oct 19], 2016.
Hooijmans CR, Rovers MM, de Vries RBM, Leenaars M, Ritskes-Hoitinga M, Langendam MW. SYRCLE's risk of bias tool for animal studies. BMC Med Res Methodol. 2014;14:43.
Deng SP, Chen HP, Zhai Y, Jia LY, Liu JY, Wang M, et al. Molecular cloning, characterization and expression analysis of spexin in spotted scat (Scatophagus argus). Gen Comp Endocrinol. 2018;266:60–6.
Wang S, Wang B, Chen S. Spexin in the half-smooth tongue sole (Cynoglossus semilaevis): molecular cloning, expression profiles, and physiological effects. Fish Physiol Biochem. 2018;44(3):829–39.
Ma A, He M, Bai J, Wong MK, Ko WK, Wong AO. Dual role of insulin in Spexin regulation: functional link between food intake and Spexin expression in a fish model. Endocrinology. 2017;158(3):560–77.
Zheng B, Li S, Liu Y, Li Y, Chen H, Tang H, et al. Spexin suppress food intake in Zebrafish: evidence from gene knockout study. Sci Rep. 2017;7(1):14643.
Wu H, Lin F, Chen H, Liu J, Gao Y, Zhang X, et al. Ya-fish (Schizothorax prenanti) spexin: identification, tissue distribution and mRNA expression responses to periprandial and fasting. Fish Physiol Biochem. 2016;42(1):39–49.
Kumar S, Hossain MDJ, Inge T, Balagopal PB. Roux-en-Y gastric bypass surgery in youth with severe obesity: 1-year longitudinal changes in spexin. Surg Obes Relat Dis. 2018;14(10):1537–43.
Lin CY, Huang T, Zhao L, Zhong LLD, Lam WC, Fan BM, et al. Circulating Spexin levels negatively correlate with age, BMI, fasting glucose, and triglycerides in healthy adult women. J Endocr Soc. 2018;2(5):409–19.
Al-Daghri NM, et al. Circulating spexin levels are influenced by the presence or absence of gestational diabetes. Cytokine. 2019;113:291–5.
Sassek M, Kolodziejski PA, Szczepankiewicz D, Pruszynska-Oszmalek E. Spexin in the physiology of pancreatic islets-mutual interactions with insulin. Endocrine. 2019;63(3):513–9.
Sassek M, Kolodziejski PA, Strowski MZ, Nogowski L, Nowak KW, Mackowiak P. Spexin modulates functions of rat endocrine pancreatic cells. Pancreas. 2018;47(7):904–9.
Lin CY, Zhao L, Huang T, Lu L, Khan M, Liu J, et al. Spexin acts as novel regulator for bile acid synthesis. Front Physiol. 2018;9:378.
Kolodziejski PA, Pruszynska-Oszmalek E, Micker M, Skrzypski M, Wojciechowicz T, Szwarckopf P, et al. Spexin: a novel regulator of adipogenesis and fat tissue metabolism. Biochim Biophys Acta Mol Cell Biol Lipids. 2018;1863(10):1228–36.
Petrescu O, Fan X, Gentileschi P, Hossain S, Bradbury M, Gagner M, et al. Long-chain fatty acid uptake is upregulated in omental adipocytes from patients undergoing bariatric surgery for obesity. Int J Obes. 2005;29(2):196–203.
Volkoff H. The neuroendocrine regulation of food intake in fish: a review of current knowledge. Front Neurosci. 2016;10:540.
Waters SM, Krause JE. Distribution of galanin-1, −2 and −3 receptor messenger RNAs in central and peripheral rat tissues. Neuroscience. 2000;95(1):265–71.
Legakis I, Mantzouridis T, Mountokalakis T. Positive correlation of galanin with glucose in type 2 diabetes. Diabetes Care. 2005;28(3):759–60.
Fang P, Yu M, Shi M, Zhang Z, Sui Y, Guo L, et al. Galanin peptide family as a modulating target for contribution to metabolic syndrome. Gen Comp Endocrinol. 2012;179(1):115–20.
Fang P, Yu M, Guo L, Bo P, Zhang Z, Shi M. Galanin and its receptors: a novel strategy for appetite control and obesity therapy. Peptides. 2012;36(2):331–9.
Fang P, Min W, Sun Y, Guo L, Shi M, Bo P, et al. The potential antidepressant and antidiabetic effects of galanin system. Pharmacol Biochem Behav. 2014;120:82–7.
Guo L, Shi M, Zhang L, Li G, Zhang L, Shao H, et al. Galanin antagonist increases insulin resistance by reducing glucose transporter 4 effect in adipocytes of rats. Gen Comp Endocrinol. 2011;173(1):159–63.
Brunner SM, Farzi A, Locker F, Holub BS, Drexel M, Reichmann F, et al. GAL3 receptor KO mice exhibit an anxiety-like phenotype. Proc Natl Acad Sci U S A. 2014;111(19):7138–43.
Zhang X. Regulatory functions of innate-like B cells. Cell Mol Immunol. 2013;10(2):113–21.
Cojocaru M, Cojocaru IM, Siloşi I, Rogoz S. Role of leptin in autoimmune diseases. Maedica (Buchar). 2013;8(1):68–74.
Procaccini C, Pucino V, Mantzoros CS, Matarese G. Leptin in autoimmune diseases. Metabolism. 2015;64(1):92–104.
Iikuni N, Kwan Lam Q, Lu L, Matarese G, Cava A. Leptin and inflammation. Curr Immunol Rev. 2008;4(2):70–9.
Engeli S, Feldpausch M, Gorzelniak K, Hartwig F, Heintze U, Janke J, et al. Association between adiponectin and mediators of inflammation in obese women. Diabetes. 2003;52(4):942–7.
Kim DK, Yun S, Son GH, Hwang JI, Park CR, Kim JI, et al. Coevolution of the spexin/galanin/kisspeptin family: Spexin activates galanin receptor type II and III. Endocrinology. 2014;155(5):1864–73.
Acknowledgments
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
Conceived and designed the systematic review: MB,AO.
Systematic literature search: ZF, MB.
Screened the titles and abstracts of the retrieved articles: VM,SP,EV.
Quality assessment and data extraction: MB,VM,AO.
Wrote the paper: All of authors.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Behrooz, M., Vaghef-Mehrabany, E., Maleki, V. et al. Spexin status in relation to obesity and its related comorbidities: a systematic review. J Diabetes Metab Disord 19, 1943–1957 (2020). https://doi.org/10.1007/s40200-020-00636-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40200-020-00636-8