Abstract
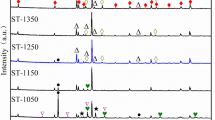
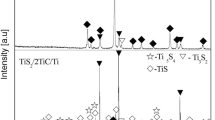
A novel simple method is presented to synthesize high-purity Ti2SC powder using Ti/C/S and Ti/C/TiS2 systems by microwave hybrid heating at different temperatures in argon atmosphere. It was confirmed that the synthesis temperature is strongly dependent on the starting composition. For Ti/C/S system, Ti2SC with small amounts of TiS and TiC was synthesized at 1200 °C. For Ti/C/TiS2 system, high-purity Ti2SC was synthesized at 800 °C and above. The synthesis of Ti2SC powder at low temperature was attributed to the combination of microwave effect by microwave hybrid heating and the introduction of TiS2 as sulfur source. Scanning electron microscopy (SEM) analysis indicated that the layered structure of Ti2SC particles is perfectly formed at 1100 °C, and the crystal particle size approaches to homogeneity which is about 2–5 μm. It was presumed that the formation mechanism of Ti/C/TiS2 system is that TiS2 firstly reacts with Ti to form Ti–S intermetallics, then Ti–S intermetallics reacts with un-reacted Ti and graphite to produce Ti2SC.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Change history
02 March 2017
An erratum has been published.
References
Barsoum MW. The MN+1AXN phases: A new class of solids: Thermodynamically stable nanolaminates. Prog Solid State Ch 2000, 28: 201–281.
Sun ZM. Progress in research and development on MAX phases: A family of layered ternary compounds. Int Mater Rev 2011, 56: 143–166.
Barsoum MW, Brodkin D, El-Raghy T. Layered machinable ceramics for high temperature application. Scripta Mater 1997, 36: 535–541.
Barsoum MW, Radovic M. Elastic and mechanical properties of the MAX phases. Annu Rev Mater Res 2011, 41: 195–227.
Barsoum MW, El-Raghy T. The MAX phases: Unique new carbide and nitride materials—Ternary ceramics turn out to be surprisingly soft and machinable, yet also heat-tolerant, strong and lightweight. Am Sci 2001, 89: 334–343
Jovic VD, Jovic BM, Gupta S, et al. Corrosion behavior of select MAX phases in NaOH, HCl and H2SO4. Corros Sci 2006, 48: 4274–4282.
Barsoum MW, El-Raghy T. A progress report on Ti3SiC2, Ti3GeC2, and the H-phases, M2BX. J Mater Synth Proces 1997, 5: 197–216.
Lin Z, Zhuo M, Zhou Y, et al. Microstructures and theoretical bulk modulus of layered ternary tantalum aluminum carbides. J Am Ceram Soc 2006, 89: 3765–3769.
Barsoum MW, Yaroschuck G, Tyagi S. Fabrication and characterization of M2SnC (M = Ti, Zr, Hf and Nb). Scripta Mater 1997, 37: 1583–1591.
Barsoum MW, El-Raghy T. Synthesis and characterization of a remarkable ceramic: Ti3SiC2. J Am Ceram Soc 1996, 79: 1953–1956.
Nowotny VH. Strukturchemie einiger Verbindungen der Übergangsmetalle mit den elementen C, Si, Ge, Sn. Prog Solid State Ch 1971, 5: 27–70.
Scabarozi TH, Amini S, Finkel P, et al. Electrical, thermal, and elastic properties of the MAX-phase Ti2SC. J Appl Phys 2008, 104: 033502.
Amini S, Barsoum MW, El-Raghy T. Synthesis and mechanical properties of fully dense Ti2SC. J Am Ceram Soc 2007, 90: 3953–3958.
Kulkarni SR, Vennila RS, Phatak NA, et al. Study of Ti2SC under compression up to 47 GPa. J Alloys Compd 2008, 448: L1–L4.
Kulkarni SR, Merlini M, Phatak N, et al. Thermal expansion and stability of Ti2SC in air and inert atmospheres. J Alloys Compd 2009, 469: 395–400.
Gupta S, Amins S, Filimonov D, et al. Tribological behavior of Ti2SC at ambient and elevated temperatures. J Am Ceram Soc 2007, 90: 3566–3571.
Zhu WB, Song JH, Mei BC. Kinetics and microstructure evolution of Ti2SC during in situ synthesis process. J Alloys Compd 2013, 566: 191–195.
Wan FF. Study on preparation and properties of ternary layered carbide Ti2SC. M.S. Thesis. Wuhan, China: Wuhan University of Technology, 2010.
Chen K, Ye Q, Zhou J, et al. Synthesis of Ti2SC phase using iron disulfide or iron sulfide post-treated with acid. J Am Ceram Soc 2015, 98: 1074–1079.
Li X, Liang B, Li Z. Combustion synthesis of Ti2SC. Int J Mater Res 2013, 104: 1038–1040.
Liang B, Wang L, Wang Z, et al. Synthesis of Ti2SC material by self-propagation high temperature synthesis. Materials Science and Engineering of Powder Metallurgy 2013, 18: 675–679. (in Chinese)
Wang Q, Hu C, Cai S, et al. Synthesis of high-purity Ti3SiC2 by microwave sintering. Int J Appl Ceram Tec 2014, 11: 911–918.
Liang B, Wang Y, Zhang W, et al. Synthesis of ternary titanium aluminum carbides using microwave synthesis technique. Journal of Ceramics 2015, 36: 476–480. (in Chinese)
Thompson P, Cox DE, Hasting JB. Rietveld refinement of Debye–Scherrer synchrotron X-ray data from Al2O3. J Appl Cryst 1987, 20: 79–83.
Yadoji P, Peelamedu R, Agrawal D, et al. Microwave sintering of Ni–Zn ferrites: Comparison with conventional sintering. Mat Sci Eng B 2003, 98: 269–278.
Clark DE, Folz DC, West JK. Processing materials with microwave energy. Mat Sci Eng A 2000, 287: 153–158.
Wu E, Gray EMA, Kisi EH. Modelling dislocation-induced anisotropic line broadening in Rietveld refinements using a Voigt function. I. General principles. J Appl Cryst 1998, 31: 356–362.
Barin I. Thermochemical Data of Pure Substances. Cheng NL, Niu ST, Xu GY, Translation. Beijing: Science Press, 2003. (in Chinese)
Acknowledgements
This work was supported by Natural Science Fundamental Research of Education Department of Henan Province (Nos. 14A430032 and 15A430021) and Plan of Natural Science Fundamental Research in Henan University of Technology (No. 2013JCYJ06).
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
Open Access The articles published in this journal are distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Guan, C., Sun, N. Synthesis of high-purity Ti2SC powder by microwave hybrid heating. J Adv Ceram 5, 337–343 (2016). https://doi.org/10.1007/s40145-016-0207-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40145-016-0207-z