Abstract
Presbyopia is a condition that affects many as the lens ages, and is a secondary effect of cataract surgery. Multifocal lenses for the treatment of presbyopia have been undergoing cycles of development and improvement over the past two decades. The latest advancements have yielded lenses with a continuous range of optical powers, otherwise known as continuous transitional focus (CTF) lenses. Modern CTF lenses provide a more natural experience for patients while minimizing unwanted optical side effects.
Similar content being viewed by others
Introduction
Presbyopia is an ophthalmic condition in which the eye undergoes progressive degeneration and loses the ability to focus clearly on objects, particularly at near vision—this is due to physiological changes that are generally caused by the aging of the eye. These changes are related to the decrease in elasticity of the lens that leads to an inability to respond to ciliary muscle contraction, resulting in the loss of the accommodating power of the lens [1]. Spectacles have been widely used to correct presbyopia, however, the development of more permanent, spectacle-free options, such as intraocular lenses (IOLs), have proved to be an attractive alternative. The use of IOLs is especially advantageous in cases in which surgery is required to remove the damaged original lens (for example, to treat cataract) which then has to be replaced by an artificial one in order to restore vision.
The pioneering operation resulting in a successful implantation of the first artificial acrylic lens was performed by Dr. Harold Ridley on 29 November 1949 [2]. Prior to this first implantation surgery, a number of questions had to be addressed regarding the appropriate material and design for the IOL, as well as the techniques for performing the required surgery and fixing the IOL in place [3]. While ophthalmic surgeons have historically been cautious with new materials and technologies, scientific focus and corresponding progress has been accelerating in recent years, resulting in a more advanced landscape. Currently, there are several different types of IOLs commercially available, and the surgery itself and implantation of IOLs have become routine procedures for correcting a range of conditions. Nevertheless, after nearly 70 years and 200 million IOL surgeries, ophthalmic surgeons still find themselves trying to better address the same concerns faced by Harold Ridley leading up to the first IOL implantation [4].
This brief review describes the evolution of currently available IOLs to address presbyopia, and a look toward new technologies, such as continuous transition focus (CTF) lenses, is highlighted. This article is based on previously conducted studies and does not contain any studies with human participants or animals performed by any of the authors.
Evolution of Presbyopic IOLs
IOLs have historically been indicated for treatment of lens opacification, and the materials and design have evolved over the last 70 years. The first lenses were rigid and made from PMMA (poly methyl methacrylate), because Dr. Ridley had observed that PMMA shrapnel remained benign in the eyes of World War II pilots. With the advent of phacoemulsification, which allowed for much smaller incisions into the eye for lens removal, foldable lenses were developed to allow placement through a small hole. Modern IOLs, made from silicon or intelligently designed polymers, have been found to provide excellent optical results and a high degree of patient satisfaction [5].
Whether or not presbyopia is present before IOL implantation, it is a frequent outcome following surgery, because virtually all IOLs are unable to adjust in response to patients’ ciliary muscle contraction. Early lenses used for treating presbyopia offered single focus corrections, either for distance or near vision correction. In many cases patients often received different powered lenses in each eye (one for distance and one for near) in order to accommodate for both conditions. If only one type of lens were to be used in both of the eyes, the patients would have required additional spectacles to satisfy the need for correcting near vision. While effective, these options are less than ideal, resulting in reduced patient satisfaction. Alternatively, IOLs designed to compensate for lack of accommodation have been developed to correct presbyopia, whether it is the principle concern facing a patient or incidental to cataract surgery.
The first multifocal IOL was developed by Precision-Cosmet (Buffalo, NY, USA), later transferred to IOLAB Corporation (CA, USA) and finally Bausch & Lomb (Rochester, NY, USA), who marketed the lens as NuVue. The novel design of NuVue facilitated two focal points [6]. Multiple focal points were created by combining two radii in a single lens in a bullseye pattern, where the center of the bullseye had one power (power A), the first ring had a second power (power B), the next ring power A, then B, and so on. The result is two focal points simultaneously present in the eye: one for distance viewing and another for near. The relative contribution of each focus is modulated by the pupil diameter under bright and dark lighting conditions. Other similarly designed lenses were soon developed, with more than two focal points, including the TrueVista 68STUV, from Storz, with three focal points [6, 7].
Another modality of IOL multifocality is the use of a rotationally asymmetric refractive profile containing an aspheric distance-vision zone combined with a sector-shaped near-vision zone in the inferior area of the IOL. This concept is the basis of the multifocal IOL Lentis Mplus LS-312 (Oculentis GmbH, Berlin, Germany). Studies of this IOL have shown good near and distance visual outcomes, combined with postoperative contrast sensitivity within physiological ranges and a positive impact on patient quality of life [8,9,10]. Other studies, however, have described an increase in optical aberrations after the implantation of the sectorial asymmetric IOL Mplus (Oculentis GmbH, Berlin, Germany), namely (i) the coma, a third-order Zernike aberration, associated with the presence of a high-power refractive sector in the inferior part of the lens optical zone [11], and (ii) decreased contrast sensitivity [12].
The first IOL approved by the USFDA (United States Food and Drug Administration) for treatment of presbyopia was the Array lens manufactured by (the former) Advanced Medical Optics (now Johnson & Johnson Vision). In terms of visual acuity, the Array IOL performed very well in clinical trials and reported a high level of patient satisfaction [13, 14]. The Array lens, with its zones of near and distant foci, was the predecessor to ReZoom (Advanced Medical Optics, Inc., Santa Ana, CA, USA), a hydrophobic acrylic multifocal IOL that contains five zones of focus which accommodate simultaneously both distance and near, under a variety of lighting conditions [15].
Alternating rings of optical power is not the only way to produce multiple focal points from a single lens. Diffractive lenses, as a special configuration of the Fresnel lens, function by combining a refractive lens with a cylindrically symmetric diffractive pattern. The diffractive pattern acts effectively as a second lens for a portion of the light, resulting in multiple focal points. Because a cylindrically symmetrical diffractive pattern is a series of concentric rings, diffractive lenses look similar under microscopy to their refractive counterparts, but the underlying physics of operation is very different. The first diffractive IOLs in the United States were the 3M 815LE (3M Healthcare) and the Pharmacia 811E (Advanced Medical Optics, Inc., Santa Ana, CA, USA). The 815LE was purchased by Alcon (Fort Worth, TX, USA) and later named the ReSTOR, the first diffractive IOL to be USFDA approved.
One of the key challenges and complaints with all IOLs—especially multifocal lenses—is related to the presence of unwanted optical phenomena such as halos and glare. Halos and glare appear to patients when light reflects or diffracts off the sharp edges on the lens [16]. Early IOLs were designed with very soft and rounded edges with the goal of avoiding irritation of the capsular bag. Unfortunately, the soft and round edges can lead to the unintended consequence of posterior capsule opacification (PCO) [17]. Changes in design and manufacturing to produce sharp edges on the lens inhibited PCO, but also increased the likelihood of glare and halos under certain conditions. With both refractive and diffractive multifocal IOLS, concentric circles are fabricated into the lens surface, with sharp discrete boundaries. These boundaries likewise produce glare and halos. Efforts to limit these phenomena have been made by utilizing an aspheric transition between boundaries.
Strengths and Weaknesses of Presbyopic IOLs on the Market
In their 2012 review, Lichtinger and Rootman elegantly highlight the more prominent technological improvements made in the development of presbyopic IOLs [6]. Advancements have mostly been targeted towards enhancing high-level uncorrected and corrected focus for both distance and near vision, as demonstrated for Array multifocal IOLs (Allergan, Irvine, USA) and ReZoom (Advanced Medical Optics), ReSTOR (Alcon), and M-Flex 630F +3 (Rayner). Additionally, Tecnis lenses (Abbott Medical Optics) are designed to split the light among near and distance focus regardless of the pupil size, resulting in a demonstrated superior performance in low light conditions compared with other multifocal lenses [18]. Other patient studies have also shown relative “pupil independency” leading to better patient acceptance [19]. In addition, ReSTOR toric lenses incorporate the astigmatic correction to provide a single platform to correct astigmatism and improve near and intermediate vision [6].
Despite the tremendous advancements made in the field of IOL design and manufacturing, leading to improved presbyopia and visual acuity outcomes, none of the above described IOLs currently on the market remain problem-free. With ever-growing patient expectations for visual outcomes following surgery with IOL implantation, surgeons are often faced with reports of unwanted dystopic phenomena, such as halos and glare. This continues to be an issue regardless of the type of IOL used [20,21,22,23]. Other disadvantages, particularly associated with currently available accommodative IOL, Crystalens (Bausch + Lomb, Rochester, NY, USA), are related to tilting and decentration of the lens caused by capsular contraction and fibrosis [24].
Extended Range of Vision (ERV) Lenses
ERV lenses use diffractive echelettes, embedded on the optical surface of the lens, which theoretically extend the depth of focus and create a greater range of useful uncorrected vision. Negative dispersion refractive technology is also used to adjust for chromatic and spherical aberration, improving quality of the image on the retina, by collapsing the aberration curve as available light increases. These refractive and diffractive technologies were used in the Tecnis Symfony Extended Range of Vision (ERV) IOL (Johnson and Johnson Vision, Santa Ana, CA, USA). A recent study of 176 eyes comparing ERV lenses to monofocal IOLs demonstrated no difference between groups in uncorrected binocular visual acuity at distance, however visual acuity at intermediate and near were significantly better in the ERV group [25].
Continuous Transitional Focus (CTF) Lenses
Most multifocal IOL designs, such as bi- and trifocal IOLs, generate multiple foci on the retina to capture the necessary clarity of vision for multiple focal distances. In these types of lens implants, IOL centration assumes critical importance since a decentered IOL, relative to the visual axis, may induce high-order aberrations and decrease post-operative visual function due to “looking through” diffractive rings of the IOL instead of the central optical zone as intended [26]. Thus, decentration could directly lead to a negative impact on the visual outcomes. Additionally, as a result of the distinct foci with different properties in the IOL, severe changes near the transition points could have a negative impact on the patient’s vision, as optical distortions are likely to take place. Alternatively, pupil diameter may also influence the ratio between distance and near additive power provided by the IOL. It has been previously described that patients with a small pupillary diameter (below 3.0 mm) are not good candidates for the traditional asymmetric refractive multifocal IOL, especially the Lentis Mplus Toric (Oculentis GmbH, Berlin, Germany) [26]. Also, patients with a large pupil are considered at increased risk for glare dysfunction, and may experience optical aberrations, diffraction, and photic phenomena such as glare, starbursts, and halos around light sources [27, 28]. A recent study comparing through-focus vision performance and light disturbances of FineVision (PhysIOL, Liège, Belgium) and PanOptix (Alcon Laboratories, Fort Worth, TX, USA) trifocal IOLs to the Symfony extended depth of focus IOL was published by Escandón-García and colleagues in Portugal [29]. They found that the trifocal IOLs grant better performance at near distance while extended depth of focus IOLs performed better at intermediate distance. Additionally, objective dysphotopsia was not reduced in extended depth of focus IOLs as compared to the trifocal IOLs [29].
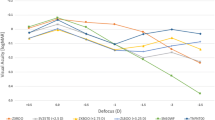
To address the disadvantages of preexisting IOLs, a presbyopic IOL, Precizon Presbyopic IOL (Ophtec BV, Netherlands), has been designed, applying a novel optical approach. Precizon IOL is a hybrid material implant based on hydrophilic acrylic, with a hydrophobic surface modification (Benz25 material), a C-loop design with modified haptics, 12.5 mm of total diameter and a 6.00 mm optical zone. The IOL optic is designated as CTF (“continuous transitional focus”) divided into three concentric sectors: the central sector, of higher diameter, is dedicated to distance correction; two peripheral sectors present a bimodal (50–50%) distribution of distance and near correction, and this distribution changes along four segments in each sector (Fig. 1). This refractive lens provides the ability for a transition in focus between 11 distinct segments (five for distance and six for near vision) within the optical zones. A CTF design creates a continuous defocus curve (Fig. 2) to facilitate a sharp image on the retina from infinity to intermediate, and from intermediate to near focus [30]. Providing smoother transition allows for the potential of quicker neuroadaptation, more closely mimicking the eye’s natural process of accommodation and providing a more natural-like vision.
A multi-zonal design allows the lens to maintain the light distribution and exposure on the foci, regardless of the tilt or decentering of the lens—addressing challenges related to pupil size and decentration of earlier designs [31].
In April 2017, Precizon Presbyopic IOL (Ophtec BV, Groningen, Netherlands) favorably concluded clinical trials (Clinical Trial NCT02409771) and results were presented at the 2017 European Society of Cataract and Refractive Surgeons meeting [32]. The prospective, multicenter European trial included 60 patients scheduled for cataract or refractive lens exchange surgery with a follow-up of 3 months. Investigators reported good uncorrected visual acuity at near, intermediate and distance and 80% of patients achieved spectacle independence, with an additional 18% requiring occasional spectacle use. The majority of patients did not report issues with halos and glare, however approximately one-third of patients did report little to moderate disturbance. The lens delivered a concept of an IOL more tolerant to pupil size and dislocation, receiving its CE marking in May 2017. Additionally, in September 2017, Swiss Advanced Vision launched EDEN, an IOL that uses the Instant Focus EDOF (extended-depth-of-focus) technology that offers continuous vision with minimized dysphotopsia, while preserving resolution and contrast sensitivity.
As with all IOL implantation to manage expectations, patient selection is important, and it is essential to have a perfectly transparent cornea and to rule out progressive ocular diseases, such as glaucoma or maculopathy, as well as moderate-to-severe amblyopia, strabismus and abnormal binocular vision. Notably, progressive systemic diseases such as diabetes or autoimmune diseases are contraindications for multifocal IOLs.
Mechanism and Technology of CTF
CTF lenses are a logical evolution of the previously commercialized bifocal, trifocal, and multifocal lenses. CTF lenses can be made by combining multiple optical elements. Diffractive lenses have multiple focal points corresponding to the diffraction order. When combined with a refractive lens, these focal points are a series where (if fr is the refractive focus and fd is the first-order diffractive focus) [33]:
The number of focal points produced by a diffractive lens depends on the specific details of photonic engineering, including the number of concentric rings and the pattern of their spacing. As more focal points are added to a diffractive lens, combined with a refractive lens, the difference between the focal points becomes indistinguishable to the eye, producing an apparent continuous gradient of focus on the retina. Despite precision of design and fabrication, benefits may be limited by unwanted optical effects due to sharp edges.
Alternatively, CTF lenses can be fabricated through a reexamination of purely refractive designs. The original bifocal refractive lenses were manufactured by lathe-cutting concentric rings with different optical powers, producing very precise and sharp edges [34]. The Precizon Presbyopic IOL is manufactured differently than conventional lathe-cutting, by arranging varying powers over the lens area in a non-cylindrically symmetric fashion.
CTF lenses can provide focus within the entire depth of field, meaning that the image more closely mimics the visual experience of the natural lens. Because the focal points are not limited to one or two discrete locations, but graduated over a larger area, some alignment issues are reduced, making placement of the lens “more forgiving” and less sensitive to pupil size.
Conclusion
Since IOLs were originally conceived, they have been on a path of continual improvement in materials and design, always with the goal of enhancing visual outcomes and patient quality of life, while limiting complications following surgery. As with many journeys of technological development, the path has been nonlinear, and the latest developments in IOLs for treatment of presbyopia are no exception. Layers of technology with corresponding challenges in manufacturing have been added with success, and much has been learned regarding unintended optical phenomena. However, as each layer of complexity is added, the required uniformity and precision of the subject increases. It should be noted that the human eye is not a precision piece of laser optics but contains natural deviation from uniformity. Optimally performing IOLs for treatment of presbyopia allow for achievement of full spectacle independence, with continual focus from distant to near, while reducing unwanted optical effects. Further developments of CTF lens technology, with improvements in the distance central area are anticipated for a further improvement of the distance visual acuity and contrast sensitivity.
References
Glasser A, Campbell MCW. Presbyopia and the optical changes in the human crystalline lens with age. Vis Res. 1998;38(2):209–29.
Auffarth GU, Apple DJ. History of the development of intraocular lenses. Der Ophthalmologe: Zeitschrift der Deutschen Ophthalmologischen Gesellschaft. 2001;98(11):1017–28.
Artal P, et al. Compensation of corneal aberrations by the internal optics in the human eye. J Vis. 2001;1(1):1.
O’Conner A. Harold Ridley, Eye doctor, 94, early developer of lens implants. In: New York Times. 2001. Available at: https://www.nytimes.com/2001/06/06/world/harold-ridley-eye-doctor-94-early-developer-of-lens-implants.html. Accessed 6 June 2018.
Tetz M, Jorgensen MR. New hydrophobic IOL materials and understanding the science of glistenings. Curr Eye Res. 2015;40(10):969–81.
Lichtinger A, Rootman DS. Intraocular lenses for presbyopia correction: past, present, and future. Curr Opin Ophthalmol. 2012;23(1):40–6.
Greer RB. Presbyopia lens surgery: a clinical guide to current technology. Optom Vis Sci. 2008;85(9):793.
Ramón ML, Piñero DP, Pérez-Cambrodí RJ. Correlation of visual performance with quality of life and intraocular aberrometric profile in patients implanted with rotationally asymmetric multifocal IOLs. J Refract Surg. 2012;28(2):93–9.
van der Linden JW, et al. Comparison of a new-generation sectorial addition multifocal intraocular lens and a diffractive apodized multifocal intraocular lens. J Cataract Refract Surg. 2012;38(1):68–73.
McNeely RN, et al. Visual quality and performance comparison between 2 refractive rotationally asymmetric multifocal intraocular lenses. J Cataract Refract Surg. 2017;43(8):1020–6.
Montés-Micó R, et al. In vitro optical performance of nonrotational symmetric and refractive–diffractive aspheric multifocal intraocular lenses: impact of tilt and decentration. J Cataract Refract Surg. 2012;38(9):1657–63.
Rosa AM, et al. Comparison of visual function after bilateral implantation of inferior sector-shaped near-addition and diffractive–refractive multifocal IOLs. J Cataract Refract Surg. 2013;39(11):1653–9.
Steinert RF, et al. A prospective comparative study of the AMO ARRAY zonal-progressive multifocal silicone intraocular lens and a monofocal intraocular lens. Ophthalmology. 1999;106(7):1243–55.
Sen HN, et al. Quality of vision after AMO Array multifocal intraocular lens implantation. J Cataract Refract Surg. 2004;30(12):2483–93.
Chang DF. Mastering refractive IOLs: the art and science. Thorofare: SLACK Incorporated; 2008.
Erie JC, Bandhauer MH. Intraocular lens surfaces and their relationship to postoperative glare. J Cataract Refract Surg. 2003;29(2):336–41.
Kohnen T, et al. Optic edge design as long-term factor for posterior capsular opacification rates. Ophthalmology. 2008;115(8):1308–1314.e3.
Hütz WW, et al. Reading ability with 3 multifocal intraocular lens models. J Cataract Refract Surg. 2006;32(12):2015–21.
Sood P, Woodward MA. Patient acceptability of the Tecnis® multifocal intraocular lens. Clin Ophthalmol (Auckland, NZ). 2011;5:403.
Petermeier K, et al. Effect of +3.00 diopter and +4.00 diopter additions in multifocal intraocular lenses on defocus profiles, patient satisfaction, and contrast sensitivity. J Cataract Refract Surg. 2011;37(4):720–6.
de Vries NE, et al. Dissatisfaction after implantation of multifocal intraocular lenses. J Cataract Refract Surg. 2011;37(5):859–65.
de Vries NE, Nuijts RM. Multifocal intraocular lenses in cataract surgery: literature review of benefits and side effects. J Cataract Refract Surg. 2013;39(2):268–78.
Kamiya K, et al. Multifocal intraocular lens explantation: a case series of 50 eyes. Am J Ophthalmol. 2014;158(2):215–220.e1.
Cazal J, et al. Accommodative intraocular lens tilting. Am J Ophthalmol. 2005;140(2):341–4.
Hogarty DT, et al. Comparing visual acuity, range of vision and spectacle independence in the extended range of vision and monofocal intraocular lens. Clin Exp Ophthalmol. 2018. https://doi.org/10.1111/ceo.13310
Park CY, Oh SY, Chuck RS. Measurement of angle kappa and centration in refractive surgery. Curr Opin Ophthalmol. 2012;23(4):269–75.
Wang M, et al. Pupil influence on the visual outcomes of a new-generation multifocal toric intraocular lens with a surface-embedded near segment. J Refract Surg. 2016;32(2):90–5.
Salati C, et al. Pupil size influence on the intraocular performance of the multifocal AMO-array intraocular lens in elderly patients. Eur J Ophthalmol. 2007;17(4):571–8.
Escandón-García S, et al. Through-focus vision performance and light disturbances of 3 new intraocular lenses for presbyopia correction. J Ophthalmol. 2018;2018:6165493.
CTF?! A Completely Different Approach! 2017. http://theophthalmologist.com/issues/0917/ctf-a-completely-different-approach.
Kim M-J, et al. Evaluation of optical performance of 4 aspheric toric intraocular lenses using an optical bench system: influence of pupil size, decentration, and rotation. J Cataract Refract Surg. 2015;41(10):2274–82.
Holzer M. Functional outcomes and patient satisfaction of the new Precizon presbyopic multifocal intraocular lens. In: Presented at European society of cataract and refractive surgeons meeting (ESCRS) 2017; Lisbon, Portugal. Available at: http://forum.escrs.org/escrs-presentations-and-videos/functional-outcomes-and-patient-satisfaction-of-the-new-precizon-presbyopic-multifocal-intraocular-lens. Accessed 6 June 2018.
Herzig HP. Design of refractive and diffractive micro-optics. In: Martellucci S, Chester AN, editors. Diffractive optics and optical microsystems. New York: Springer; 2013.
Kunzler WF. Mold for and method of making contact and intraocular lenses. In: Google Patents; 1988. Available at: https://patents.google.com/patent/EP0325673A3/un. Accessed 6 June 2016.
Acknowledgements
Funding
No funding or sponsorship was received for this study or publication of this article. The article processing charges were funded by the authors.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Disclosures
Ramón Ruiz Mesa and Tiago Monteiro have nothing to disclose. All tables and figures are original and have been produced by the authors for this manuscript.
Compliance with Ethics Guidelines
This article is based on previously conducted studies and does not contain any studies with human participants or animals performed by any of the authors.
Open Access
This article is distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/), which permits any noncommercial use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Author information
Authors and Affiliations
Corresponding author
Additional information
Enhanced digital features
To view enhanced digital features for this article go to https://doi.org/10.6084/m9.figshare.6396065.
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Mesa, R.R., Monteiro, T. Continuous Transitional Focus (CTF): A New Concept in Ophthalmic Surgery. Ophthalmol Ther 7, 223–231 (2018). https://doi.org/10.1007/s40123-018-0134-x
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40123-018-0134-x