Abstract
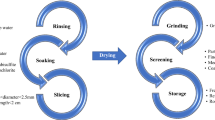
Spray-dried extracts are prepared as powders or granules after solvent removal, which can be obtained in the presence or absence of pharmaceutical adjuvants. This work aimed to optimize the process of obtaining dried extracts of Peperomia pellucida L. (HBK) by spray drying. The characterization of the extract was performed by thermal analysis, specific surface area, particle size and high performance liquid chromatography (HPLC); then, capsules were developed for antimicrobial treatment, evaluating four bench lots by the determination of the angle of repose and time of flow, scanning electron microscopy, porosity and physicochemical quality control. There were no significant differences between the extracts obtained by spray drying at atomization temperatures of 140 °C, 160 °C and 180 °C, which was confirmed by thermal analysis. Specific surface area varied inversely with the mean particle size. Regarding the marker content by HPLC, no significant differences were found between the samples, although the flavonoid fraction was more stable at 160 °C. Bench lots (I to IV) were developed using the diluents Flowlac®, Starch® 1500, microcrystalline cellulose 250 and Cellactose® 80. Based on the results, the bench lot I, containing Flowlac®, was selected. The results of physicochemical quality control demonstrated that the selected formulation meets the pre-established parameters, and proving to be economically viable.








Similar content being viewed by others
References
Lachmann L, Lieberman HÁ, Kanig JL. Teoria e Prática na Indústria Farmacêutica. Lisboa: Fundação Calouste GuBLenkian; 2001.
Hermes AG, Hermes EG, Cavalcante FLM, Luna MS, Leal RA, Cuoco V, Lins AFA, Soriano S. Ações Anti-hipertensivas da Peperomia pellucida(L.) em Cães e na Espécie Humana. Cl. Bio. Cl. Saúde; 1981. pp.47–52.
Majumder P, Abraham P, Satya V. Ethno-medicinal, Phytochemical and Pharmacological review of an amazing medicinal herb Peperomia pellucida (L.) HBK. 2011. pp. 358–364.
Bojo AC, ABLano-Garcia E, Pocsídio GN. The antibacterial activity of Peperomia pellucida(L.) H.B.K. (Piperaceae). Ásia Life Sciences; 1994. pp. 35–44.
Aziba PI, Adedeji A, Ekor M, Adeyemi O. Analgesic activity of Peperomia pellucida aerial parts in mice. Fitoterapia. 2001. pp. 57–58. https://doi.org/10.1016/S0367-326X(00)00249-5.
Khan MR, Omoloso AD. Antibacterial activity of Hygrophilastrictaand Peperomia pellucida. Fitoterapia. 2002. pp. 251–254. https://doi.org/10.1016/S0367-326X(02)00066-7.
Xiang-Ng Z, Than MJY, Yong PH. Peperomia pellucida (L.) Kunth herbal tea: Effect of fermentation and drying methods on the consumer acceptance, antioxidant and anti-inflammatory activities. Food Chemistry. 2020. https://doi.org/10.1016/j.foodchem.2020.128738.
Khan A, Rahman M, Islam S. Antipyretic Activity of Peperomia pellucida Leaves in Rabbit. Turk J. Biol. 2008. pp. 37–41.
Hamzah RU., Odetola AA, Erukainure OL, Oyagbemi AA. Peperomia pellucida in diets modulates hyperglyceamia, oxidative stress and dyslipidemia in diabetic rats. J AcuteDis. 2012. pp. 135–140. https://doi.org/10.1016/S2221-6189(13)60031-1.
Dorla E, Grondin I, Hue T, Clerc P, Dumas S, Gauvin-Bialecki A, Laurent P. Traditional uses, antimicrobial and acaricidal activities of 20 plants selected among Reunion Island's flora. South African Journal of Botany. 2018. pp.1–10. https://doi.org/10.1016/j.sajb.2018.04.014
Mendes LPM, Maciel KM, Vieira ABR, Mendonça LCV, Silva RMF, Rolim Neto PJ, Barbosa WLR, Vieira JMS. Atividade Antimicrobiana de Extratos Etanólicos de Peperomia pellucidae Portulaca pilosa. Rev. Ciênc. Farm. Básica Apl. 2011. pp. 121-125.
Oliveira OW, Petrovick PR. Secagem por aspersão (spray drying) de extratos vegetais: bases e aplicações. Rev. Bras. Farmacogn. 2010. pp. 641–650. https://doi.org/10.1590/S0102-695X2010000400026.
Chaves JS, Costa FB, Freitas LAP Development of enteric coated tablets from spray dried extract of feverfew (Tanacetum parthenium L.). Braz. J. Pharm. Sci. 2009. pp. 573–583. https://doi.org/10.1590/S1984-82502009000300024.
Brazil. Brazilian Pharmacopoeia, volume 1. National Agency of Sanitary Surveillance, Brasília, Brazil. 2010.
Liu W, Chenc XD, Selomulya C. On the spray drying of uniform functional microparticles. Particuology. 2015. pp. 1–12. https://doi.org/10.1016/j.partic.2015.04.001.
Severo AAL, Souza TP, Rolim LA, Sobrinho JLS, Medeiros FPM, Rolim Neto PJ Otimização das condições de extração de senosídeos por soluções hidroetanólicas das folhas de Senna alexandrina MILL empregando planejamento fatorial. Rev. Ciênc. Farm. Básica Apl. 2013. pp. 603-609.
Silva RMM. Flavonóides de Peperomia pellucida. Trabalho de Conclusão de Curso. Faculdade de Farmácia, Instituto de Ciências da Saúde, Universidade Federal do Pará. 2010. 48p.
Silva RMF, Ribeiro JFA, Freitas MCC, Arruda MSP, Nascimento MN, Barbosa WLR, Rolim Neto PJ. Caracterização físico-química e análises por espectrofotometria e cromatografia de Peperomia pellucida L. (H. B. K.). Rev. Bras. Plantas Med. 2013. pp. 717–726.
Costa SPM, Silva KER, Medeiros GCR, Rolim LA, Oliveira JF, Lima MCA, Galdino SL, Pitta IR, Rolim Neto PJ Thermal behavior and compatibility analysis of the new chemical entity LPSF/FZ4. Thermochim. Acta. 2013. pp. 29–34.
Storpirtis S, Gonçalves JE, Chiann C, Gai MN Ciências Farmacêuticas: Biofarmacotécnica. Editora Guanabara Koogan, Rio de Janeiro. 2009. pp. 32–65.
Silva RMF Peperomia pellucida L. (H.B.K.): Obtenção tecnológica de formas farmacêuticas. Tese de doutorado. Programa de Pós-Graduação em Ciências Farmacêuticas. Departamento de Ciências Farmacêuticas, Universidade Federal de Pernambuco. 2010. 173p.
Silva RMF, Gomes TCBL, Medeiros GCR, Peixoto MSP, Pina EML, Costa SPM, Freitas MCC, Silva MN, Arruda MSP, Rolim-Neto PJ Influence of the seasonal changes and of the extraction method on flavonoid content in Peperomia pellucida L. (H.B.K.). IJPSR. 2014. pp. 354–360.
Alves FC Desenvolvimento de formas farmacêuticas de β-lapachona para utilização em terapias antineoplásicas. Dissertação de mestrado. Programa de Pós-graduação em Ciências Farmacêuticas. Departamento de Ciências Farmacêuticas, Universidade Federal de Pernambuco. 2002. 157p.
Silva RMF, Oliveira FHC, Rolim CMB, Pimentel MF, Medeiros FPM, Albuquerque MM; Rolim Neto PJ. Cápsulas de sulfato de indinavir: desenvolvimento farmacotécnico industrial, desenvolvimento do método de dissolução e avaliação. Rev. Bras. Cienc. Farm. 2007. pp 385–395. https://doi.org/10.1590/S1516-93322007000300007.
Nigro PRA, Carazzatto R A Farmacotécnica das Cápsulas Revista Racine 1999. pp. 19–22.
Prista LN, Alves AC, Morgado R. Tecnologia farmacêutica. 4.ed., Fundação Calouste GuBLenkian, Lisboa. 1997. pp. 291–297.
Guedes LC. Controle de qualidade na indústria farmacêutica. Rio de Janeiro: Editora confederação Nacional da Indústria; 1987. p. 33–68.
Brazil. Brazilian Pharmacopoeia, volume 1. National Agency of Sanitary Surveillance, Brasília, Brazil. 2019.
United States Pharmacopoeia Convention (USP). United States Pharmacopoeia 33 - National Formulary 28. The Stationery Office, London, United Kingdom. 2010.
Vasconcelos EAF, Medeiros MGF, Raffin FN, Moura TFAL Influência da temperatura de secagem e da concentração de Aerosil® 200 nas características dos extratos secos por aspersão da Schinusterebinthifolius Raddi (Anacardiaceae). Rev. Bras. Farmacogn. 2005. pp. 243–249. https://doi.org/10.1590/S0102-695X2005000300015.
Couto RO, Oliveira EMS, Martins FS, Freitas O, Bara MTF, Paula JR, Conceicao EC Processing of Apeibatibourbou Aubl. Extract Via Spray Drying. Lat. Am. J. Pharm. 2012. pp. 104–111.
León-Martínez FM, Méndez-Lagunas LL, Rodríguez-Ramírez J Spray drying of nopal mucilage (Opuntiaficus-indica): Effects on powder properties and characterization. Carbohydr Polym. 2010. pp. 864–870. https://doi.org/10.1016/j.carbpol.2010.03.061.
Cheow WS, Li S, Hadinoto K Spray drying formulation of hollow spherical aggregates of silica nanoparticles by experimental design. Chem. Eng. Res. Des. 2010. pp. 673–685. https://doi.org/10.1016/j.cherd.2009.11.012.
Sosnik A, Seremeta KP Advantages and challenges of the spray-drying technology for the production of pure drug particles and drug-loaded polymeric carriers. Adv. Colloid Interface Sci. 2015. pp. 40–54. https://doi.org/10.1016/j.cis.2015.05.003.
Gallo L, Llabot JM, Allemandi D, Bucalá V, Piña J Influence of spray-drying operating conditions on Rhamnuspurshiana (Cáscarasagrada) extract powder physical properties. Powder Technol. 2011. pp. 205–214. https://doi.org/10.1016/j.powtec.2010.12.021.
Gallo L, Ramírez-Rigo MV, Piña J, ucalá, V. A comparative study of spray-dried medicinal plant aqueous extracts. Drying performance and product quality.Chem. Eng. Res. Des. 2015. pp. 681–694. https://doi.org/10.1016/j.cherd.2015.10.009.
Bridgwater J. Mixing of powders and granular materials by mechanical means—A perspective. Particuology. 2012. pp. 397–427. https://doi.org/10.1016/j.partic.2012.06.002.
Shenoy P, Viau M., Tammel P, Innings F, Fitzpatrick J, Ahrné L. Effect of powder densities, particle size and shape on mixture quality of binary food powder mixtures. Powder Technol. 2015. pp. 165–172. https://doi.org/10.1016/j.powtec.2014.11.023.
Rios M. Debating excipients functionality. Pharm Technol http://pharmtech.findpharma.com/pharmtech/Special+Report/Debating-Excipient-Functionality/ArticleStandard/Article/detail/3711322. Accessed 03 March 2010.
Lyra MAM Desenvolvimento de forma farmacêutica granulado efervescente à base de extratos de Bauhiniaforficata Link para obtenção de uma associação – dose – fixa como alternativa no tratamento do Diabetes Melittus. Tese de doutorado. Programa de Pós-Graduação em Ciências Farmacêuticas. Departamento de Ciências Farmacêuticas, Universidade Federal de Pernambuco. 2013. 187p.
Nery CGC, Pires MAS, Pianetti GA, Vianna-Soares CD Caracterização do fármaco hipoglicemiante glibenclamida. Rev. Bras. Cienc. Farm. 2008. pp. 61–73. https://doi.org/10.1590/S1516-93322008000100008.
Acknowledgments
The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Not applicable.
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
da Silva, R.M.F., Gomes, T.C.B.L., Campos, A.F. et al. Obtainment of the spray-dried extracts of Peperomia pellucida L. (H.B.K.) using different atomization temperatures: physicochemical characterization and technological development for pharmaceutical applications. DARU J Pharm Sci 29, 147–158 (2021). https://doi.org/10.1007/s40199-021-00393-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40199-021-00393-w