Abstract
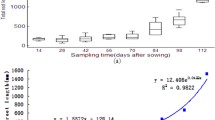
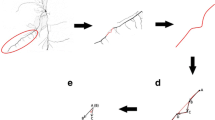
A method consisting of soil core with undisturbed sample of root architecture, digitizer and commercial software was developed to visualize wheat root system architecture (RSA) in a field experiment. At first, root-zone soil was excavated and wheat RSA was digitized in every 5-mm-thick soil layers. By then, the 3D root morphological data were re-integrated by Pro-E software, where 3D models of wheat RSAs could be reconstructed, scaled and calculated. Results revealed markedly distorted morphological structures of wheat axial roots in both curvature and helical morphology. Scaled RSA models also revealed severe restrictions on overall RSAs, illustrating the intense constraint of root topology by the heterogeneous environment. Time-series presentation of RSAs clearly displayed the fine structural details of root morphological dynamics, indicating that a high accuracy of data had been achieved for field crop physiology. Laminated digitization combined with Pro-E modelling was featured as a deterministic, data-driven and model-assisted procedure, which was particularly suitable for crop RSAs in field. Wheat RSA dynamics and temporal variations of 3D root topology illustration could be realized by undisturbed sampling, digitizing and modelling method for field-state crops with fine and fibrous root systems. It had potential to provide a means of illustrating the influence of anisotropy of the soil environment.




Similar content being viewed by others
References
A.H. Fitter, An architectural approach to the comparative ecology of plant root systems. New Phytol. 106, 61–77 (1987)
J. Lynch, Root architecture and plant productivity. Plant Physiol. 109, 7–13 (1995)
P. Armengaud, K. Zambaux, A. Hills, R. Sulpice, R.J. Pattison, M.R. Blatt, A. Amtmann, EZ-Rhizo: integrated software for the fast and accurate measurement of root system architecture. Plant J. 57, 945–956 (2009)
Y. Uga, K. Sugimoto, S. Ogawa, J. Rane, M. Ishitani, N. Hara, Y. Kitomi, Y. Inukai, K. Ono, N. Kanno, H. Inoue, H. Takehisa, R. Motoyama, Y. Nagamura, J. Wu, T. Matsumoto, T. Takai, K. Okuno, M. Yano, Control of root system architecture by DEEPER ROOTING1 increases rice yield under drought conditions. Nat. Genet. 45, 1097–1102 (2013)
M.B. Jackson, W. Armstrong, Formation of aerenchyma and the processes of plant ventilation in relation to soil flooding and submergence. Plant Biol. 1(3), 274–287 (1999)
S. Trachsel, S.M. Kaeppler, K.M. Brown, J.P. Lynch, Shovelomics: high throughput phenotyping of maize (Zea mays L.) root architecture in the field. Plant Soil 341, 75–87 (2011)
U. Rascher, S. Blossfeld, F. Fiorani, Non-invasive approaches for phenotyping of enhanced performance traits in bean. Funct. Plant Biol. 38, 968–983 (2011)
R.J. Flavel, C.N. Guppy, M. Tighe, M. Watt, A. McNeill, I.M. Young, Non-destructive quantification of cereal roots in soil using high-resolution X-ray tomography. J. Exp. Bot. 63, 2503–2511 (2012)
S. Mairhofer, S. Zappala, T.R. Saoirse, C. Sturrock, M. Bennett, S.J. Mooney, RooTrak: automated recovery of three-dimensional plant root architecture in soil from X-ray microcomputed tomography images using visual tracking. Plant Physiol. 158, 561–569 (2012)
S. Mooney, T. Pridmore, J. Helliwell, M. Bennett, Developing X-ray computed tomography to non-invasively image 3-D root systems architecture in soil. Plant Soil 352, 1–22 (2012)
R.O. Kuchenbuch, H.H. Gerke, U. Buczko, Spatial distribution of maize roots by complete 3D soil monolith sampling. Plant Soil 315, 297–314 (2009)
J. Wu, L. Pagès, Q. Wu, B. Yang, Y. Guo, Three-dimensional architecture of axile roots of field-grown maize. Plant Soil 387, 363–377 (2015)
X.X. Chen, Q.S. Ding, Z. Błaszkiewicz, J.A. Sun, Q. Sun, R.Y. He, Y.N. Li, Phenotyping for the dynamics of field wheat root system architecture. Sci. Rep. 7, 37649 (2017). https://doi.org/10.1038/srep37649
X.X. Chen, Y.N. Li, R.Y. He, Q.S. Ding, Phenotyping field-state wheat root system architecture for root foraging traits in response to environment × management interactions. Sci. Rep. 8, 2624 (2018). https://doi.org/10.1038/s41598-018-20361-w
M.O. Adu, L. Wiesel, M.J. Bennett, M.R. Broadley, P.J. White, L.X. Dupuy, A scanner system for high-resolution quantification of variation in root growth dynamics of Brassica rapa genotypes. J. Exp. Bot. 65, 2039–2048 (2014)
J. Wu, Y. Guo, An integrated method for quantifying root architecture of field-grown maize. Ann. Bot. 114, 841–851 (2014)
M. Watt, L.J. Magee, M.E. McCully, Types, structure and potential for axial water flow in the deepest roots of field-grown cereals. New Phytol. 178, 135–146 (2008)
L. Pagès, S. Pellerin, Evaluation of parameters describing the root system architecture of field grown maize plants II. Density, length, and branching of first-order lateral roots. Plant Soil 164, 169–176 (1994)
Studio of Erdai LZ. Pro/engineer Wildfire 2.0 Senior Design. Beijing Publishing House of Electronic Industry. (2005). (in Chinese)
J.A. Postma, U. Schurr, F. Fiorani, Dynamic root growth and architecture responses to limiting nutrient availability: linking physiological models and experimentation. Biotechnol. Adv. 32, 53–65 (2014)
F. Tan, L. Tang, C.J. Hu, Three-dimensional morphological modelling and visualization of wheat root system. Chin. J. Appl. Ecol. 22, 137–143 (2011). (in Chinese with English abstract)
A.G. Bengough, A. Castrignana, L. Pagès, M. VanNoordwijk, Sampling strategies, scaling, and statistics, in Root Methods: A Hand book, ed. by A.L. Smit, A.G. Bengough, C. Engels, M. VanNoordwijk, S. Pellerin, S.C. VandeGeijn (Springer, Berlin, 2000), pp. 147–174
H.F. Downie, M.O. Adu, S. Schmidt, W. Otten, L.X. Duputy, P.J. White, T.A. Valentine, Challenges and opportunities for quantifying roots and rhizosphere interactions through imaging and image analysis. Plant Cell Environ. 38, 1213–1232 (2015)
M. Blouin, S. Barot, C. Roumet, A quick method to determine root biomass distribution in diameter classes. Plant Soil 290, 371–381 (2007)
W. Böhm, Methods of Studying Root Systems (Springer, Berlin, 1979)
D.A. Heeraman, N.G. Juma, A comparison of minirhizotron, core and monolith methods for quantifying barley (Hordeum vulgare L.) and fababean (Vicia faba L.) root distribution. Plant Soil 148(1), 29–41 (1993)
M.C.G. Costa, I.M.D.L. Cunha, L.A.D.C. Jorge, I.C.D.S. Araújo, Public-domain software for root image analysis. Rev bras ciênc solo 38, 1359–1366 (2014)
M. Aono, T.L. Kunii, Botanical Tree Image Generation. IEEE Comput. Graph. Appl. 4, 10–34 (1984)
D.L. Bell, S.E. Sultan, Dynamic phenotypic plasticity for root growth in Polygonum: a comparative study. Am. J. Bot. 86, 807–819 (1999)
A. Larigauderie, J.H. Richards, Root proliferation characteristics of seven perennial arid-land grasses in nutrient-enriched microsites. Oecologia 99, 102–111 (1994)
A.P. Hamblin, D. Tennant, H. Cochrane, Tillage and the growth of a wheat crop in a loamy sand. Aust. J. Agric. Res. 33, 887–897 (1982)
M. Dracup, P.J. Gregory, R.K. Belford, Restricted growth of lupin and wheat roots in the sandy a horizon of a yellow duplex soil. Aust. J. Agric. Res. 44, 1273–1290 (1993)
D. Bhaduri, T.J. Purakayastha, Long-term tillage, water and nutrient management in rice–wheat cropping system: assessment and response of soil quality. Soil Tillage Res. 144, 83–95 (2014)
Acknowledgements
Financial support from the State Key Program of China (2016YFD0300900) and Juangsu Agri. Mech. Fund was acknowledged. We are grateful to Zbigniew Błaszkiewicz, the professor of Institute of Biosystems Engineering, Poznań University of Life Sciences, who provided suggestive contributions to the discussions and the critical reading.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chen, X., He, R., Ding, Q. et al. A Digitization and Visualization Procedure for 3D Wheat Root System Architecture in Rice–Wheat Rotation. J. Inst. Eng. India Ser. A 100, 1–8 (2019). https://doi.org/10.1007/s40030-018-0331-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40030-018-0331-6