Abstract
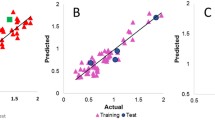
From two QSAR studies realized with the PRECLAV computer program, the database of the series of aromatic benzene sulfonamides incorporating 1,3,5-triazine moieties derivative, give without outliers N = 59, Se = 0.2858, r 2 = 0.8734, r 2CV = 0.8444, F = 74.5237. The virtual molecular fragments that lead to a significant increase of the inhibitor activity of CA IX are C3H2N5Cl, NH2, C3H5N6, CHO2, and C3HN6. The virtual fragment OH, C5H2NO, N atom, CHO2, C3HN6 leads to a significant decrease of the inhibitor activity value. The CA IX inhibitor activity is favorably influenced by the size of the molecule. With a view to external validation, the calibration set includes 46 molecules (Se = 0.234, r 2 = 0.9114, F = 84.3447, r 2CV = 0.8872) and the validation set includes 13 molecules (Se = 0.279, r 2 = 0.8741, F = 56.9143, r 2CV = 0.8246). Identification of molecules in validation set with high estimated value of inhibitory activity of CA IX is correct enough to have practical value, even if the calibration/validation set contains aromatic benzene sulfonamides incorporating 1,3,5-triazine moieties derivatives with very different chemical structures.

Similar content being viewed by others
References
Supuran CT (2008) Carbonic anhydrases: novel therapeutic applications for inhibitors and activators. Nat Rev Drug Discov 7:168–181
Mincione F, Menabuoni L, Supuran CT (2004) Clinical applications of the carbonic anhydrase inhibitors in ophthalmology. In: Supuran CT, Scozzafava A, Conway J (eds) Carbonic anhydrase, its inhibitors and activator. CRC Press, Boca Raton
Supuran CT (1994) Carbonic anhydrases: catalytic and inhibition mechanisms, distribution and physiological roles. In: Pacscas I (ed) Carbonic anhydrase and modulation of physiologic and pathologic process in the organism. Helicon. Timisoara
Supuran CT, Scozzafava A, Casini A (2004) Development of sulfonamide carbonic anhydrase inhibitors (CAIs). In: Supuran CT, Scozzafava A, Conway J (eds) Carbonicanhydrase, its inhibitors and activators. CRC Press, Boca Raton
Supuran CT, Scozzafava A, Casini A (2003) Carbonic anhydrase inhibitors. Med Res Rev 23:146–189
Korkolopoulou P, Perdiki M, Thymara I, Boviatsis E, Agrogiannis G, Kotsiakis X, Angelidakis D, Rologis D, Diamantopoulou K, Thomas Tsagli E, Kaklamanis L, Gatter K, Patsouris E (2007) Expression of hypoxia-related tissue factors in astrocytic gliomas. A multivariate survival study with emphasis upon carbonic anhydrase IX. Hum Pathol 38:629–638
Giatromanolaki A, Koukourakis MI, Sivridis E, Pastorek J, Wykoff CC, Gatter KC, Harris AL (2001) Expression of hypoxia-inducible carbonic anhydrase-9 relates to angiogenic pathways and independently to poor outcome in non-small cell lung cancer. Cancer Res 61:7992–7998
Chia SK, Wykoff CC, Watson PH, Han C, Leek RD, Pastorek J, Gatter KC, Ratcliffe P, Harris AL (2001) Prognostic significance of a novel hypoxia-regulated marker, carbonic anhydrase IX, in invasive breast carcinoma. J Clin Oncol 19:3660–3668
Singh S (2011) A comparative molecular field (CoMFA) studies on carbonic anhydrase inhibitor hCA IX-tumor-associated (hypoxia). Lett Drug Des Discov 8:877–883
Pastorekova S, Parkkila S, Pastorek J, Supuran CT (2004) Carbonic anhydrase isozymes. J Enzyme Inhib Med Chem 19:199–229
Svastova E, Hulikova A, Rafajova M, Zat ovicova M, Gibadulinova A, Casini A, Cecchi A, Scozzafava A, Supuran CT, Pastorek J, Pastorekova S (2004) Hypoxia activates the capacity of tumor-associated carbonic anhydrase IX to acidify extracellular pH. FEBS Lett 577:439–445
Wykoff CC, Beasley NJ, Watson PH, Turner KJ, Pastorek J, Sibtain A, Wilson GD, Turley H, Talks KL, Maxwell PH, Pugh CW, Ratcliffe PJ, Harris AL (2000) Hypoxia-inducible expression of tumor-associated carbonic anhydrases. Cancer Res 60:7075–7083
Potter CPS, Harris AL (2003) Diagnostic, prognostic and therapeutic implications of carbonic anhydrases in cancer. Br J Cancer 89:2–7
Robertson N, Potter C, Harris AL (2004) Role of carbonic anhydrase IX in human tumor cell growth. Survival and invasion. Cancer Res 64:6160–6165
Thiry A, Dogne JM, Masereel B, Supuran CT (2006) Targeting tumor-associated carbonic anhydrase IX in cancer therapy. Trends Pharmacol Sci 27:566–573
Winum JY, Rami M, Scozzafava A, Montero JL, Supuran CT (2008) A new druggable target for the design of antitumor agents. Med Res Rev 28:445
Lakshman S, Kumar A (2009) QSAR study on some series of sodium and potassium channel blockers. Med Chem 5:570–576
Garaj V, Puccetti L, Fasolis G, Winum JY, Montero JL, Scozzafava A, Vullo D, Innocentia A, Supurana CT (2005) Carbonic anhydrase inhibitors: novel sulfonamides incorporating 1,3,5-triazine moieties as inhibitors of the cytosolic and tumour-associated carbonic anhydrase isozymes I, II and IX. Bioorg Med Chem Lett 15:3102–3108
Winum Y, Jean I, Alessio V, Daniela M, Louis J, Supuran CT (2009) Carbonic anhydrase inhibitors: fluorinated phenyl sulfamates show strong inhibitory activity and selectivity for the inhibition of the tumor-associated isozymes IX and XII over the cytosolic ones I and II. Bioorg Med Chem Lett 19:5082–5085
Rzewski FS, Innocenti A, Sławin Sk, Jarosław KA, Brzozowsk Z, Pomarnacka E, Scozzafava A, Supuran CT (2008) Carbonic anhydrase inhibitors: inhibition of human cytosolic isozymes I and II and tumor-associated isozymes IX and XII with S-substituted 4-chloro-2-mercapto-5-methyl-benzene sulfonamide. Bioorg Med Chem 16:3933–3940
HyperChem Software7.52 (2006) Hypercube Inc. Florida Science and Technology Park
Allinger NL (1977) Conformational analysis 130 MM2 A hydrocarbon force field utilizing V1 and V2 torsional terms. J Am Chem Soc 99:8127
Allinger NL, Yuh YH (1982) Quantam chemistry program exchange, Bloomington, Indiana. In: Burkert U, Allinger NL (eds) Program # 395, Molecular mechanics, Monograph 177. ACS, Washington, D.C.
Stewart JJP (2007) The Mopac programVersion 8.301W (October 2008). http://www.openmopac.net/
Tarko L (2005) Calcule QSPR/QSAR cu ajutorul programului PRECLAV. Rev Chim (Bucuresti) 56:639
Tarko L, Lupescu I, Groposila CD (2005) Sweetness power QSARs by PRECLAV software. Arkivoc x:254
The PRECLAV v.0809 (2008) program. Center of Organic Chemistry (CCO), Romanian Academy, Bucharest
Talete SRL (2007) DRAGON Plus for windows, software for molecular descriptor calculations version 5.5-http://www.tate.mi.it/
Tarko L (2007) QSAR studies regarding the inhibition of the carbonic anhydrase by the sulfonamides containing a picolinoyl group. Rev Chim (Bucuresti) 58:191–194
Tarko L, Supuran CT (2007) QSAR studies for the inhibition of the transmembrane isozymes XII and XIV of human carbonic anhydrase with a series of sulfonamides. Bioorg Med Chem 15:5666–5671
Singh S, Khadikar PV, Scozzafava A, Supuran CT (2009) QSAR studies for the inhibition of the trans membrane carbonic anhydrase isozyme XIV with sulfonamides using PRECLAV software. J Enzyme Inhib Med Chem 24:337–349
Singh S (2009) Comparative QSAR studies on the novel series of thiazolones and tetrazole derivatives as HCV NS5B polymerase allosteric inhibitors. Lett Drug Des Discov 6:286–297
Singh S, Singh S, Shukla P (2010) Modeling of novel HIV-1 protease inhibitors incorporating N-aryl-oxazolidinone-5-carboxamides as P2 ligands using quantum chemical and topological finger print descriptors. Med Chem Res. doi: 10.1007/s00044-010-9416-0
Singh S, Supuran CT (2011). QSARs on human carbonic anhydrase VA and VB inhibitors of some new not yet synthesized, substituted aromatic/heterocyclic sulphonamides as anti-obesity agent. J Enzyme Inhib Med Chem. doi:10.3109/14756366.2011.606544
Tarko L (2004) A procedure for virtual fragmentation of molecules into functional groups. ARKIVOC xiv:74–82
Acknowledgment
The author (Shalini Singh) expresses her thanks to the University Grants Commission, New Delhi, India for providing financial support under UGC Research Award No.F.30-29/2011(SA-II)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Singh, S. In Silico Modeling of Carbonic Anhydrase Inhibitor hCA IX-Tumor Associated (Hypoxia). Proc. Natl. Acad. Sci., India, Sect. A Phys. Sci. 82, 113–123 (2012). https://doi.org/10.1007/s40010-012-0018-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40010-012-0018-8