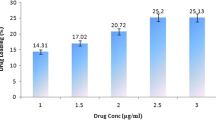
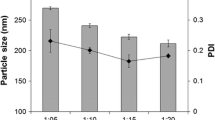
Abstract
Artemisinin (ART)-loaded niosome and pegylated niosomes were prepared using two different techniques. Nanosized lipid vesicles were physically characterized for entrapment efficacy and stability. Particle sizes were determined and release kinetic of the optimized formulation was carried out by dialysis method. The efficacy of the developed formulation was tested on MCF7 cells and cytotoxicity was accomplished by MTT assay. Common observation was the effect of pegylation on the reduction of vesicle size due to its hydrophilic nature. Span 60 niosomes had slightly larger vesicle size than Span 20 niosomes. Over all the good stability was observed over 60 days. In vitro drug release studies indicate gradual release of niosome over 40 h. similar trend in drug release was observed for most formulation except for the multilammellar pegylated niosomes. Pegylation of niosomes causes increased stability and efficacy of ART. Cytotoxicity (IC50) was evaluated at different time of incubation at 48 and 72 h for selected niosomal formulations. Pegylated ART niosomes show great advantages in term of interaction with MCF-7 cell membrane. Results suggest that pegylated niosomes may be an appropriate candidate for the clinical administration of ART.



Similar content being viewed by others
References
Chen Y, Lin X, Park H, Greever R (2009) Study of artemisinin nanocapsules as anticancer drug delivery system. Nanomedicine 5:316–322
Efferth T, Oesch F (2004) Oxidative stress response of tumor cells: microarray-based comparison between artemisinins and anthracyclines. Biochem Pharmacol 68:3–10
Fang J-Y, Hong C-T, Chiu W-T, Wang Y-Y (2001) Effect of liposomes and niosomes on skin permeation of Enoxacin. Int J Pharm 219:61–72
Fathi Azarbayjani A, Lin H, Yap CW, Chan YW, Chan SY (2010) Surface tension and wettability in transdermal delivery: a study on the in-vitro permeation of haloperidol with cyclodextrin across human epidermis. J Pharm Pharmacol 62:770–778
Fathi Azarbayjani A, Khu JV, Chan YW, Chan SY (2011) Development and characterization of skin permeation retardants and enhancers: a comparative study of Levothyroxine-loaded PNIPAM, PLA, PLGA and EC microparticles. Biopharm Drug Dispos 32:380–388
Fathi-Azarbayjani A, Tan PL, Chan YY, Chan SY (2013) Ascorbic acid for the safe use of a sunscreen agent: accumulation of nano zinc oxide and titanium dioxide on the skin. Sci Pharm 81(4):1141–1150
Fathi-Azarbayjani A, Ng KX, Chan YW, Chan SY (2015) Lipid vesicles for the skin delivery of diclofenac: cerosomes vs. other lipid suspensions. Adv Pharm Bull 5(1): 25–33
Ibrahim N, Ibrahim H, Sabater AM, Mazier D, Valentin A, Nepveu F (2015) Artemisinin nanoformulation suitable for intravenous injection: preparation, characterization and antimalarial activities. Int J Pharm 495:671–679
Iwunze MO (2004) Binding and distribution characteristics of curcumin solubilized in CTAB micelle. J Mol Liq 111:161–165
Jing Y, Danielsson A, Dobšíček Trefná H, Persson M, Svedhem S (2016) Asymmetric cationic liposomes designed for heat-activated association with cells. Colloids Surf B. doi:10.1016/j.colsurfb.2016.06.041
Khan MI, Madni A, Peltonen L (2016) Development and in-vitro characterization of sorbitan monolaurate and poloxamer 184 based niosomes for oral delivery of diacerein. Eur J Pharm Sci 95:88–95
Lai H, Sasaki T, Singh NP (2005) Targeted treatment of cancer with artemisinin and artemisinin-tagged iron-carrying compounds. Expert Opin Ther Targets 9:995–1007
Navaratnam V, Mansor SM, Sit NW, Grace J, Li Q, Olliaro P (2000) Pharmacokinetics of artemisinin-type compounds. Clin Pharmacokinet 39(4):255–270
Palozza P, Muzzalupo R, Trombino S, Valdannini A, Picci N (2006) Solubilization and stabilization of β-carotene in niosomes: delivery to cultured cells. Chem Phys Lipids 139:32–42
Pasut G, Paolino D, Celia C, Mero A, Joseph AS, Wolfram J, Cosco D, Schiavon O, Shen H, Fresta M (2015) Polyethylene glycol (PEG)-dendron phospholipids as innovative constructs for the preparation of super stealth liposomes for anticancer therapy. J Control Release 199:106–113
Payghan SA, Bhat M (2008) Albumin microspheres containing artemisinin. Pharmacist 3:33–36
Shahzad Y, Sohail S, Arshad MS, Hussain T, Shah SNH (2013) Development of solid dispersions of artemisinin for transdermal delivery. Int J Pharm 457:197–205
Shaker DS, Shaker MA, Hanafy MS (2015) Cellular uptake, cytotoxicity and in-vivo evaluation of Tamoxifen citrate loaded niosomes. Int J Pharm 493:285–294
Vyas SP, Singh RP, Jain S, Mishra V, Mahor S, Singh P, Gupta PN, Rawat A, Dubey P (2005) Non-ionic surfactant based vesicles (niosomes) for non-invasive topical genetic immunization against hepatitis B. Int J Pharm 296:80–86
Wang Z, Yu Y, Ma J, Zhang H, Wang X, Wang J, Zhang X, Zhang Q (2012) LyP-1 modification to enhance delivery of artemisinin or fluorescent probe loaded polymeric micelles to highly metastatic tumor and its lymphatics. Mol Pharm 9(9):2646–2657
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
This statement is to certify that the authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Asgharkhani, E., Fathi Azarbayjani, A., Irani, S. et al. Artemisinin-loaded niosome and pegylated niosome: physico-chemical characterization and effects on MCF-7 cell proliferation. J. Pharm. Investig. 48, 251–256 (2018). https://doi.org/10.1007/s40005-017-0331-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40005-017-0331-y