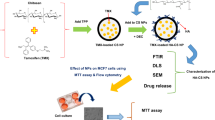
Abstract
Background
treatment of breast cancer as one of the most common cancers in the world remains an important area of drug development based on nanoparticulate systems. Effective targeted therapy of affected cells based on ligand conjugate biocompatible polymeric nanoparticles is an attractive perspective in this context.
Objective
In this study, a novel double effect nanoparticle based on Chitosan-Raloxifene conjugate was prepared for adjuvant therapy (hormone and chemo therapy) and drug targeting to breast cancer cells via estrogen receptor (ER).
Methods
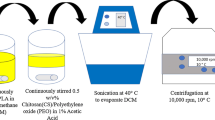
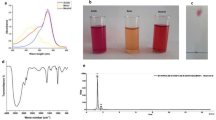
Chitosan-raloxifene conjugate was synthesized. Related nanoparticles containing doxorubicin (DOX) were prepared and characterized. Experimental design study was performed to determine the optimum levels of variables in the preparation of nanoparticle. Drug loading, release, nanoparticle stability, and the effect of nanoparticles on cell viability were evaluated. Further, inhibition tests were performed to demonstrate that the function of these novel nanoparticles is mediated via ER.
Results
Chitosan-raloxifene conjugate was successfully synthesized. The prepared nanoparticles showed sizes within 25–35 nm, more than 95% drug loading, about 60% of drug release and desired stability after 24 h. XTT assay on MCF-7 cell line illustrated that these nanoparticles could inhibit the cellular growth up to 60%. The results from inhibition tests revealed that prepared nanoparticles can inhibit cell growth via ER blocking.
Conclusion
This study introduced chitosan-raloxifene nanoparticles containing doxorubicin as a novel targeting agent for adjuvant therapy of breast cancer.

Graphical abstract





Similar content being viewed by others
References
Chidambaram M, Manavalan R, Kathiresan K. Nanotherapeutics to overcome conventional cancer chemotherapy limitations. J Pharm Pharm Sci. 2011;14(1):67–77.
Yousefpour P, Atyabi F, Vasheghani-Farahani E, Movahedi A-AM, Dinarvand R. Targeted delivery of doxorubicin-utilizing chitosan nanoparticles surface-functionalized with anti-Her2 trastuzumab. Int J Nanomedicine. 2011;6:1977.
Islam MS, Haque P, Rashid TU, Khan MN, Mallik AK, Khan MNI, et al. Core–shell drug carrier from folate conjugated chitosan obtained from prawn shell for targeted doxorubicin delivery. J Mater Sci Mater Med. 2017;28(4):55.
Naruphontjirakul P, Viravaidya-Pasuwat K, Editors. Development of doxorubicin—Core Shell chitosan nanoparticles to treat Cancer. Proceedings of the international conference on biomedical engineering and technology; IACSIT Press: Singapore; 2011.
Bernkop-Schnürch A, Dünnhaupt S. Chitosan-based drug delivery systems. Eur J Pharm Biopharm. 2012;81(3):463–9.
Samadi FY, Mohammadi Z, Yousefi M, Majdejabbari S. Synthesis of raloxifene–chitosan conjugate: a novel chitosan derivative as a potential targeting vehicle. Int J Biol Macromol. 2016;82:599–606.
Qi L-F, Xu Z-R, Li Y, Jiang X, Han X-Y. In vitro effects of chitosan nanoparticles on proliferation of human gastric carcinoma cell line MGC803 cells. World J Gastroenterol: WJG. 2005;11(33):5136–41.
Jing H, Guo Z, Guo W, Yang W, Xu P, Zhang X. Synthesis and characterization of folic acid modified water-soluble chitosan derivatives for folate-receptor-mediated targeting. Bioorg Med Chem Lett. 2012;22(10):3418–24.
Mourya V, Inamdar NN. Chitosan-modifications and applications: opportunities galore. React Funct Polym. 2008;68(6):1013–51.
Mohammadi Z, Abolhassani M, Dorkoosh F, Hosseinkhani S, Gilani K, Amini T, et al. Preparation and evaluation of chitosan–DNA–FAP-B nanoparticles as a novel non-viral vector for gene delivery to the lung epithelial cells. Int J Pharm. 2011;409(1):307–13.
Gil EMC. Targeting the PI3K/AKT/mTOR pathway in estrogen receptor-positive breast cancer. Cancer Treat Rev. 2014;40(7):862–71.
Pritchard K. Endocrine therapy: is the first generation of targeted drugs the last? J Intern Med. 2013;274(2):144–52.
Wood AJ, Riggs BL, Hartmann LC. Selective estrogen-receptor modulators—mechanisms of action and application to clinical practice. N Engl J Med. 2003;348(7):618–29.
Fan P, Jordan VC. Acquired resistance to selective estrogen receptor modulators (SERMs) in clinical practice (tamoxifen & raloxifene) by selection pressure in breast cancer cell populations. Steroids. 2014;90:44–52.
Goldstein SR, Siddhanti S, Ciaccia AV, Plouffe L. A pharmacological review of selective oestrogen receptor modulators. Hum Reprod Update. 2000;6(3):212–24.
Muchmore DB. Raloxifene: a selective estrogen receptor modulator (SERM) with multiple target system effects. Oncologist. 2000;5(5):388–92.
Dutertre M, Smith CL. Molecular mechanisms of selective estrogen receptor modulator (SERM) action. J Pharmacol Exp Ther. 2000;295(2):431–7.
Abdel-Hafez SM, Hathout RM, Sammour OA. Towards better modeling of chitosan nanoparticles production: screening different factors and comparing two experimental designs. Int J Biol Macromol. 2014;64:334–40.
Motwani SK, Chopra S, Talegaonkar S, Kohli K, Ahmad FJ, Khar RK. Chitosan–sodium alginate nanoparticles as submicroscopic reservoirs for ocular delivery: formulation, optimisation and in vitro characterisation. Eur J Pharm Biopharm. 2008;68(3):513–25.
Gazori T, Khoshayand MR, Azizi E, Yazdizade P, Nomani A, Haririan I. Evaluation of alginate/chitosan nanoparticles as antisense delivery vector: formulation, optimization and in vitro characterization. Carbohydr Polym. 2009;77(3):599–606.
Janes KA, Fresneau MP, Marazuela A, Fabra A. Alonso MaJ. Chitosan nanoparticles as delivery systems for doxorubicin. J Control Release. 2001;73(2):255–67.
Acknowledgements
The authors would like to acknowledge Iran National Science Foundation (INSF) for financial support (grant number 91004795), and Mrs. Saadat for her valuable assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None of the authors has any financial or personal relationships that could inappropriately influence or bias the content of the paper.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 16 kb)
Rights and permissions
About this article
Cite this article
Mohammadi, Z., Samadi, F.Y., Rahmani, S. et al. Chitosan-Raloxifene nanoparticle containing doxorubicin as a new double-effect targeting vehicle for breast cancer therapy. DARU J Pharm Sci 28, 433–442 (2020). https://doi.org/10.1007/s40199-020-00338-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40199-020-00338-9