Abstract
Purpose
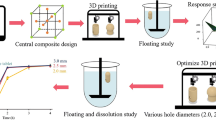
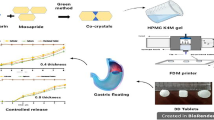
Helicobacter pylori is a common cause of peptic ulcer disease, but patient’s low compliance to treatment can cause antibiotic resistance. The aim of the study was to describe a gastroretentive drug delivery system using an anti–flip-up floating tablet design prepared by using 3D printing with the goal of achieving better therapeutic outcomes.
Methods
Polylactic acid (PLA) was the printing material for the floating tablet housing, with metronidazole as the model drug. The core tablet was prepared by direct compression and placed in the middle of the housing. The floating system was optimized by using a Box–Behnken experimental design with core tablet main factors of drug release orifice, air volume (height was varied), and compression force. The optimized formulation was prepared to test the prediction efficiency of the model.
Results
The optimized model required a drug release orifice of 23.10 mm2, air compartment height of 2.0 mm, and compression force of 3.63 tons, providing constant drug release for 8 h (average R2 = 0.9920). The optimized anti–flip-up tablets did not flip-up during the test.
Conclusion
This gastroretentive drug release model shows possibility for constant extended drug dissolution which could enhance therapeutic efficiency of peptic ulcer disease from Helicobacter pylori.






Similar content being viewed by others
References
Abdelbary A, El-Gazayerly ON, El-Gendy NA, Ali AA (2010) Floating tablet of trimetazidine dihydrochloride: an approach for extended release with zero-order kinetics. Aaps Pharmscitech 11(3):1058–1067
Arora S, Ali J, Ahuja A, Khar RK, Baboota S (2005) Floating drug delivery systems: a review. Aaps PharmSciTech 6(3):E372–E390
Arunachalam A, Karthikeyan M, Konam K, Prasad PH, Sethuraman S, Ashutoshkumar S, Manidipa S (2011) Floating drug delivery systems: A review. Int J Res Pharm Sci 2(1):76–83
Chey WD, Leontiadis GI, Howden CW, Moss SF (2017) ACG clinical guideline: treatment of Helicobacter pylori infection. Am J Gastroenterol 112(2):212–239
Dokoumetzidis A, Macheras P (2006) A century of dissolution research: from Noyes and Whitney to the biopharmaceutics classification system. Int J Pharm 321(1–2):1–11
Hadi MA, Rao NR, Rao AS (2013) Formulation and evaluation of compression coated tablets of lornoxicam for targeting early morning peak symptoms of Rheumatoid arthritis. Dhaka Univ J Pharm Sci 12(2):109–117
Hattori Y, Haruna Y, Otsuka M (2013) Dissolution process analysis using model-free Noyes-Whitney integral equation. Colloids Surf, B 102:227–231
He Y, Xue G-H, Fu J-Z (2014) Fabrication of low cost soft tissue prostheses with the desktop 3D printer. Sci Rep 4(1):1–7
Huanbutta K, Sangnim T (2019) Design and development of zero-order drug release gastroretentive floating tablets fabricated by 3D printing technology. J Drug Delivery Sci Technol 52:831–837
Jamróz W, Szafraniec J, Kurek M, Jachowicz R (2018) 3D printing in pharmaceutical and medical applications – recent achievements and challenges. Pharm Res 35(9):176
Khaled SA, Burley JC, Alexander MR, Yang J, Roberts CJ (2015) 3D printing of five-in-one dose combination polypill with defined immediate and sustained release profiles. J Control Release 217:308–314
Kimura S-I, Ishikawa T, Iwao Y, Itai S, Kondo H (2019) Fabrication of zero-order sustained-release floating tablets via fused depositing modeling 3d printer. Chem Pharm Bull 67(9):992–999
Kitazawa S, Johno I, Ito Y, Teramura S, Okada J (1975) Effects of hardness on the disintegration time and the dissolution rate of uncoated caffeine tablets. J Pharm Pharmacol 27(10):765–770
Kotta S, Nair A, Alsabeelah N (2018) 3D printing technology in drug delivery: recent progress and application. Curr Pharm Des 24(42):5039–5048
Lee K-J, Kang A, Delfino JJ, West TG, Chetty D, Monkhouse DC, Yoo J (2003) Evaluation of critical formulation factors in the development of a rapidly dispersing captopril oral dosage form. Drug Dev Ind Pharm 29(9):967–979
Li Q, Guan X, Cui M, Zhu Z, Chen K, Wen H, Jia D, Hou J, Xu W, Yang X (2018) Preparation and investigation of novel gastro-floating tablets with 3D extrusion-based printing. Int J Pharm 535(1–2):325–332
Malaty HM (2007) Epidemiology of Helicobacter pylori infection. Best Pract Res Clin Gastroenterol 21(2):205–214
Maroni A, Melocchi A, Parietti F, Foppoli A, Zema L, Gazzaniga A (2017) 3D printed multi-compartment capsular devices for two-pulse oral drug delivery. J Control Release 268:10–18
Nomura A, Stemmermann GN, Chyou P-H, Perez-Perez GI, Blaser MJ (1994) Helicobacter pylori infection and the risk for duodenal and gastric ulceration. Ann Intern Med 120(12):977–981
Pickover C (2008) Archimedes to hawking: laws of science and the great minds behind them. Oxford University Press, New Yorks.
Rees A, Powell LC, Chinga-Carrasco G, Gethin DT, Syverud K, Hill KE, Thomas DW (2015) 3D bioprinting of carboxymethylated-periodate oxidized nanocellulose constructs for wound dressing applications. BioMed Res Int. https://doi.org/10.1155/2015/925757
Stern PW (1976) Effects of film coatings on tablet hardness. J Pharm Sci 65(9):1291–1295
Timmermans J, Moës AJ (1990) How well do floating dosage forms float? Int J Pharm 62(2–3):207–216
Wu G, Wu W, Zheng Q, Li J, Zhou J, Hu Z (2014) Experimental study of PLLA/INH slow release implant fabricated by three dimensional printing technique and drug release characteristics in vitro. Biomed Eng Online 13(1):97
Zhao S, Lv Y, Zhang J-B, Wang B, Lv G-J, Ma X-J (2014) Gastroretentive drug delivery systems for the treatment of Helicobacter pylori. World J Gastroenterol: WJG 20(28):9321
Acknowledgements
The authors acknowledge the Faculty of Pharmaceutical Sciences, Burapha University for their financial support. The assistance of Mr. Kasitpong Thanawuth from the Faculty of Pharmacy, Silpakorn University with laboratory work is greatly appreciated.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Research involving human and animal rights
This article does not contain any studies with human and animal subjects performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Huanbutta, K., Sriamornsak, P., Kittanaphon, T. et al. Development of a zero-order kinetics drug release floating tablet with anti–flip-up design fabricated by 3D-printing technique. J. Pharm. Investig. 51, 213–222 (2021). https://doi.org/10.1007/s40005-020-00507-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40005-020-00507-7