Abstract
Background
Diabetes mellitus is a group of metabolic disorders in which there are high blood glucose levels over a prolonged period. Diabetes is one of many diseases associated with pathologically elevated levels of endothelin (ET)-1. We have recently proposed the development of ET-traps, which are an antibody – based fusion protein that potently bind and sequester pathologically elevated levels of endothelin-1.
Methods
We constructed ET-traps that were found to be very potent binders to ET-1, with a KD of 32.5ρM. We then treated human retinal microvascular endothelial cells (HRMECs), which are an in vitro model of glucose induced cellular damage, with 10 nM ET-1 or high glucose levels (25 mM).
Results
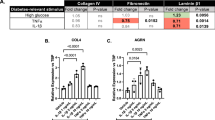
In this study, we investigated the effects of our ET-trap constructs on the expression levels of both collagen 4α1 and fibronectin, which are both important pathologic markers in diabetes. Treating HRMECs with 10 nM ET-1 or 25 mM glucose significantly induces the expression of the ECM proteins fibronectin and collagen 4α1, as is found in chronic diabetic complications; Incubation of the cells with the ET-traps significantly prevented the increased expression of fibronectin and collagen 4α1 back to basal levels. This was found with both mRNA and protein expression levels of the two ECM proteins.
Conclusion
Our results provide the first evidence of the efficacy of ET-traps in reducing pathologic markers in an in vitro model (of diabetes). Further research is warranted to determine the efficacy of ET-traps as a therapeutic tool for diabetes, which is a major public health burden around the world.




Similar content being viewed by others
References
Eizirik DL, Cardozo AK, Cnop M. The role for endoplasmic reticulum stress in diabetes mellitus. Endocr Rev. 2008;29(1):42–61.
Cerf ME. Beta cell dysfunction and insulin resistance. Front Endocrinol (Lausanne). 2013;4:37.
Mathis D, Vence L, Benoist C. Beta-cell death during progression to diabetes. Nature. 2001;414(6865):792–8.
Chen S, et al. Hyperhexosemia induced functional and structural changes in the kidneys: role of endothelins. Nephron. 2002;90(1):86–94.
Chen S, et al. Diabetes-induced myocardial structural changes: role of endothelin-1 and its receptors. J Mol Cell Cardiol. 2000;32(9):1621–9.
Chen S, et al. Differential activation of NF-kappa B and AP-1 in increased fibronectin synthesis in target organs of diabetic complications. Am J Physiol Endocrinol Metab. 2003;284(6):E1089–97.
Evans T, et al. Endothelin receptor blockade prevents augmented extracellular matrix component mRNA expression and capillary basement membrane thickening in the retina of diabetic and galactose-fed rats. Diabetes. 2000;49(4):662–6.
Chen S, et al. High glucose-induced, endothelin-dependent fibronectin synthesis is mediated via NF-kappa B and AP-1. Am J Physiol Cell Physiol. 2003;284(2):C263–72.
Simonson MS, Ismail-Beigi F. Endothelin-1 increases collagen accumulation in renal mesangial cells by stimulating a chemokine and cytokine autocrine signaling loop. J Biol Chem. 2011;286(13):11003–8.
Seligman BG, et al. Increased plasma levels of endothelin 1 and von Willebrand factor in patients with type 2 diabetes and dyslipidemia. Diabetes Care. 2000;23(9):1395–400.
Schneider JG, et al. Elevated plasma endothelin-1 levels in diabetes mellitus. Am J Hypertens. 2002;15(11):967–72.
Stehouwer CD, et al. Endothelial dysfunction and pathogenesis of diabetic angiopathy. Cardiovasc Res. 1997;34(1):55–68.
De Caterina, R., Endothelial dysfunctions: common denominators in vascular disease. Curr Opin Clin Nutr Metab Care, 2000. 3(6): p. 453–467.
Deanfield J, et al. Endothelial function and dysfunction. Part I: methodological issues for assessment in the different vascular beds: a statement by the working group on endothelin and endothelial factors of the European Society of Hypertension. J Hypertens. 2005;23(1):7–17.
Bohm F, Pernow J. The importance of endothelin-1 for vascular dysfunction in cardiovascular disease. Cardiovasc Res. 2007;76(1):8–18.
Jain A, et al. Creating a soluble binder to Endothelin-1 based on the natural ligand binding domains of the Endothelin-1 (G-protein-coupled) receptor. Int J Pept Res Ther. 2017.
Patel S, et al. Selection of a high-affinity WW domain against the extracellular region of VEGF receptor isoform-2 from a combinatorial library using CIS display. Protein Eng Des Sel. 2013;26(4):307–15.
Khan ZA, et al. Oncofetal fibronectin in diabetic retinopathy. Invest Ophthalmol Vis Sci. 2004;45(1):287–95.
Khan ZA, Chakrabarti S. Chronic diabetic complications: endothelial cells at the frontline. In: Rahman AU, Choudhary I, editors. Frontiers in cardiovascular drug discovery. Bentham Science publishers; 2010. p. 121–137.
Kaur H, et al. Diabetes-induced extracellular matrix protein expression is mediated by transcription coactivator p300. Diabetes. 2006;55(11):3104–11.
Jain A, et al. Endothelin-1 induces endoplasmic reticulum stress by activating the PLC-IP(3) pathway: implications for placental pathophysiology in preeclampsia. Am J Pathol. 2012;180(6):2309–20.
Manea SA, Todirita A, Manea A. High glucose-induced increased expression of endothelin-1 in human endothelial cells is mediated by activated CCAAT/enhancer-binding proteins. PLoS One. 2013;8(12):e84170.
Chen S, et al. Interaction of endothelin-1 with vasoactive factors in mediating glucose-induced increased permeability in endothelial cells. Lab Investig. 2000;80(8):1311–21.
De Miguel, C. and J.S. Pollock, Does endoplasmic reticulum stress mediate endothelin-1-induced renal inflammation? Am J Physiol Regul Integr Comp Physiol, 2013. 305(2): p. R107–R109.
Jain A. Endothelin-1-induced endoplasmic reticulum stress in disease. J Pharmacol Exp Ther. 2013;346(2):163–72.
Jain A, Migdalska-Richards A, Jain A. Endothelin-1 -induced endoplasmic reticulum stress in Parkinson's disease. Pharmacologia. 2014;5(3):84–90.
Funding
This project was funded by private funding.
Author information
Authors and Affiliations
Contributions
AJ and SC participated in the research design. HY and SC conducted the experiments. AJ and SC contributed new reagents or analytic tools. SC performed the data analysis. AJ, SC and SC wrote or contributed to the writing of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The author(s) declare that they have no competing interests.
Declaration statement
Any inquiries on data of this study can be directed to Dr. Arjun Jain.
Additional information
Key points
ET-traps have a very high binding affinity.
ET-traps significantly return to basal levels different pathologic markers both at the mRNA and at the protein level.
ET-traps are non-cytotoxic.
Rights and permissions
About this article
Cite this article
Jain, A., Chen, S., Yong, H. et al. Endothelin-1 traps potently reduce pathologic markers back to basal levels in an in vitro model of diabetes. J Diabetes Metab Disord 17, 189–195 (2018). https://doi.org/10.1007/s40200-018-0360-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40200-018-0360-8