Abstract
Background
Weight gain after initiation of combination antiretroviral therapy (cART) is a possible side effect of all antiretroviral regimens, but it seems to be more evident in association with integrase strand transfer inhibitors (INSTIs). So, we aimed to evaluate weight change associated with an initial cART including one INSTI or darunavir–ritonavir (DRV/r).
Methods
A retrospective, observational, cohort study of antiretroviral therapy-naive adult HIV-positive patients starting an initial cART including raltegravir (RAL), dolutegravir (DTG), elvitegravir–cobicistat (EVG), or DRV/r. We compared changes in weight and body mass index (BMI) across the four groups during a 12-month follow-up.
Results
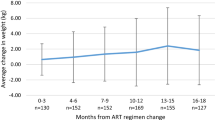
As a whole, 680 patients (470 males, mean age 42.1 years) were enrolled: 196 starting RAL, 174 DTG, 158 EVG/c, and 152 DRV/r. Baseline mean CD4 lymphocyte count was 455 cells/mm3 and 7.3% had an AIDS diagnosis. After 12 months, mean increase in body weight was 1.93 kg in the RAL group, 2.38 kg in the DTG group, 2.14 kg in the EVG group, and 1.85 in the DRV/r group. Mean increase in BMI was 0.71, 0.84, 0.77 and 0.63 kg/m2, respectively (p > 0.05 for each comparison). Therefore, no significant increases in weight and BMI were reported in each group, and no significant differences in weight and BMI changes were described across the four treatment groups.
Conclusions
In our study, patients starting an initial cART including one INSTI or DRV/r after 12 months showed a small and comparable, but not significant, increase in body weight, whose long-term clinical consequences are unknown.


Similar content being viewed by others
References
Demir OM, Candilio L, Fuster D, Muga R, Barbaro G, Colombo A, et al. Cardiovascular disease burden among human immunodeficiency virus-infected individuals. Int J Cardiol. 2018;265:195–203.
Sinha A, Feinstein MJ. Coronary artery disease manifestations in HIV: what, how, and why. Can J Cardiol. 2019;35:270–9.
Guaraldi G, Oralndo G, Zona S, Menozzi M, Carli F, Garlassi E, et al. Premature age-related comorbidities among HIV-infected persons compared with the general population. Clin Infect Dis. 2011;53:1120–6.
Rao SG, Galaviz KI, Gay HC, Wei J, Armstrong WS, del Rio C, et al. Factors associated with excess myocardial infarction risk in HIV-infected adults: a systematic review and meta-analysis. J Acquir Immune Defic Syndr. 2019 Feb 20 [Epub ahead of print].
Lang S, Mary-Krause M, Cotte L, Gilquin J, Partisani M, Simon A, et al. French Hospital Database on HIV-ANRS CO4. Increased risk of myocardial infarction in HIV-infected patients in France, relative to the general population. AIDS. 2010;24:1228–30.
Kumar S, Samaras K. The impact of weight gain during HIV treatment on risk of pre-diabetes, diabetes mellitus, cardiovascular disease, and mortality. Front Endocrinol (Lausanne). 2018;9:705.
Herrin M, Tate JP, Akgün KM, Butt AA, Crothers K, Freiberg MS, et al. Weight gain and incident diabetes among HIV-infected veterans initiating antiretroviral therapy compared with uninfected individuals. J Acquir Immune Defic Syndr. 2016;73:228–36.
Sattler FR, He J, Letendre S, Wilson C, Sanders C, Heaton R, et al. Abdominal obesity contributes to neurocognitive impairment in HIV-infected patients with increased inflammation and immune activation. J Acquir Immune Defic Syndr. 2015;68:281–8.
Bhagwat P, Ofotokun I, McComsey GA, Brown T, Moser C, Sugar CA, et al. Predictors of severe weight/body mass index gain following antiretroviral initiation. In: [Abstract 695] 24th Conference on Retroviruses and Opportunistic Infections (CROI) 13–16 February 2017.
Taramasso L, Ricci E, Menzaghi B, Orofino G, Passerini S, Madeddu G, et al. Weight gain: a possible effect of all antiretrovirals. Open Forum Infect Dis. 2017;4:ofx239.
Lakey W, Yang LY, Yancy W, Chow SC, Hicks C. Short communication: from wasting to obesity: initial antiretroviral therapy and weight gain in HIV-infected persons. AIDS Res Hum Retrovir. 2013;29:435–40.
Erlandson KM, Kitch D, Tierney C, Sax PE, Daar ES, Tebas P, et al. Weight and lean body mass change with antiretroviral initiation and impact on bone mineral density. AIDS. 2013;27:2069–79.
McComsey GA, Moser C, Currier J, Ribaudo HJ, Paczuski P, Dubé MP, et al. Body composition changes after initiation of raltegravir or protease inhibitors: actg A5260s. Clin Infect Dis. 2016;62:853–62.
Bourgi K, Jenkins C, Rebeiro PF, Lake JE, Moore RD, Mathews WC, et al. Greater weight gain among treatment-naïve persons starting integrase inhibitors. In: [Abstract 670] 26th Conference on Retroviruses and Opportunistic Infections (CROI) 4–7 March 2019.
Menard A, Meddeb L, Tissot-Dupont H, Ravaux I, Dhiver C, Mokhtari S, et al. Dolutegravir and weight gain: an unexpected bothering side effect? AIDS. 2017;31:1499–500.
Norwood J, Turner M, Bofill C, Rebeiro P, Shepherd B, Bebawy S, et al. Weight gain in persons with HIV switched from efavirenz-based to integrase strand transfer inhibitor-based regimens. J Acquir Immune Defic Syndr. 2017;76:527–31.
Kerchberger AM, Sheth AN, Angert CD, Mehta CC, Summers NA, Ofotokun I, et al. Integrase strand transfer inhibitors are associated with weight gain in women. In: [Abstract 672] 26th Conference on Retroviruses and Opportunistic Infections (CROI) 4–7 March 2019.
Lake JE, Wu K, Erlandson KM, Bares SH, Debroy P, Godfrey C, et al. Risk factors for excess weight gain following switch to integrase inhibitor-based ART. In: [Abstract 669] 26th Conference on Retroviruses and Opportunistic Infections (CROI) 4–7 March 2019.
Bares SH, Smeaton LM, Xu A, Godfrey C, McComsey GA. HIV-infected women gain more weight than HIV-infected men following the initiation of antiretroviral therapy. J Womens Health (Larchmt). 2018;27:1162–9.
Debroy P, Sim M, Erlandson KM, Falutz J, Prado CM, Brown TT, et al. Progressive increases in fat mass occur in adults living with HIV on antiretroviral therapy, but patterns differ by sex and anatomic depot. J Antimicrob Chemother. 2019;74:1028–34.
Gomez M, Seybold U, Roider J, Harter G, Bogner JR. A retrospective analysis of weight changes in HIV-positive patients switching from a tenofovir disoproxil fumarate (TDF)- to a tenofovir alafenamide fumarate (TAF)-containing treatment regimen in one German university hospital in 2015-2017. Infection. 2019;47:95–102.
Kouanfack C, Mpoudi-Etame M, Omgba Bassega P, Eymard-Duvernay S, Leroy S, Boyer S, NAMSAL ANRS 12313 Study Group, et al. Dolutegravir-based or low-dose efavirenz-based regimen for the treatment of HIV-1. N Engl J Med. 2019;381:816–26.
Venter WD, Moorhouse M, Sokhela S, Fairlie L, Mashabane N, Masenya M, et al. Dolutegravir plus two different prodrugs of tenofovir to treat HIV. N Engl J Med. 2019;381:803–15.
Achhra AC, Mocroft A, Reiss P, Sabin C, Ryom L, de Wit S, et al. Short-term weight gain after antiretroviral therapy initiation and subsequent risk of cardiovascular disease and diabetes: the D:A:D study. HIV Med. 2016;17:255–68.
Yuh B, Tate J, Butt AA, Crothers K, Freiberg M, Leaf D, et al. Weight change after antiretroviral therapy and mortality. Clin Infect Dis. 2015;60:1852–9.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Calza, L., Colangeli, V., Borderi, M. et al. Weight gain in antiretroviral therapy-naive HIV-1-infected patients starting a regimen including an integrase strand transfer inhibitor or darunavir/ritonavir. Infection 48, 213–221 (2020). https://doi.org/10.1007/s15010-019-01376-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s15010-019-01376-5