Abstract
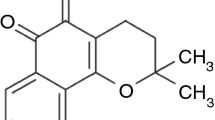
Propolis is a resinous product collected by honeybees from various plant sources; it is widely used in traditional medicine and has been reported to have a broad spectrum of pharmacological effects (i.e., antibacterial, antifungal, antiviral and anti-inflammatory effects). The most commonly used propolis formulations are gargles, in which propolis tinctures are diluted with water. The dilution process is accompanied by nanoprecipitation, and the propolis droplets are dispersed in the prepared gargle. In the present study, we investigated the dependence of the properties of propolis nanodispersions on the method of preparation. The particle size was found to be approximately 150 nm and was observed to decrease with increasing dilution as the zeta potential of the particles became more negative, which stabilized the dispersion. The dispersion dissolved upon alkalization and reprecipitated during acidification. The addition of salt destabilized the dispersion. The uptake of propolis from the dispersion was modeled using 1-octanol and was found to be rapid and only slightly dependent on the nanoparticle size. Propolis susceptibility tests showed that the most effective dispersion of propolis was tenfold-diluted EEP (P-80-10 and JP-80-10). The disc diffusion method was used to evaluate the antibacterial activity of chosen dispersions of propolis against Staphylococcus aureus, Escherichia coli and Candida albicans. Propolis samples with different locations of origin exhibited different effects against the strain of C. albicans.








Similar content being viewed by others
References
Banskota AH, Tezuka Y, Prasain JK, Matsushige K, Saiki I, Kadota S (1998) Chemical constituents of Brazilian propolis and their cytotoxic activities. J Nat Prod 61:896–900
Banskota AH, Tezuka Y, Adnyana IK (2001) Hepatoprotective and anti Helicobacterpylori activities of constituents from Brazilian propolis. Phytomedicine 8:16–23
Bender KI, Lutsevich AN (1988) Dependence of the absorption through the human oral mucosa of antihistaminic (H1) preparations on their physicochemical properties. Farmakol Toksikol 51:75–79
Burdock GA (1998) Review of the biological properties and toxicity of bee propolis (propolis). Food Chem Toxicol 36:347–363
Dimov V, Ivanovska N, Manolova N, Bankova V, Nikolov N, Popov S (1991) Immunomodulatory action of propolis: influence on anti-infectious protection and macrophage function. Apidologie 22:155–162
Ghisalberti EL (1979) Propolis: a review. Bee World 60:59–84
Hruby M, Filippov SK, Panek J, Novakova M, Mackova H, Kucka J, Vetvicka D, Ulbrich K (2010) Polyoxazoline thermoresponsive micelles as radionuclide delivery systems. Macromol Biosci 10:916–924
Isla MI, Moreno MIN, Sampietro AR, Vattuone MA (2001) Antioxidant activity of Argentina propolis extracts. J Ethnopharmacol 76:165–170
NCCLS, National Committee for Clinical Laboratory Standards (2000) Performance standards for antimicrobial disk susceptibility tests. Approved standard, 7th ed. NCCLS document M2-A7. NCCLS, Wayne, PA, USA
Ota C, Unterkicher C, Fantinato V, Shimizu MT (2001) Antifungal activity of propolis on different species of Candida. Mycoses 44:375–378
Pepeljnjak S, Jalsenjak I, Maysinger D (1985) Flavonoid content in propolis extracts and growth inhibition of Bacillussubtilis. Pharmazie 40:122–123
Silici S, Kutluca S (2005) Chemical composition and antibacterial activity of propolis collected by three different races of honeybees in the same region. J. Ethnopharmacol 99:69–73
Stepanek P, Konak C (1984) Quasielastic light scattering from polymers, colloids and gels. Adv Colloid Interface Sci 21:195–274
Sun F, Hayami S, Haruna S, Ogiri Y, Tanaka K, Yamada Y, Ikeda K, Yamada H, Sugimoto H, Kawai N, Kojo S (2000) In vivo antioxidative activity of propolis evaluated by the interaction with vitamin C and vitamin E and the level of lipid hydroperoxides in rats. J Agric Food Chem 48:1462–1465
Uzel A, Sorkun K, Oncag O, Cogulu D, Gencay M, Salih B (2005) Chemical compositions and antimicrobial activities of four different Anatolian propolis samples. Microbiol Res 160:189–195
Vasiluk L (2006). Development of an in vitro system to assess the oral bioavailability of hydrophobic contaminants. Dissertation, Simon Fraser University, Burnaby, Canada, p 176
Velikova M, Bankova V, Tsvetkova I, Kujumgiev A, Marcucci MC (2000) Antibacterial ent-kaurene from Brazilian propolis of native stingless bees. Fitoterapia 71:693–696
Vohlidal J, Julak A, Stulik K (1999) Chemical and analytical tables, 1st edn. Grada Publishing, Prague
Volpert R, Elstner F (1993) Biochemical activities of propolis extracts. Part I: standardization and antioxidative properties of ethanolic and aqueous derivatives. Z Naturforsch 48C:851–857
Waring MJ (2009) Defining optimum lipophilicity and molecular weight ranges for drug candidates: molecular weight dependent lower log D limits based on permeability. Bioorg Med Chem Lett 19:2844–2851
Acknowledgments
This work was supported by grant #GA202/09/2078 of the Grant Agency of the Czech Republic, grant #M200501201 of the Academy of Sciences of the Czech Republic and grant #MPO TIP FR-TI4/625 of the Ministry of Industry and Trade of the Czech Republic.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Trousil, J., Panek, J., Hruby, M. et al. Self-association of bee propolis: effects on pharmaceutical applications. Journal of Pharmaceutical Investigation 44, 15–22 (2014). https://doi.org/10.1007/s40005-013-0104-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40005-013-0104-1