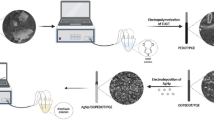
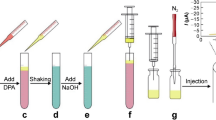
Abstract
Esterase enzymes play an important role in biology because they are responsible for the hydrolysis of choline esters. In their absence, the original state of the post synaptic membranes cannot be reestablished. Therefore, the aim of the work is to study the inhibiting action exerted by the group of compounds on these enzymes. Among these class of inhibiting compounds, pesticides are important because of the potential danger as a result of their large scale use in agriculture. Pesticides are generally determined using liquid or gas chromatography methods with various detection techniques. These methods are very sensitive and discriminating, however they require sample pretreatment such as extraction, preconcentration and clean up, which are skilled techniques and high cost treatment and also time consuming. In this study, acetyl cholinesterase and butyrylcholinesterase based biosensors have emerged as a promising tool for the detection and characterization of pesticides which are inhibitors of these enzymes. Although the physiological function of butyrylcholinesterase in comparison with acetyl cholinesterase is ambiguous, it has larger substrate specificity towards choline esters. Therefore, the development of a more selective electrode against choline, can lead to more sensitive determination of the inhibitor being investigated. Hence in the present work, a method based on inhibition of butyrylcholinesterase was attempted for quantification of carbofuran on the basis of cholinesterase inhibition. Butyrylcholinesterase with an activity of 10.2 units/mg was immobilized on a solid surface by cross linking with glutaraldehyde. The immobilized system was calibrated by correlating the inhibition of the butyrylcholinesterase activity with varying concentrations of the butyryl choline chloride and carbofuran. The sensing mechanism was investigated for its response to carbofuran concentrations ranging from 125 to 1,000 ppm. The effects of butyryl choline chloride concentration on the response of the sensing strip were also determined. Through this work, a sensing element made up of a carbon based electrode, containing the immobilized butyrylcholinesterase, has been described. This element has certain practical advantages like smaller size, user friendly, easy portability and disposability.










Similar content being viewed by others
References
V. Simeon, C.E. Reiner, A. Vernon, Effect of temperature and pH on carbamoylation and phosphorylation of serum cholinesterases. Biochem. J. 130, 515 (1972)
T. Imato, N. Ishibashi, Potentiometric butyrylcholine sensor for organophosphate pesticides. Biosens. Bioelectron. 10, 435 (1995)
S. Kumaran, M. Morita, Application of a cholinesterase biosensor to screen for organophosphorus pesticides extracted from soil. Talanta 42, 649 (1995)
J.D. Faxon, A.L. Ghindilis, P. Atanasov, E. Wilkins, Direct electron transfer based on tri-enzyme electrode for monitoring of organophosphorus pesticide. Sens. Actuat. B 35–36, 448 (1996)
Y.A. Cho, S.H. Lee, G.S. Cha, Y.T. Lee, Fabrication of butyrylcholinesterase sensor using polyurethane-based ion selective membrane. Biosens. Bioelectron. 14(4), 435 (1999)
P. Mulchandani, A. Mulchandani, I. Kaneva, W. Chen, Biosensor for direct determination of organophosphate nerve agents. 1. Potentiometric enzyme electrode. Biosens. Bioelectron. 14, 77 (1999)
S. Andreescu, J.-L. Marty, Twenty years research in cholinesterase biosensors—from basic research to practical application. Biomol. Eng. 23, 1 (2006)
H. Mohammadi, A. Amine, S. Cosine, C. Mousty, Mercury-enzyme inhibition assays with an amperometric sucrose biosensor based on a trienzymatic-clay matrix. Anal. Chim. Acta 543, 143 (2005)
A. Singh, Enzyme Assays (Regency Publications, New Delhi, 2007), p. 199
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dey, M., Bhuvanagayathri, R. & Daniel, D.K. Biosensor based on Butyrylcholinesterase for Detection of Carbofuran. J. Inst. Eng. India Ser. E 96, 9–14 (2015). https://doi.org/10.1007/s40034-015-0060-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40034-015-0060-6