Abstract
Purpose
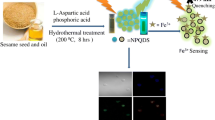
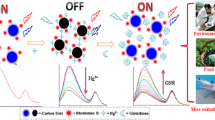
This article details the application of pristine nitrogen and sulfur doped carbon quantum dots (CQDs) as a novel fluorescence biosensor for the detection of glutathione. The second object of this study is to evaluate reduction of cellular nitric oxide in microglial cells.
Methods
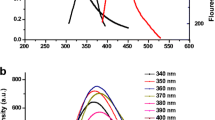
Microwave assisted hydrothermal method was used for the fabrication of CQDs. Unlike conventional methods which utilize metallic or transition metal coating over CQDs for the fabrication of fluorescence switch on/off probes, our simple yet efficient CQDs itself performed as a biosensor that is both selective and sensitive towards glutathione (GSH). Particle size analyzer, scanning electron microscope, atomic force microscopy, high-performance X-ray photoelectron spectroscopy, fourier-transform infrared spectroscopy were used for physicochemical characterization of developed CQDs. Photoluminescence properties of CQDs were analyzed using photoluminescence spectroscope for glutathione detection. Furthermore, microglial cells were used to evaluate reduction of cellular nitric oxide.
Results
The developed biosensor was able to detect GSH within a short time of 2 min. Hemolysis assay confirmed negligible red blood cell lysis even at a higher concentration of 0.2 mg/mL. Furthermore, the developed CQDs demonstrated enhanced cellular uptake, which resulted in generating fluorescence from the BV-2 microglial cells. Interestingly, the developed CQDs were able to mitigate the secretion of toxic pro-inflammatory cytokine, nitric oxide (NO) from the lipopolysaccharide (LPS) insulted BV-2 microglial cells. A 50% reduction in the secretion of NO was observed after treating with CQDs in the LPS treated BV-2 cells.
Conclusion
These novel fluorescent CQDs with low manufacturing costs, high selectivity and sensitivity towards GSH and shorter detection time manifest them as a promising nanomaterial for diverse biomedical applications.






Similar content being viewed by others
References
Baker SN, Baker GA (2010) Luminescent carbon nanodots: emergent nanolights. Angew Chem Int Ed 49:6726–6744. https://doi.org/10.1002/anie.200906623
Bansal A, Simon MC (2018) Glutathione metabolism in cancer progression and treatment resistance. J Cell Biol 217:2291–2298. https://doi.org/10.1083/jcb.201804161
Bhattacharyay D, Dutta K, Banerjee S et al (2008) Disposable amperometric sensors for thiols with special reference to glutathione. Electroanalysis 20:1947–1952. https://doi.org/10.1002/elan.200804247
Bourlinos AB, Stassinopoulos A, Anglos D et al (2008) Surface functionalized carbogenic quantum dots. Small 4:455–458. https://doi.org/10.1002/smll.200700578
Cai Q-Y, Li J, Ge J et al (2015) A rapid fluorescence “switch-on” assay for glutathione detection by using carbon dots–MnO2 nanocomposites. Biosens Bioelectron 72:31–36. https://doi.org/10.1016/j.bios.2015.04.077
Cao M, Chen H, Chen D et al (2016) Naphthalimide-based fluorescent probe for selectively and specifically detecting glutathione in the lysosomes of living cells. Chem Commun 52:721–724. https://doi.org/10.1039/C5CC08328A
Chae A, Choi Y, Jo S et al (2017) Microwave-assisted synthesis of fluorescent carbon quantum dots from an A 2 /B 3 monomer set. RSC Adv 7:12663–12669. https://doi.org/10.1039/C6RA28176A
Chang PV, Bertozzi CR (2012) Imaging beyond the proteome. Chem Commun 48:8864. https://doi.org/10.1039/c2cc31845h
Deng R, Xie X, Vendrell M et al (2011) Intracellular glutathione detection using MnO2-nanosheet- modified upconversion nanoparticles. J Am Chem Soc 133:20168–20171
Dutta Choudhury S, Chethodil JM, Gharat PM et al (2017) pH-elicited luminescence functionalities of carbon dots: mechanistic insights. J Phys Chem Lett 8:1389–1395. https://doi.org/10.1021/acs.jpclett.7b00153
Flohé L, Günzler WA (1984) [12] Assays of glutathione peroxidase. Methods Enzymol 105:114–120
Huang GG, Han XX, Hossain MK, Ozaki Y (2009) Development of a heat-induced surface-enhanced raman scattering sensing method for rapid detection of glutathione in aqueous solutions. Anal Chem 81:5881–5888. https://doi.org/10.1021/ac900392s
Janeš L, Lisjak K, Vanzo A (2010) Determination of glutathione content in grape juice and wine by high-performance liquid chromatography with fluorescence detection. Anal Chim Acta 674:239–242. https://doi.org/10.1016/j.aca.2010.06.040
Kacimi R, Giffard RG, Yenari MA (2011) Endotoxin-activated microglia injure brain derived endothelial cells via NF-κB, JAK-STAT and JNK stress kinase pathways. J Inflamm (Lond) 8:7. https://doi.org/10.1186/1476-9255-8-7
Kowalska K, Zalewska M, Milnerowicz H (2015) The application of capillary electrophoresis in the determination of glutathione in healthy women’s blood. J Chromatogr Sci 53:353–359. https://doi.org/10.1093/chromsci/bmu035
Kundu A, Nandi S, Layek RK, Nandi AK (2013) Fluorescence resonance energy transfer from sulfonated graphene to riboflavin: a simple way to detect vitamin B 2. ACS Appl Mater Interfaces 5:7392–7399. https://doi.org/10.1021/am4017208
Li ZQ, Lu CJ, Xia ZP et al (2007) X-ray diffraction patterns of graphite and turbostratic carbon. Carbon N Y 45:1686–1695. https://doi.org/10.1016/j.carbon.2007.03.038
Li G, Lv N, Zhang J, Ni J (2017) MnO2 in situ formed into the pores of C-dots/ZIF-8 hybrid nanocomposites as an effective quencher for fluorescence sensing ascorbic acid. RSC Adv 7:16423–16427. https://doi.org/10.1039/C7RA00307B
Li L, Jia C, Wang F et al (2018) Facile synthesis of magnetic fluorescent nanoparticles: adsorption and selective detection of Hg( %3cscp%3eii%3c/scp%3e ) in water. J Mater Chem C 6:2360–2369. https://doi.org/10.1039/C7TC05564A
Liu X, Zhang N, Bing T, Shangguan D (2014) Carbon dots based dual-emission silica nanoparticles as a ratiometric nanosensor for Cu 2+. Anal Chem 86:2289–2296. https://doi.org/10.1021/ac404236y
Liu Q, Zhang N, Shi H et al (2018) One-step microwave synthesis of carbon dots for highly sensitive and selective detection of copper ions in aqueous solution. New J Chem 42:3097–3101. https://doi.org/10.1039/C7NJ05000C
Lu W, Qin X, Liu S et al (2012) Economical, green synthesis of fluorescent carbon nanoparticles and their use as probes for sensitive and selective detection of mercury(II) ions. Anal Chem 84:5351–5357. https://doi.org/10.1021/ac3007939
Lushchak VI (2012) Glutathione homeostasis and functions: potential targets for medical interventions. J Amino Acids 2012:1–26. https://doi.org/10.1155/2012/736837
Mleczko J, Defort A, Kozioł JJ et al (2016) Limitation of tuning the antibody-antigen reaction by changing the value of pH and its consequence for hyperthermia. J Biochem 159:421–427. https://doi.org/10.1093/jb/mvv120
Mudunkotuwa IA, Al MA, Grassian VH (2014) ATR-FTIR spectroscopy as a tool to probe surface adsorption on nanoparticles at the liquid–solid interface in environmentally and biologically relevant media. Analyst 139:870–881. https://doi.org/10.1039/C3AN01684F
Nandi S, Routh P, Layek RK, Nandi AK (2012) Ethidium bromide-adsorbed graphene templates as a platform for preferential sensing of DNA. Biomacromol 13:3181–3188. https://doi.org/10.1021/bm3009632
Niu L-Y, Guan Y-S, Chen Y-Z et al (2012) BODIPY-based ratiometric fluorescent sensor for highly selective detection of glutathione over cysteine and homocysteine. J Am Chem Soc 134:18928–18931. https://doi.org/10.1021/ja309079f
Nurunnabi M, Parvez K, Nafiujjaman M et al (2015) Bioapplication of graphene oxide derivatives: drug/gene delivery, imaging, polymeric modification, toxicology, therapeutics and challenges. RSC Adv 5(52):42141–42161. https://doi.org/10.1039/c5ra04756k
Pan J, Zheng Z, Yang J et al (2017) A novel and sensitive fluorescence sensor for glutathione detection by controlling the surface passivation degree of carbon quantum dots. Talanta 166:1–7. https://doi.org/10.1016/j.talanta.2017.01.033
Pravda J (2014) Metabolic theory of septic shock. World J Crit care Med 3:45–54. https://doi.org/10.5492/wjccm.v3.i2.45
Revuri V, Cherukula K, Nafiujjaman M et al (2018) White-light-emitting carbon nano-onions: a tunable multichannel fluorescent nanoprobe for glutathione-responsive bioimaging. ACS Appl Nano Mater 1:662–674. https://doi.org/10.1021/acsanm.7b00143
Sobhakumari A, Love-Homan L, Fletcher EVM et al (2012) Susceptibility of human head and neck cancer cells to combined inhibition of glutathione and thioredoxin metabolism. PLoS ONE 7:e48175. https://doi.org/10.1371/journal.pone.0048175
Tajarrod N, Rofouei MK, Masteri-Farahani M, Zadmard R (2016) A quantum dot-based fluorescence sensor for sensitive and enzymeless detection of creatinine. Anal Methods 8:5911–5920. https://doi.org/10.1039/C6AY01609J
Tang L, Ji R, Cao X et al (2012) Deep ultraviolet photoluminescence graphene quantum dots. ACS Nano 6:5102–5110. https://doi.org/10.1021/nn300760g
Wang C, Chen L, Wang P et al (2019) A novel ultrasensitive electrochemiluminescence biosensor for glutathione detection based on poly-L-lysine as co-reactant and graphene-based poly(luminol/aniline) as nanoprobes. Biosens Bioelectron 133:154–159. https://doi.org/10.1016/j.bios.2019.03.016
Wendel A (1981) [44] Glutathione peroxidase. Academic Press, Cambridge, pp 325–333
Xu Y, Niu X, Zhang H et al (2015) Switch-on fluorescence sensing of glutathione in food samples based on a graphitic carbon nitride quantum dot (g-CNQD)–Hg2+ chemosensor. J Agric Food Chem 63:1747–1755. https://doi.org/10.1021/jf505759z
Yang S, Sun J, Li X et al (2014) Large-scale fabrication of heavy doped carbon quantum dots with tunable-photoluminescence and sensitive fluorescence detection. J Mater Chem A 2:8660. https://doi.org/10.1039/c4ta00860j
Yang C, Deng W, Liu H et al (2015) Turn-on fluorescence sensor for glutathione in aqueous solutions using carbon dots–MnO2 nanocomposites. Sens Actuators B Chem 216:286–292. https://doi.org/10.1016/j.snb.2015.04.055
Ye Z, Tang R, Wu H et al (2014) Preparation of europium complex-conjugated carbon dots for ratiometric fluorescence detection of copper( %3cscp%3eii%3c/scp%3e ) ions. New J Chem 38:5721–5726. https://doi.org/10.1039/C4NJ00966E
Zhang C, Wei L, Wei C et al (2015) A FRET–ICT dual-quenching fluorescent probe with large off–on response for H2S: synthesis, spectra and bioimaging. Chem Commun 51:7505–7508. https://doi.org/10.1039/C5CC01184A
Zheng XT, Ananthanarayanan A, Luo KQ, Chen P (2015) Glowing graphene quantum dots and carbon dots: properties, syntheses, and biological applications. Small 11:1620–1636. https://doi.org/10.1002/smll.201402648
Zhou H, Tang J, Lv L et al (2018) Intracellular endogenous glutathione detection and imaging by a simple and sensitive spectroscopic off–on probe. Analyst 143:2390–2396. https://doi.org/10.1039/C8AN00102B
Zhu S, Song Y, Wang J et al (2017) Photoluminescence mechanism in graphene quantum dots: quantum confinement effect and surface/edge state. Nano Today 13:10–14. https://doi.org/10.1016/j.nantod.2016.12.006
Acknowledgements
This research was graciously supported by National Research Foundation of Korea (Grant Numbers: NRF-2017R1C1B2010276, NRF-2018R1D1A1A09083269). This work was also supported by Korea National University of Transportation in 2018.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing financial interest.
Research involving human and animal rights
This article does not contain any studies with human and animal subjects performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mondal, J., Revuri, V., Choochana, P. et al. Sulfur and nitrogen doped carbon quantum dots for detection of glutathione and reduction of cellular nitric oxide in microglial cells. J. Pharm. Investig. 50, 209–218 (2020). https://doi.org/10.1007/s40005-019-00466-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40005-019-00466-8