Abstract
Purpose
To investigate the microorganisms in culture-proven endophthalmitis and their susceptibilities to antimicrobial agents commonly used in South Korea.
Methods
Medical records of consecutive patients with culture-proven endophthalmitis at eight institutions between 1 January 2004 and 31 July 31 2010 were reviewed. Four categories of endophthalmitis were studied: postoperative, posttraumatic, endogenous, and unspecified. Outcome measures were culture-proven infectious organisms, antimicrobial susceptibilities, and final visual acuity in the patients.
Results
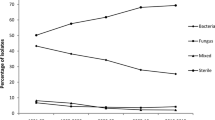
A total of 93 microorganisms were identified from 103 patients during the study period. The positive culture rate was 59.2 % (103/174). The most common organisms identified were Enterococcus faecalis (in 20.8 % of patients, 20/96), Staphylococcus epidermidis (18.8 %, 18/96), other coagulase-negative staphylococci (10.4 %, 10/96), Pseudomonas aeruginosa (6.3 %, 6/96), and Klebsiella pneumoniae (6.3 %, 6/96). Two cases of Enterococcus faecium (2.1 %) were recognized. Overall, 70 of 96 (73.0 %) isolates were Gram-positive bacteria, 22 (23.0 %) were Gram-negative bacteria, and 4 (4.2 %) were fungi. The most common organisms resulting in reduced light perception were E. faecalis and K. pneumoniae.
Conclusions
The emergence of E. faecalis in endophthalmitis is mainly caused by the high incidence of E. faecalis in postoperative endophthalmitis. This increase also impacts the final visual acuity of the patients.
Similar content being viewed by others
References
Mamalis N. Endophthalmitis. J Cataract Refract Surg. 2002;28:729–30.
[No Authors Listed]. Results of the Endophthalmitis Vitrectomy Study. A randomized trial of immediate vitrectomy and of intravenous antibiotics for the treatment of postoperative bacterial endophthalmitis. Endophthalmitis Vitrectomy Study Group. Arch Ophthalmol. 1995;113:1479–96.
Kattan HM, Flynn HW Jr, Pflugfelder SC, Robertson C, Forster RK. Nosocomial endophthalmitis survey: current incidence of infection after intraocular surgery. Ophthalmology. 1991;98:227–38.
Aaberg TM Jr, Flynn HW Jr, Schiffman J, Newton J. Nosocomial acute-onset postoperative endophthalmitis survey: a 10-year review of incidence and outcomes. Ophthalmology. 1998;105:1004–10.
Taban M, Behrens A, Newcomb RL, Nobe MY, Saedi G, Sweet PM, et al. Acute endophthalmitis following cataract surgery: a systematic review of the literature. Arch Ophthalmol. 2005;123:613–20.
[No Authors Listed]. Microbiologic factors and visual outcome in the Endophthalmitis Vitrectomy Study. Am J Ophthalmol. 1996;122:830–846.
Benz MS, Scott IU, Flynn HW Jr, Unonius N, Miller D. Endophthalmitis isolates and antibiotic sensitivities: a 6-year review of culture-proven cases. Am J Ophthalmol. 2004;137:38–42.
Pijl BJ, Theelen T, Tilanus MA, Rentenaar R, Crama N. Acute endophthalmitis after cataract surgery: 250 consecutive cases treated at a tertiary referral center in the Netherlands. Am J Ophthalmol. 2010;149:482–7.
Choi S, Hahn TW, Osterhout G, O’Brien TP. Comparative intravitreal antibiotic therapy for experimental Enterococcus faecalis endophthalmitis. Arch Ophthalmol. 1996;114:61–5.
Han DP, Wisniewski SR, Wilson LA, Barza M, Vine AK, Doft BH, et al. Spectrum and susceptibilities of microbiologic isolates in the Endophthalmitis Vitrectomy Study. Am J Ophthalmol. 1996;122:1–17.
Chen KJ, Lai CC, Sun MH, Chen TL, Yang KJ, Kuo YH, et al. Postcataract endophthalmitis caused by Enterococcus faecalis. Ocul Immunol Inflamm. 2009;17:364–9.
Scott IU, Loo RH, Flynn HW Jr, Miller D. Endophthalmitis caused by Enterococcus faecalis: antibiotic selection and treatment outcomes. Ophthalmology. 2003;110:1573–7.
Struelens MJ. The epidemiology of antimicrobial resistance in hospital acquired infections: problems and possible solutions. BMJ. 1998;317:652–4.
Callegan MC, Ramirez R, Kane ST, Cochran DC, Jensen H. Antibacterial activity of the fourth-generation fluoroquinolones gatifloxacin and moxifloxacin against ocular pathogens. Adv Ther. 2003;20:246–52.
Rishi E, Rishi P, Nandi K, Shroff D, Therese KL. Endophthalmitis caused by Enterococcus faecalis: a case series. Retina. 2009;29:214–7.
Clinical and Laboratory Standards Institute (CLSI). Clinical and Laboratory Standards Institute document. CLSI, Wayne, Pa.; 2005.
Kunimoto DY, Das T, Sharma S, Jalali S, Majji AB, Gopinathan U, et al. Microbiologic spectrum and susceptibility of isolates: part I. Postoperative endophthalmitis. Endophthalmitis Research Group. Am J Ophthalmol. 1999;128:240–2.
Park SH, Lim JA, Choi JS, Kim KA, Joo CK. The resistance patterns of normal ocular bacterial flora to 4 fluoroquinolone antibiotics. Cornea. 2009;28:68–72.
Friling E, Lundstrom M, Stenevi U, Montan P. Six-year incidence of endophthalmitis after cataract surgery: Swedish national study. J Cataract Refract Surg. 2013;39:15–21.
Acknowledgment
This work was supported by the 2013 Inje University research grant.
Conflict of interest
No conflicting relationships exist for any authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
H.W. Kim, S.Y. Kim, and I.Y. Chung contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kim, H.W., Kim, S.Y., Chung, I.Y. et al. Emergence of Enterococcus species in the infectious microorganisms cultured from patients with endophthalmitis in South Korea. Infection 42, 113–118 (2014). https://doi.org/10.1007/s15010-013-0530-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s15010-013-0530-z