Abstract
Background
Myocardial ischemia/reperfusion injury (MIRI) seriously threatens the health of people. The mitochondrial dysfunction in cardiomyocytes can promote the progression of MIRI. Dexmedetomidine (Dex) could alleviate the myocardial injury, which was known to reverse mitochondrial dysfunction in lung injury. However, the function of Dex in mitochondrial dysfunction during MIRI remains unclear.
Objective
To assess the function of Dex in mitochondrial dysfunction during MIRI.
Methods
To investigate the function of Dex in MIRI, H9C2 cells were placed in condition of hypoxia/reoxygenation (H/R). CCK8 assay was performed to test the cell viability, and the mitochondrial membrane potential was evaluated by JC-1 staining. In addition, the binding relationship between Sirt3 and Prdx3 was explored by Co-IP assay. Furthermore, the protein expressions were examined using western blot.
Results
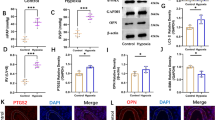
Dex could abolish H/R-induced mitochondrial dysfunction in H9C2 cells. In addition, H/R treatment significantly inhibited the expression of Sirt3, while Dex partially restored this phenomenon. Knockdown of Sirt3 or Prdx3 obviously reduced the protective effect of Dex on H/R-induced mitochondrial injury. Meanwhile, Sirt3 could enhance the function of Prdx3 via deacetylation of Prdx3.
Conclusion
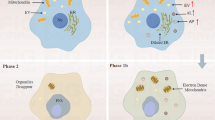
Dex was found to attenuate H/R-induced mitochondrial dysfunction in cardiomyocytes via activation of Sirt3/Prdx3 pathway. Thus, this study might shed new lights on exploring new strategies for the treatment of MIRI.
Graphical abstract





Similar content being viewed by others
Data availability
The supplementary materials_raw data were shown in: https://osf.io/hn9z6/; DOI: https://doi.org/10.17605/OSF.IO/HN9Z6.
Abbreviations
- Dex:
-
Dexmedetomidine
- Sirt3:
-
Sirtuin-3
- Prdx3:
-
Peroxiredoxin-3
- MMP:
-
Mitochondrial membrane potential
References
Wen J, Peng H, Wang D, Wen Z, Liu Y, Qu J, et al. Lipopolysaccharides protect mesenchymal stem cell against cardiac ischemia-reperfusion injury by HMGB1/STAT3 signaling. J Geriatr Cardiol. 2023;20:801–12.
Luo X, Zhang C, Yue C, Jiang Y, Yang F, Xian YJCs. A near-infrared light-activated nanoprobe for simultaneous detection of hydrogen polysulfide and sulfur dioxide in myocardial ischemia-reperfusion injury. Chem Sci. 2023;14:14290–301.
Shi P, Wu J, Li M, Cao Y, Wu J, Ren P, et al. Upregulation of Hsp27 via further inhibition of histone H2A ubiquitination confers protection against myocardial ischemia/reperfusion injury by promoting glycolysis and enhancing mitochondrial function. Cell Death Discov. 2023;9:466.
Jin J, Xu F, Zhang Y, Guan J, Fu J. Myocardial ischemia-reperfusion injury is probably due to the excessive production of mitochondrial ROS caused by the activation of 5-HT degradation system mediated by PAF receptor. Mol Immunol. 2023;155:27–43.
Chen M, Zhong G, Liu M, He H, Zhou J, Chen J, et al. Integrating network analysis and experimental validation to reveal the mitophagy-associated mechanism of Yiqi Huoxue (YQHX) prescription in the treatment of myocardial ischemia/reperfusion injury. Pharmacol Res. 2023;189:106682.
Liao CL, Liu Y, Huang MZ, Liu HY, Ye ZL, Su Q. Myocardial ischemia reperfusion injury is alleviated by curcumin-peptide hydrogel via upregulating autophagy and protecting mitochondrial function. Stem Cell Res Ther. 2021;12:89.
Zhang P, Yu Y, Wang P, Shen H, Ling X, Xue X, et al. Role of hydrogen sulfide in myocardial ischemia-reperfusion injury. J Cardiovasc Pharmacol. 2021;77:130–41.
Tang C, Hu Y, Gao J, Jiang J, Shi S, Wang J, et al. Dexmedetomidine pretreatment attenuates myocardial ischemia reperfusion induced acute kidney injury and endoplasmic reticulum stress in human and rat. Life Sci. 2020;257:118004.
Yu W, Lyu J, Jia L, Sheng M, Yu H, Du H. Dexmedetomidine ameliorates hippocampus injury and cognitive dysfunction induced by hepatic ischemia/reperfusion by activating SIRT3-mediated mitophagy and inhibiting activation of the NLRP3 inflammasome in young rats. Oxid Med Cell Longev. 2020;2020:7385458.
Li H, Zhang X, Chen M, Chen J, Gao T, Yao S. Dexmedetomidine inhibits inflammation in microglia cells under stimulation of LPS and ATP by c-Fos/NLRP3/caspase-1 cascades. EXCLI J. 2018;17:302–11.
Liang S, Wang Y, Liu Y. Dexmedetomidine alleviates lung ischemia-reperfusion injury in rats by activating PI3K/Akt pathway. Eur Rev Med Pharmacol Sci. 2019;23:370–7.
Yang J, Wu Y, Xu Y, Jia J, Xi W, Deng H, et al. Dexmedetomidine resists intestinal ischemia-reperfusion injury by inhibiting TLR4/MyD88/NF-kappaB signaling. J Surg Res. 2021;260:350–8.
Yu P, Zhang J, Ding Y, Chen D, Sun H, Yuan F, et al. Dexmedetomidine post-conditioning alleviates myocardial ischemia-reperfusion injury in rats by ferroptosis inhibition via SLC7A11/GPX4 axis activation. Hum Cell. 2022;35:836–48.
Wang Z, Yao M, Jiang L, Wang L, Yang Y, Wang Q, et al. Dexmedetomidine attenuates myocardial ischemia/reperfusion-induced ferroptosis via AMPK/GSK-3beta/Nrf2 axis. Biomed Pharmacother. 2022;154:113572.
He J, Shangguan X, Zhou W, Cao Y, Zheng Q, Tu J, et al. Glucose limitation activates AMPK coupled SENP1-Sirt3 signalling in mitochondria for T cell memory development. Nat Commun. 2021;12:4371.
Diao Z, Ji Q, Wu Z, Zhang W, Cai Y, Wang Z, et al. SIRT3 consolidates heterochromatin and counteracts senescence. Nucleic Acids Res. 2021;49:4203–19.
Chen J, Chen S, Zhang B, Liu J. SIRT3 as a potential therapeutic target for heart failure. Pharmacol Res. 2021;165:105432.
Sun W, Liu C, Chen Q, Liu N, Yan Y, Liu B. SIRT3: A new regulator of cardiovascular diseases. Oxid Med Cell Longev. 2018;2018:7293861.
Fu Z, Pang Z, He L, Zhang L, Fan Y, Zhao C, et al. Dexmedetomidine confers protection against neuronal oxygen glucose deprivation-reperfusion by regulating SIRT3 mediated autophagy. Neurochem Res. 2022;47:3490–505.
Zhang Q, Liu XM, Hu Q, Liu ZR, Liu ZY, Zhang HG, et al. Dexmedetomidine inhibits mitochondria damage and apoptosis of enteric glial cells in experimental intestinal ischemia/reperfusion injury via SIRT3-dependent PINK1/HDAC3/p53 pathway. J Transl Med. 2021;19:463.
Tao WH, Shan XS, Zhang JX, Liu HY, Wang BY, Wei X, et al. Dexmedetomidine attenuates ferroptosis-mediated renal ischemia/reperfusion injury and inflammation by inhibiting ACSL4 via alpha2-AR. Front Pharmacol. 2022;13:782466.
Li H, Wang Z, Xie X, Luo M, Shen H, Li X, et al. Peroxiredoxin-3 plays a neuroprotective role in early brain injury after experimental subarachnoid hemorrhage in rats. Brain Res Bull. 2022;193:95–105.
Sonn SK, Song EJ, Seo S, Kim YY, Um JH, Yeo FJ, et al. Peroxiredoxin 3 deficiency induces cardiac hypertrophy and dysfunction by impaired mitochondrial quality control. Redox Biol. 2022;51:102275.
Li X, He P, Wang XL, Zhang S, Devejian N, Bennett E, et al. Sulfiredoxin-1 enhances cardiac progenitor cell survival against oxidative stress via the upregulation of the ERK/NRF2 signal pathway. Free Radic Biol Med. 2018;123:8–19.
Li M, Jiao L, Shao Y, Li H, Sun L, Yu Q, et al. LncRNA-ZFAS1 promotes myocardial ischemia-reperfusion injury through DNA methylation-mediated Notch1 Down-regulation in mice. JACC Basic Transl Sci. 2022;7:880–95.
Qiu Z, He Y, Ming H, Lei S, Leng Y, Xia ZY. Lipopolysaccharide (LPS) aggravates high glucose- and hypoxia/reoxygenation-induced injury through activating ROS-dependent NLRP3 inflammasome-mediated pyroptosis in H9C2 cardiomyocytes. J Diabetes Res. 2019;2019:8151836.
Xu H, Ba Z, Liu C, Yu XJTo. Long noncoding RNA DLEU1 promotes proliferation and glycolysis of gastric cancer cells via APOC1 upregulation by recruiting SMYD2 to induce trimethylation of H3K4 modification. Transl Oncol. 2023;36:101731.
Wang Z, Sun R, Wang G, Chen Z, Li Y, Zhao Y, et al. SIRT3-mediated deacetylation of PRDX3 alleviates mitochondrial oxidative damage and apoptosis induced by intestinal ischemia/reperfusion injury. Redox Biol. 2020;28:101343.
Liang C, Peng Y, Sun H, Wang L, Jiang L, Zou S. Silencing lncRNA KCNQ1OT1 reduced hepatic ischemia reperfusion injury-induced pyroptosis by regulating miR-142a-3p/HMGB1 axis. Mol Cell Biochem. 2022;478:1293–305.
Wu T, Shi G, Ji Z, Wang S, Geng L, Guo Z. Circulating small extracellular vesicle-encapsulated SEMA5A-IT1 attenuates myocardial ischemia-reperfusion injury after cardiac surgery with cardiopulmonary bypass. Cell Mol Biol Lett. 2022;27:95.
Cong D, Yu Y, Meng Y, Qi X. Dexmedetomidine (Dex) exerts protective effects on rat neuronal cells injured by cerebral ischemia/reperfusion via regulating the Sphk1/S1P signaling pathway. J Stroke Cerebrovasc Dis. 2023;32:106896.
Shan X, Zhang J, Wei X, Tao W, Peng K, Liu H, et al. Dexmedetomidine attenuates renal ischemia-reperfusion injury through activating PI3K/Akt-eNOS signaling via alpha(2) adrenoreceptors in renal microvascular endothelial cells. FASEB J. 2022;36:e22608.
Zhang S, Wu X, Wang J, Shi Y, Hu Q, Cui W, et al. Adiponectin/AdiopR1 signaling prevents mitochondrial dysfunction and oxidative injury after traumatic brain injury in a SIRT3 dependent manner. Redox Biol. 2022;54:102390.
Funding
This work was supported by the Scientific Research Project of Heilongjiang Health Commission (20210404110049) and Basic scientific research funds for colleges and universities in Heilongjiang Province (2023-KYYWF-0636).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interests
These authors declared no competing interests in this research.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Qingyun Tan and Wenming Dong are the co-first authors.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tan, Q., Dong, W., Wang, Q. et al. Dexmedetomidine alleviates Hypoxia/reoxygenation-induced mitochondrial dysfunction in cardiomyocytes via activation of Sirt3/Prdx3 pathway. DARU J Pharm Sci (2024). https://doi.org/10.1007/s40199-024-00504-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s40199-024-00504-3