Abstract
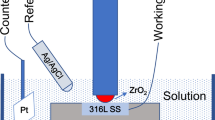
Tribocorrosion behavior and degradation mechanism of 316L stainless steel (SS) in alkaline solution were studied. The SS was worn in 0.1 mol/L NaOH solution with different potentials to investigate the synergism between wear and corrosion. The SS showed larger material loss at a cathodic potential of − 0.8 V and lower material loss at anodic potential when compared with that under pure wear condition. This was inverse when compared with that in other corrosive media, such as H2SO4 and NaCl solutions. The formation of tribo-films with different properties at different potentials played decisive role in the degradation process. Tribo-films were formed at cathodic potential (− 0.8 V) and anodic potentials (0 and 0.4 V). The tribo-film formed at − 0.8 V had the highest O content and was very brittle. It resulted in the easily peeling off of the film and then the acceleration of material degradation. By contrast, the tribo-films formed at anodic potentials were more complete and the O content was much lower. Such kind of ductile tribo-films could protect the SS from wear. The locally high concentration of OH− produced in the reduction reaction of oxygen at − 0.8 V could react with the tribo-film which consist of nano-particles (NPs) with high chemical activity and finally led to deep oxidation and embrittlement of the film. The passivation of the NPs at the anodic potentials could inhibit the oxidation of tribo-film to maintain its ductility.













Similar content being viewed by others
References
Y. Sun, R. Bailey, Corros. Sci. 139, 197 (2018)
C.M. Abreu, M. Cristóbal, R. Losada, X. Nóvoa, G. Pena, M. Pérez, Electrochim. Acta 49, 3049 (2004)
J. Chen, Q. Zhang, Q. Li, S. Fu, J. Wang, T. Nonferr, Metal. Soc. 24, 1022 (2014)
B.A. Obadele, A. Andrews, M.B. Shongwe, P.A. Olubambi, Mater. Chem. Phys. 171, 239 (2016)
Y. Wang, J. Zhang, S. Zhou, Y. Wang, Q. Xue, Appl. Surf. Sci. 528, 147061 (2020)
L. Tan, Z. Wang, Y. Ma, Y. Yan, L. Qiao, Mater. Res. Express 8, 086501 (2021)
A. Dalmau, C. Richard, A.I. Muñoz, Tribol. Int. 121, 167 (2018)
S.M.P. Jemmely, D. Landolt, Wear 237, 63 (2000)
A. de Frutos, M.A. Arenas, G.G. Fuentes, R.J. Rodríguez, R. Martínez, J.C. Avelar-Batista, J.J. de Damborenea, Surf. Coat. Technol. 204, 1623 (2010)
N. Diomidis, J.P. Celis, P. Ponthiaux, F. Wenger, Wear 269, 93 (2010)
Q. Zeng, Y. Xu, Mater. Today Commun. 24, 101261 (2020)
Y. Sun, V. Rana, Mater. Chem. Phys. 129, 138 (2011)
A. Dalmau, W. Rmili, C. Richard, A. Igual-Muñoz, Wear 368–369, 146 (2016)
Y. Liao, R. Pourzal, M.A. Wimmer, J.J. Jacobs, A. Fischer, L.D. Marks, Science 334, 1687 (2011)
L. Tan, Z. Wang, Y. Ma, Acta Metall. Sin. -Engl. Lett. 34, 813 (2021)
J. Perret, E. Boehm-Courjault, M. Cantoni, S. Mischler, A. Beaudouin, W. Chitty, J.P. Vernot, Wear 269, 383 (2010)
M. Favero, P. Stadelmann, S. Mischler, J. Phys. D: Appl. Phys. 39, 3175 (2006)
Y. Sun, E. Haruman, Corros. Sci. 53, 4131 (2011)
R. Büscher, A. Fischer, Wear 254, 1318 (2003)
D. Bitounis, J. Pourchez, V. Forest, D. Boudard, M. Cottier, J.P. Klein, Biomaterials 76, 302 (2016)
A. López, R. Bayón, F. Pagano, A. Igartua, A. Arredondo, J.L. Arana, J.J. González, Wear 338–339, 1 (2015)
A. Atrens, B. Baroux, M. Mantel, J. Electrochem. Soc. 144, (1997).
J. Huang, X. Wu, E.H. Han, Corros. Sci. 52, 3444 (2010)
I. Betova, M. Bojinov, T. Laitinen, K. Mäkelä, P. Pohjanne, T. Saario, Corros. Sci. 44, 2695 (2002)
P. Radhakrishnamurty, P. Adaikkalam, Corros. Sci. 13, 753 (1982)
J.A. Richardson, Shreir’s Corrosion 2, 1191 (2010)
C. Man, C. Dong, Z. Cui, K. Xiao, Q. Yu, X. Li, Appl. Surf. Sci. 427, 763 (2018)
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China under grant No. 51801016, the Natural Science Foundation of Chongqing under grant No. cstc2019jcyj-msxmX0134, the Science and Technology Research Program of Chongqing Municipal Education Commission under grant No. KJQN201901132, the University Innovation Research Group of Chongqing under grant No. CXQT20023 and the Scientific Research Foundation of Chongqing University of Technology under grant No. 2019ZD02.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors state that there are no conflicts of interest to disclose.
Additional information
Available online at http://link.springer.com/journal/40195.
Rights and permissions
About this article
Cite this article
Zou, J., Wang, Z., Ma, Y. et al. Tribocorrosion Behavior and Degradation Mechanism of 316L Stainless Steel in Alkaline Solution: Effect of Tribo-Film. Acta Metall. Sin. (Engl. Lett.) 35, 1365–1375 (2022). https://doi.org/10.1007/s40195-022-01374-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40195-022-01374-x