Abstract
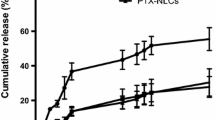
The aim of this study was to compare physicochemical properties of solid lipid nanoparticles (SLNs) on drug nature. These SLNs were fabricated for formulating paclitaxel (PTX) or docetaxel (DTX). To make small, and highly encapsulated SLNs, the SLNs consisted of compritol 888 ATO (glyceryl behenate) as lipid matrix, poloxamer 188 or solutol HS 15 as surfactant and soya lecithin as co-surfactant. The SLNs were characterized by particle size, zeta potential, in vitro release study and cytotoxicity study. In this study, SLNs showed different physicochemical properties and release profiles according to used solid lipid. In case of particle size, PS1 and DS1 showed bigger particle size than those of PS2 and DS2. Encapsulation efficiency (%) of DTX-loaded SLNs exhibited higher than that of PTX-loaded SLNs. And, DTX-loaded SLNs showed prolonged release up to 24 h. We found that SLN using solutol HS 15 as surfactant exhibited enhanced cellular uptake on MCF-7/ADR cells. These results suggest that different drugs and surfactant effect on physicochemical properties regardless same composition.






Similar content being viewed by others
References
Baek JS, Shin SC, Cho CW (2012a) Effect of lipid on physicochemical properties of solid lipid nanoparticle of paclitaxel. J Pharm Investig 42(5):279–283
Baek JS, So JW, Shin SC, Cho CW (2012b) Solid lipid nanoparticles of paclitaxel strengthened by hydroxypropyl-β-cyclodextrin as an oral delivery system. Int J Mol Med 30:953–959
Bittner B, Gonzalez RC, Walter I, Kapps M, Huwyler J (2003) Impact of Solutol HS 15 on the pharmacokinetic behavior of colchicine upon intravenous administration to male Wistar rats. Biopharm Drug Dispos 24(4):173–181
Coon JS, Knudson W, Clodfelter K, Lu B, Weinstein RS (1991) Solutol HS 15, nontoxic polyoxyethylene esters of 12-hydroxystearic acid, reverses multidrug resistance. Cancer Res 51(3):897–902
Cortes JE, Pazdur R (1995) Docetaxel. J Clin Oncol 13:2643–2655
Garcia-Fuentes D, Loukili N, Garcia-Fuentes M, Thomson TM (2002) Influence of dipyridamole on the fatty acid composition of the main lipid classes of chick serum. Environ Toxicol Pharmacol 12(4):189–194
He C, Hu Y, Yin L, Tang C, Yin C (2010) Effects of particle size and surface charge on cellular uptake and biodistribution of polymeric nanoparticles. Biomaterials 31(3):3657–3666
Hortobagyi GN (1999) High-dose chemotherapy for primary breast cancer: facts versus anecdotes. J Clin Oncol 17(11):25–29
Hu FQ, Jiang SP, Du YZ, Yuan H, Ye YQ, Zeng S (2005) Preparation and characterization of stearic acid nanostructured lipid carriers by solvent diffusion method in an aqueous system. Colloids Surf B Biointerfaces 45:167–173
Joshi M, Patravale V (2006) Formulation and evaluation of nanostructured lipid carrier (NLC)-based gel of Valdecoxib. Drug Dev Ind Pharm 32(8):911–918
Kumar VV, Chandrasekar D, Ramakrishna S, Kishan V, Rao YM, Diwan PV (2007) Development and evaluation of nitrendipine loaded solid lipid nanoparticles: influence of wax and glyceride lipids on plasma pharmacokinetics. Int J Pharm 335(1–2):167–175
Lee MK, Lim SJ, Kim CK (2007) Preparation, characterization and in vitro cytotoxicity of paclitaxel loaded sterically stabilized solid lipid nanoparticles. Biomaterials 28:2137–2146
Marengo E, Cavalli R, Caputo O, Rodriguez L, Gasco MR (2000) Scale-up of the preparation process of solid lipid nanospheres. Part I. Int J Pharm 205(1–2):3–13
Mehnert W, Mader K (2001) Solid lipid nanoparticles: production, characterization and applications. Adv Drug Deliv Rev 47:165–196
Mo Y, Lim LY (2005) Preparation and in vitro anticancer activity of wheat germ agglutinin (WGA)-conjugated PLGA nanoparticles loaded with paclitaxel and isopropyl myristate. J Control Release 107:30–42
Muller RH, Mader K, Gohla S (2000) Solid lipid nanoparticles (SLN) for controlled drug delivery—a review of the state of the art. Eur J Pharm Biopharm 50(1):161–177
Reinhart T, Bauer KH (1995) The hemolysis and solubilization behavior of nonionic polymer surface-active agents classes. Pharmazie 50(6):403–407
Rose PG, Maxson JH, Fusco N, Mossbruger K, Rodriquez M (2001) Liposomal doxorubicin in ovarian, peritoneal, and tubal carcinoma: a retrospective comparative study of single-agent dosages. Gynecol Oncol 82(2):323–328
Ruchatz F, Schuch H (1998) Physicochemical properties of Solutol HS 15 and its solubisates. BASF ExAct 1:6–7
Subedi RK, Kang KW, Choi HK (2009) Preparation and characterization of solid lipid nanoparticles loaded with doxorubicin. Eur J Pharm Sci 37(3–4):508–513
Trickler WJ, Nagvekar AA, Dash AK (2008) A novel nanoparticle formulation for sustained paclitaxel delivery. AAPS PharmSciTech 9:486–493
Trotta M, Gallarate M, Carlotti ME, Morel S (2003) Preparation of griseofulvin nanoparticles from water-dilutable microemulsions. Int J Pharm 254(2):235–242
Wang Y, Yu L, Han L, Sha X, Fang X (2007) Difunctional pluronic copolymer micelles for paclitaxel delivery: synergistic effect of folate-mediated targeting and pluronic-mediated overcoming multidrug resistance in tumor cell lines. Int J Pharm 337:63–73
Wissing SA, Kayser O, Muller RH (2004) Solid lipid nanoparticles for parenteral drug delivery. Adv Drug Deliv Rev 56:1257–1272
Zur Muhlen A, Schwarz C, Mehnert W (1998) Solid lipid nanoparticles (SLN) for controlled drug delivery—drug release and release mechanism. Eur J Pharm Biopharm 45(2):149–155
Acknowledgments
This article does not contain any studies with human and animal subjects performed by any of the authors. All authors (J. S. Baek, C. W. Cho) declare that they have no conflict of interest. This work was supported by the Basic Science Research Program (2009-009385 and 2012R1A1B5003358) through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Baek, JS., Cho, CW. Comparison of solid lipid nanoparticles for encapsulating paclitaxel or docetaxel. Journal of Pharmaceutical Investigation 45, 625–631 (2015). https://doi.org/10.1007/s40005-015-0182-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40005-015-0182-3