Abstract
Background
In previous studies, authors have completed the total synthesis of several phloroglucinol natural products and synthesized a series of their derivatives, which were tested with good biological activities.
Objectives
To discover anti-MRSA lead compound and study their mechanism of action.
Methods
Phloroglucinol derivatives were tested to investigate their activities against several gram-positive strains including Methicillin-resistant Staphylococcus aureus (MRSA). The mechanism study was conducted by determining extracellular potassium ion concentration, intracellular NADPH oxidase content, SOD activity, ROS amount in MRSA and MRSA survival rate under A5 treatment. The in vitro cytotoxicity test of A5 was conducted.
Results
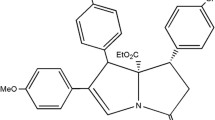
The activity of monocyclic compounds was stronger than that of bicyclic compounds, and compound A5 showed the best MIC value of 0.98 μg/mL and MBC value of 1.95 μg/mL, which were 4–8 times lower than that of vancomycin. The mechanism study of A5 showed that it achieved anti-MRSA effect through membrane damage, which is proved by increased concentration of extracellular potassium ion after A5 treatment. Another possible mechanism is the over ROS production induced cell death, which is suggested by observed alternation of several reactive oxygen species (ROS) related indicators including NADPH concentration, superoxide dismutase (SOD) activity, ROS content and bacterial survival rate after A5 treatment. The cytotoxicity results in vitro showed that A5 was basically non-toxic to cells.
Conclusion
Acylphloroglucinol derivative A5 showed good anti-MRSA activity, possibly via membrane damage and ROS-mediated oxidative stress mechanism. It deserves further exploration to be a potential lead for the development of new anti-MRSA agent.
Graphical Abstract










Similar content being viewed by others
References
Allemani C, Matsuda T, Di Carlo V, et al. Global surveillance of trends in cancer survival 2000–14 (CONCORD-3): analysis of individual records for 37 513 025 patients diagnosed with one of 18 cancers from 322 population-based registries in 71 countries. Lancet. 2018;391(10125):1023–75. https://doi.org/10.1016/S0140-6736(17)33326-3.
Fisher RA, Gollan B, Helaine S. Persistent bacterial infections and persister cells. Nat Rev Microbiol. 2017;15(8):453–64. https://doi.org/10.1038/nrmicro.2017.42.
Fernández J, Gustot T. Management of bacterial infections in cirrhosis. J Hepatol. 2012;56(Suppl 1):S1-12. https://doi.org/10.1016/S0168-8278(12)60002-6.
Hassoun A, Linden PK, Friedman B. Incidence, prevalence, and management of MRSA bacteremia across patient populations—a review of recent developments in MRSA management and treatment. Crit Care. 2017;21(1):211. https://doi.org/10.1186/s13054-017-1801-3.
Peacock SJ, Paterson GK. Mechanisms of Methicillin Resistance in Staphylococcus aureus. Annu Rev Biochem. 2015;84:577–601. https://doi.org/10.1146/annurev-biochem-060614-034516.
Lakhundi S, Zhang K. Methicillin-Resistant Staphylococcus aureus: Molecular Characterization, Evolution, and Epidemiology. Clin Microbiol Rev. 2018;31(4):e00020-e118. https://doi.org/10.1128/cmr.00020-18.
Santajit S, Indrawattana N. Mechanisms of Antimicrobial Resistance in ESKAPE Pathogens. Biomed Res Int. 2016;2016:2475067. https://doi.org/10.1155/2016/2475067.
Mun SH, Kang OH, Kong R, et al. Punicalagin suppresses methicillin resistance of Staphylococcus aureus to oxacillin. J Pharmacol Sci. 2018;137(4):317–23. https://doi.org/10.1016/j.jphs.2017.10.008.
Kitahara T, Aoyama Y, Hirakata Y, et al. In vitro activity of lauric acid or myristylamine in combination with six antimicrobial agents against methicillin-resistant Staphylococcus aureus (MRSA). Int J Antimicrob Agents. 2006;27(1):51–7. https://doi.org/10.1016/j.ijantimicag.2005.08.020.
Heilmann J, Winkelmann K, Sticher O. Studies on the antioxidative activity of phloroglucinol derivatives isolated from hypericum species. Planta Med. 2003;69:202–6. https://doi.org/10.1055/s-2003-38477.
Liu HX, Tan HB, Qiu SX. Antimicrobial acylphloroglucinols from the leaves of Rhodomyrtus tomentosa. J Asian Nat Prod Res. 2016;18(6):535–41. https://doi.org/10.1080/10286020.2015.1121997.
Hua X, Yang Q, Zhang W, et al. Antibacterial Activity and Mechanism of Action of Aspidinol Against Multi-Drug-Resistant Methicillin-Resistant Staphylococcus aureus. Front Pharmacol. 2018;9:619. https://doi.org/10.3389/fphar.2018.00619.
Jacob MR, Walker LA. Natural Products and Antifungal Drug Discovery. Methods Mol Med. 2005;118:83–109. https://doi.org/10.1385/1-59259-943-5:083.
Liu X, Liu J, Jiang T, et al. Analysis of chemical composition and in vitro antidermatophyte activity of ethanol extracts of Dryopteris fragrans (L.) Schot. J Ethnopharmacol. 2018;226:36–43. https://doi.org/10.1016/j.jep.2018.07.030.
Lin H, Liu X, Shen Z, et al. The effect of isoflavaspidic acid PB extracted from Dryopteris fragrans (L.) Schott on planktonic and biofilm growth of dermatophytes and the possible mechanism of antibiofilm. J Ethnopharmacol. 2019;241:111956. https://doi.org/10.1016/j.jep.2019.111956.
Chen NH, Qian YR, Li W, et al. Six New Acylphloroglucinols from Dryopteris championii. Chem Biodivers. 2017;14(7). https://doi.org/10.1002/cbdv.201700001.
Shi PQ. Synthesis and biological activity of fumaric acid and bleomycin compounds. Guangdong Pharmaceutical University, 2021. https://kns.cnki.net/kcms2/article/abstract?v=DxGmxfxkPoFSIbnUFVn3ilTx0dtlcyC6be1__idNCGuT7W_idJd7DmmoNLpvFY1NpVWL-wKzdNMCq8PfttMbVliNyzYRnA31Np0VuxhM16c5Umrxt_71o_PD03tIHKqa-O5n3KPc2CY=&uniplatform=NZKPT&language=CHS.
Liu HY, Du WZ, et al. Study on quality standard of Dryopteris fragrans. J Guangdong Pharmaceutical University. 2016;32(1):36–40 (https://kns.cnki.net/kcms/detail/44.1413.r.20160115.1840.011.html).
Fan HQ, Shen ZB, et al. Research progress on chemical constituents of Dryopteris fragrans and their pharmacological effects in the treatment of skin diseases. Shizhen Guoyi Guoyao. 2013;24(1):199–201 (https://kns.cnki.net/kcms2/article/abstract?v=DxGmxfxkPoHR57lvp9bMq3AwUERnn-OEMwP-504UoehrJOUGtE0HihfiKGC4bbl2GgxnxwhsTzDmiu7SHPIwrPj7Z5Zq0seoIvfU_GrbMZsFD62MVYXYEptJeqThfl1Y&uniplatform=NZKPT&language=CHS).
Rahman MM, Shiu WKP, Gibbons S, et al. Total synthesis of acylphloroglucinols and their antibacterial activities against clinical isolates of multi-drug resistant (MDR) and methicillin-resistant strains of Staphylococcus aureus. Eur J Med Chem. 2018;155:255–62. https://doi.org/10.1016/j.ejmech.2018.05.038.
Feng L, Maddox MM, Alam MZ, et al. Synthesis, structure-activity relationship studies, and antibacterial evaluation of 4-chromanones and chalcones, as well as olympicin A and derivatives. J Med Chem. 2014;57(20):8398–420. https://doi.org/10.1021/jm500853v.
Kahlmeter G, Giske CG, Kirn TJ, et al. Point-Counterpoint: Differences between the European Committee on Antimicrobial Susceptibility Testing and Clinical and Laboratory Standards Institute Recommendations for Reporting Antimicrobial Susceptibility Results. J Clin Microbiol. 2019;57(9):e01129-e1219. https://doi.org/10.1128/JCM.01129-19.
Zhang S, Qu X, Jiao J, et al. Felodipine enhances aminoglycosides efficacy against implant infections caused by methicillin-resistant Staphylococcus aureus, persisters and bi ofilms. Bioactive Mater. 2022;14:272–89. https://doi.org/10.1016/j.bioactmat.2021.11.019.
Sun K, Metzger DW. Influenza infection suppresses NADPH oxidase-dependent phagocytic bacterial clearance and enhances susceptibility to secondary methicillin-resistant Staphylococcus aureus infection. J Immunol. 2014;192(7):3301–7. https://doi.org/10.4049/jimmunol.1303049.
Thanh ND, Giang NTK, Quyen TH, et al. Synthesis and evaluation of in vivo antioxidant, in vitro antibacterial, MRSA and antifungal activity of novel substituted isatin N-(2,3,4,6-tetra-O-acetyl-beta-d-glucopyranosyl) thiosemicarbazones. Eur J Med Chem. 2016;123:532–43. https://doi.org/10.1016/j.ejmech.2016.07.074.
Pramanik A, Laha D, Bhattacharya D, et al. A novel study of antibacterial activity of copper iodide nanoparticle mediated by DNA and membrane damage. Colloids Surf B Biointerf. 2012;96:50–5. https://doi.org/10.1016/j.colsurfb.2012.03.021.
Helmerhorst EJ, Troxler RF, Oppenheim FG. The human salivary peptide histatin 5 exerts its antifungal activity through the formation of reactive oxygen species. Proc Natl Acad Sci U S A. 2001;98:14637–42. https://doi.org/10.1073/pnas.141366998.
Zhao C, Wang X, Wu L, et al. Nitrogen-doped carbon quantum dots as an antimicrobial agent against Staphylococcus for the treatment of infected wounds. Colloids Surf B Biointerf. 2019;179:17–27. https://doi.org/10.1016/j.colsurfb.2019.03.042.
Clinical and Laboratory Standards Institute (CLSI). Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically; approved standard, Ninth Edition (M07-A9). Wayne, PA: CLSI; 2012a.
Clinical and Laboratory Standards Institute (CLSI). Performance Standards for Antimicrobial Disk Susceptibility Tests; Approved Standard, Eleventh Edition (M02-A11). Wayne, PA: CLSI; 2012b.
Wu JW, Li BL, Tang C, et al. Callistemonols A and B, Potent Antimicrobial Acylphloroglucinol Derivatives with Unusual Carbon Skeletons from Callistemon viminalis. J Nat Prod. 2019;82(7):1917–22. https://doi.org/10.1021/acs.jnatprod.9b00064.
Xiang YQ, Liu HX, Zhao LY, et al. Callistemenonone A, a novel dearomatic dibenzofuran-type acylphloroglucinol with antimicrobial activity from Callistemon viminalis. Sci Rep. 2017;7(1):2363. https://doi.org/10.1038/s41598-017-02441-5.
Li N, Gao C, Peng X, et al. Aspidin BB, a phloroglucinol derivative, exerts its antibacterial activity against Staphylococcus aureus by inducing the generation of reactive oxygen species. Res Microbiol. 2014;165(4):263–72. https://doi.org/10.1016/j.resmic.2014.03.002.
Gao C, Guo N, Li N, et al. Investigation of antibacterial activity of aspidin BB against Propionibacterium acnes. Arch Dermatol Res. 2016;308(2):79–86. https://doi.org/10.1007/s00403-015-1603-x.
Yang L, Mih N, Anand A, et al. Cellular responses to reactive oxygen species are predicted from molecular mechanisms. Proc Natl Acad Sci U S A. 2019;116:14368–73. https://doi.org/10.1073/pnas.1905039116.
Jang HJ, Chung IY, Lim C, et al. Redirecting an Anticancer to an Antibacterial Hit Against Methicillin-Resistant Staphylococcus aureus. Front Microbiol. 2019;10:350. https://doi.org/10.3389/fmicb.2019.00350.
Acknowledgements
The work was supported by Innovation and strengthening project of Guangdong Pharmaceutical University- Special Projects in Key Fields of General Colleges and Universities in Guangdong Province(2022ZDZX2030) and Guangdong Province Graduate Education Innovation Program in 2021 (2021JGXM071), Medical Scientific Research Foundation of Guangdong Province (B20234214), Scientific Research Project of Guangdong Provincial Bureau of traditional Chinese Medicine (20231205).
Funding
No funding is applicable.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interest
The authors declare that they have no known competing interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yang, X., Gao, X., Ou, J. et al. Antimicrobial activity and mechanism of anti-MRSA of phloroglucinol derivatives. DARU J Pharm Sci 32, 177–187 (2024). https://doi.org/10.1007/s40199-024-00503-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40199-024-00503-4