Abstract
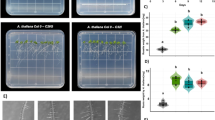
The present work is an attempt to explore immunomodulatory activity of some natural products taking into account the major cellular immune reaction, nodulation, in the short horned grasshopper Poekilocerus pictus. It has been shown that two different groups of natural products i.e. quinones, juglone and plumbagin and azadirachtins, neemazal and TN MP-100 inhibit the nodulation reaction against four different species of bacteria, Staphylococcus aureus, Pseudomonas aeruginosa, Escherichia coli, and bacteria isolated from the gut of P. pictus itself (‘self bacteria’),used to artificially infect the grasshopper. Role of eicosanoid in mediation of nodule formation has been shown by studying the effects of eicosanoid inhibitor dexamethasone and rescue experiments using the precursor, arachidonic acid. Significant reduction in the number of nodules by dexamethasone and their restoration by arachidonic acid has shown that eicosanoids function for nodulation in P. pictus. Most interesting result of the present work has been revealed by the fact that all the four natural products inhibit nodule formation in P. pictus, which would lead to impaired immunity in the insect. These results can be exploited in insect pest management by inducing immunodeficiency.





Similar content being viewed by others
References
Rembold H (1989) Azadirachtins: their structure and mode of action. In: Arnason J T, Philogene J, Morand P (eds) Insecticides of plants origin. ACS, Washington DC
Rembold H (1994) Secondary plant compounds in insect control with special reference to azadirachtins. In: Engels WE (ed) Advances in insect reproduction. Elsevier, Amsterdam, p 481
Banerji A (1988) Alternate strategies of insect control: natural products as potential biocides. Technical report Bhabha Atomic Research Centre (BARC) Bombay 1416:1-62
Banerji A (1994) Insect–plant interactions: natural insect control agents. In: Gujar GT (ed) Recent advances in insect physiology and toxicology. Agricole Publishing Academy, New Delhi, p 41
Banerjee S, Rembold H (1992) Azadirachtin A interferes with control of serotonin pools in the neuroendocrine system of locusts. Naturwissen 79:81–84
Bowers WS (1992) Insecticidal compound from plants. In: Nigg HN, Seliglier D (eds) Phytochemical resources for medicine and agriculture. Plenum Press, New York, p 227
Gupta AP (1994) Insect haemocytes: classification and immunological function. In: Gujar GT (ed) Recent advances in insect physiology and toxicology. Agricole Publishing Academy, New Delhi, p 218
Banerjee S, Oberthuer W, Rembold H (1995) In: Dutta NM, Gupta Bhattacharya SY, Mandal S, Bhattacharya KN (eds) Current topics in pollen-spore and biopollution research. Research Periodicals and Book Publishing House, Routledge
Magdum S, Banerjee S, Kalena GP, Banerji A (2001) Chemosterilant activity of naturally occurring quinones and their analogues in Dysdercus koenigii. J Appl Entomol 125:589–596
Banerjee S, Banerjj A (2004) Novel synthetic precocenoids as inhibitors of reproduction in Dysdercus koenigii. J Ecophysiol Occup Health 4:123–136
Subrahmanyam B, Mueller T, Rembold H (1989) Inhibition of turnover of neurosecretion by azadirachtin in Locusta migratoria. J Insect Physiol 35:493
Subrahmanyam B, Rembold H (1989) Effect of azadirachtin A on neuroendocrine activity in Locusta migratoria. Cell Tissue Research 256:513–517
Annadurai RS, Oberthuer W, Rembold H (1992) High resolution two-dimensional mapping of tissue-specific polypeptides in the desert locust, Schistocerca gregaria. Electrophoresis 13:456
Magdum S (1999) Investigations on bioactivity of plumbagin and some related compounds in Dysdercus koenigii (Hemiptera), Ph D thesis. Dr H S Gour University, Sagar
Hoffman JA (1970) Endocrine regulation of production and differentiation of heamocytes in insects, I orthoptera: Locusta migratoria migratoides. Gen Comp Endocrinol 15:198–219
Pathak JPN (1986) Haemogram and its endocrine control in insects. In: Brehelin M (ed) Immunity in invertebrates. Springer, Berlin, pp 49–59
Vinson SB (1993) Interactions between the insect endocrine system and the immune system. In: Pathak JPN (ed) Insect immunity. IBH Oxford Publishing, New Delhi, p 103
Gupta AP (1994) Insect haemocytes: classification and immunologic functions. In: Gujar GT (ed) Recent advances in insect physiology and toxicology. Agricole Publishing Academy, New Delhi, p 106
Rao PCC, Ray A, Rammurthy PS (1984) Effect of ligation and ecdysone on total haemocyte count of Spodopetra liture. Can J Zool 62:1461–1463
Ahmad A, Khan MA (1988) Effect of trial and makisterone A on the haemocytes of Hieroglyphus nigroreptetus Bolivar (Orthopter: Acrididoe). Proc Indian Acad Sci (Anim Sci) 97(3):203–210
Haq MR, Sabri MA, Rasheed A (2005) Toxicity of nictinile insecticides on the haemocytes of red cotton bug Dysdercus koenigii (Pyrrhocoridae: Hemiptera). J Agri Soc Sci 1(3):239–241
Haq MR, Sabri MA, Rasheed A (2006) Toxic effects of endosulfan 35 EC and Deltaphos 350 + 10EC on the haemocytes of red cotton bug Dysdercus koenigii (Pyrrhocoridae: Hemiptera). J Agric Soc Sci 2(1):29–32
Prakash B, Bhargava S, Rawat K (2007) Effect of penfluron on total haemocyte count of Dysdercus koenigii. Asian J Exp Sci 21(1):151–154
Sabri MA, Tariq B (2004) Toxicity of some insecticides on the heamocytes of red pumpkin beetle Aulacophora foveicollis larvae. Pak Entomol 26(1):109–114
Singh AP, Banerjee S (2012) Naturally occurring quinones modulate haemagglutination activity in Poekilocerus pictus (Acrididae: orthoptera). Proc Natl Acad Sci India B 82(4):543–551
Ottaviani E (2005) Insect immunorecognition. ISJ 2:142–151
Ratcliffe NA, Walters JB (1983) Studies on the in vivo cellular reactions of insects: fate of pathogenic and nonpathogenic bacteria in Galleria mellonella nodules. J Insect Physiol 29:407–415
Miller JS, Howard RW, Nguyen T, Nguyen A, Rosario RMT, Stanly-Samuelson DW (1996) Eicosanoids mediate nodulation responses to bacterial infection in larvae of the tenebrionid beetle, Zophobas atratus. J Insect Physiol 42:3–12
Jurenka RA, Miller JS, Pedibhotla VK, Raina RL, Stanley-Samuelson DW (1997) Eicosanoids mediate microaggregation and nodulation responses to bacterial infections in black cutworms, Agrotis ipsilon and true armyworms, Pseudaletia unipuncta. J Insect Physiol 43:125–133
Miller JS, Howard RW, Rana RL, Tunaz H, Stanley DW (1999) Eicosanoids mediate nodulation reactions to bacterial infections in adults of the cricket, Gryllus assimilis. J Insect Physiol 45:75–83
Tunaz H, Bedick JC, Miller JS, Hoback WW, Rana RL, Stanley DW (1999) Eicosanoids mediate nodulation reactions to bacterial infections in adults of two 17-year periodical cicadas, Magicicada septendecim and M. cassini. J Insect Physiol 45:923–931
Stanley DW (2000) Eicosanoids in invertebrate signal transduction system. Princeton University Press, Princeton, p 277
Stanley DW (2005) In: Gilbert LI, Latrou K, Gill SS (eds) Comprehensive molecular insect science. Elsevier, Pergemon Amsterdam, p 307
Stanley DW (2006) Prastaglandins and other eicosanoids in insects: biological significance. Ann Rev Entomol 51:25–44
Stanley DW, Miller JS (2006) Eicosanoids: in insect cellular immune function. Entomol Exp Appl 119:1–13
Turner RA (1965) Screening methods in pharmacology. Academic press, London, pp 60–68
Shapiro M (1979) Changes in hemocyte populations. In: Gupta AP (ed) Insect heamocytes. Cambridge University Press, Cambridge, p 539
Gupta AP (1979) Insect heamocytes. Cambridge University Press, Cambridge, p 85
Elhance DN (1977) Fundamentals of statistics. Kitab Mahal, Allahabad, pp 796–804
Strand MR, Pech LL (1995) Immunological basis for compartibility in parasitoid–host relationships. Ann Rev Entomol 40:31–56
Gillespie JP, Kanost MR, Treczer T (1997) Biological mediators of insect immunity. Annu Rev Entomol 42:611–643
Carton Y, Frey F, Nappi A (1992) Genetic determinism of the cellular immune reaction in Drosophila melanogastern. Heredity 69:393–399
Vass E, Nappi AJ (2000) Developmental and immunological aspects of Drosophila–parasitoid relationships. J Parasitol 86:1259–1270
Nappi AJ, Ottaviani E (2000) Cytotoxicity and cytotoxic molecules in invertebrates. Bio Essays 22:469–480
Nappi AJ, Christensen BM (2005) Melanogenesis and associated cytotoxic reactions: application to insect immunity. J Insect Biochem Mol biol 35:443–449
Stanley DW, Miller JS (2008) Parasitoid polydnaviruses and insect immunity. In: Beckage NE (ed) Insect immunology. Elsevier, San Diego, p 49
Krishnan N (2002) Studies on the role of some signal transduction moieties and cell–cell interactions associated with the cellular defense responses of Bombyx mori L. to bacterial infection. Ph.D. Thesis, Midnapore University
Baines D, Downer RGH (1994) Octopamine enhances phagocytosis in cockroach haemocytes: involvement of inositol triphosphate. Arch Insect Biochem Physiol 26:249–261
Diehl-Jones W, Mandato CA, Whent G, Downer RGH (1996) Monoaminergic regulation of haemocyte activity. J Insect Physiol 42:13–19
Howard RW, Miller JS, Stanley DW (1998) The influence of bacterial species and intensity of infections on nodule formation in insects. J Insect Physiol 44:157–164
Acknowledgments
The authors are thankful to Dr. A. Banerji, former Head, Bioorganic division BARC Mumbai, Prof. H. Rembold (Former Head, Insect Biochemistry, Max Planck Institute for Biochemistry, Munich), Terra Nostra, GmbH, Germany, Parry India (P) Ltd., Bangalore and Prof. J.P.N. Pathak, former Head, Department of Zoology, Madhav Science College, Ujjain, for the gift samples of plant products and bacterial cultures.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Singh, A.P., Jain, M.K. & Banerjee, S. Natural Products Modulate Eicosanoid Mediated Nodulation in Poekilocerus pictus Fab. (Acrididae, Orthoptera). Proc. Natl. Acad. Sci., India, Sect. B Biol. Sci. 84, 75–82 (2014). https://doi.org/10.1007/s40011-013-0187-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40011-013-0187-5