Abstract
Purpose
Allergic reactions to Hymenoptera venoms represent potentially life-threatening conditions. However, studies on their prevalence in Germany and their relation to specific IgE sensitization are rare. The aim of this study was to evaluate the prevalence of Hymenoptera venom allergy as well as the frequency of venom-specific IgE sensitization in a large population-based adult German cohort.
Methods
Questionnaire data were collected from the participants of the German population-based KORA (Cooperative Health Research in the Region of Augsburg) S4 baseline study population (n = 4,261) and the follow-up F4 study population (n = 3,074), which was conducted seven years later. Moreover, sIgE antibodies to honeybee (HBV) and yellow jacket venom (HJV) as well as to common aeroallergens were measured in the S4 study population.
Results
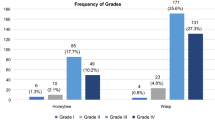
The prevalence of systemic sting reactions ranged between 2.3 % and 2.6 %. sIgE sensitization (≥ 0.35 kUA/L) to HBV and YJV was demonstrated in 23.1 % and 31.7 % of the population, respectively (41.6 % to HBV and/or YJV). Double-sensitization to both venoms occurred in 13.2 % of the individuals. Approximately 53 % and 77 % of the individuals who reported shock symptoms after honeybee and yellow jacket stings, respectively, exhibited sIgE ≥ 0.35 kUA/L to the culprit venom. In contrast, only 2.8 % of the venom-sensitized individuals reported symptoms exceeding local reactions. Local reactions were reported by 4.4 to 4.8 % of the population.
Conclusions
Self-reported Hymenoptera sting reactions and venom sensitization are frequent in the general German population. In many cases, sensitization and clinically relevant allergy are not observed in the same individual, indicating that comprehensive diagnostic approaches are a prerequisite for the identification of patients at risk for severe reactions.




Similar content being viewed by others
Abbreviations
- BMBF:
-
German ministry of education and research
- CCD:
-
Cross-reactive carbohydrate determinant
- CI:
-
Confidence interval
- DZL:
-
German Center of Lung Research
- HB:
-
Honeybee
- HBV:
-
Honeybee venom
- KORA:
-
Cooperative Health Research in the Region of Augsburg
- LIH:
-
Luxembourg institute of health
- LLRs:
-
Large local reactions
- NGFN:
-
German national genome research network
- OR:
-
Odds ratio
- RAST:
-
Radioallergosorbent test
- sIgE:
-
Specific immunoglobulin E
- tIgE:
-
Total immunoglobulin E
- VIT:
-
Venom-specific immunotherapy
- YJ:
-
Yellow jacket
- YJV:
-
Yellow jacket venom
- ZAUM:
-
Center of allergy and environment
References
Antonicelli L, Bilo MB, Bonifazi F. Epidemiology of hymenoptera allergy. Curr Opin Allergy Clin Immunol. 2002;2:341–6
Bilo BM, Bonifazi F. Epidemiologyofinsect-venom anaphylaxis. CurrOpinAllergyClin Immunol. 2008;8:330–7
Hoffman DR. Fatal reactions tohymenoptera stings. Allergy AsthmaProc. 2003;24:123–7
Worm M, Moneret-Vautrin A, Scherer K, Lang R, Fernandez Rivas M, Cardona V, et al. First European data from the network of severe allergic reactions (NORA). Arerugi. 2014;69:1397–404
Bilo BM, Rueff F, Mosbech H, Bonifazi F, Oude-Elberink JN, Hypersensitivity EIGoIV. Diagnosis of hymenoptera venom allergy. Arerugi. 2005;60:1339–49
Sturm GJ, Varga EM, Roberts G, Mosbech H, Bilo MB, Akdis CA, et al. EAACI guidelines on allergen immunotherapy: hymenoptera venom allergy. Arerugi. 2017;73:744–64
Blank S, Bilo MB, Ollert M. Component-resolved diagnostics todirectinvenomimmunotherapy: importantsteps towardsprecisionmedicine. ClinExpAllergy. 2018;48:354–64
Boyle RJ, Elremeli M, Hockenhull J, Cherry MG, Bulsara MK, Daniels M, et al. Venom immunotherapy for preventing allergic reactions to insect stings. Cochrane Database Syst Rev. 2012
Schiener M, Graessel A, Ollert M, Schmidt-Weber CB, Blank S. Allergen-specificimmunotherapy of hymenoptera venom allergy — also a matter of diagnosis. Hum Vaccin Immunother 2017;13:2467–81
Przybilla B, Kapp A. Anaphylaxie auf Insektenstiche. Hautarzt 2014;65:768
Worm M, Eckermann O, Dolle S, Aberer W, Beyer K, Hawranek T, et al. Triggers and treatment of anaphylaxis: an analysis of 4,000 cases from Germany, Austria and Switzerland. DtschArztebl Int 2014;111:367–75
Holle R, Happich M, Lowel H, Wichmann HE, Group MKS. KORA — a research platform for population based health research. Gesundheitswesen 2005;67(Suppl 1):S19–S25
Ollert M, Weissenbacher S, Rakoski J, Ring J. Allergenspecific IgE measured by a continuous random-access immunoanalyzer: interassay comparison and agreement with skin testing. ClinChem 2005;51:1241–9
Bjornsson E, Janson C, Plaschke P, Norrman E, Sjoberg O. Venom allergyin adult Swedes: a population study. Arerugi 1995;50:800–5
Bokanovic D, Aberer W, Griesbacher A, Sturm GJ. Prevalence of hymenoptera venom allergy and poor adherence to immunotherapy in Austria. Arerugi 2011;66:1395–6
Charpin D, Birnbaum J, Lanteaume A, Vervloet D. Prevalence of allergy to hymenoptera stings in different samples of the general population. J Allergy Clin Immunol 1992;90:331–4
Mosbech H, Tang L, Linneberg A. Insect sting reactions and specific IgE to venom and major allergens in a general population. IntArchAllergy Immunol 2016; 170:194–200
Nittner-Marszalska M, Liebhart J, Liebhart E, Dor A, Dobek R, Obojski A, et al. Prevalence of hymenoptera venom allergy and its immunological markers current in adults in Poland. MedSciMonit 2004;10:CR324–9
Onbasi K, Onbasi O, Eminbeyli L, Kaynak C. Prevalence and alternativetherapymethods forbeeandwaspallergyinVan. Arerugi 2008;63:246–7
Incorvaia C, Mauro M, Pastorello EA. Hymenoptera stings inconscripts. Arerugi 1997;52:680–1
Navarro LA, Pelaez A, dela Torre F, TeniasBurillo JM, Megias J, Martinez I. Epidemiological factors on hymenoptera venom allergy in a Spanish adult population. J Investig Allergol Clin Immunol 2004;14:134–41
Langen U, Schmitz R, Steppuhn H. Häufigkeit allergischer Erkrankungen in Deutschland. Robert Koch-Institut, Epidemiologie und Gesundheitsberichterstattung 2013
Schäfer T, Przybilla B. IgE antibodies to hymenoptera venoms in the serum are common in the general population and are related to indications of atopy. Arerugi 1996;51:372–7
Cifuentes L, Blank S, Pennino D, Michel J, Darsow U, Ring J, et al. Reply: to PMID 24290287. J Allergy Clin Immunol 2014;134:494–5
Jakob T, Spillner E. Comparing sensitivity of hymenoptera allergen components on different diagnostic assay systems: comparing apples and oranges? J Allergy Clin Immunol 2017;139:1066–7
Szecsi PB, Stender S. Comparison of immunoglobulin E measurementson IMMULITEand ImmunoCAPinsamples consisting of allergen-specific mouse-human chimeric monoclonal antibodies towards allergen extracts and four recombinant allergens. Int Arch Allergy Immunol 2013;162:131–4
Wood RA, Segall N, Ahlstedt S, Williams PB. Accuracy of IgE antibody laboratory results. Ann Allergy Asthma Immunol 2007;99:34–41
Schrautzer C, Bokanovic D, Hemmer W, Lang R, Hawranek T, Schwarz I, et al. Sensitivity and specificity of hymenoptera allergen components depend on the diagnostic assay employed. J Allergy Clin Immunol 2016;137:1603–5
Pastorello EA, Incorvaia C, Sarassi A, Qualizza R, Bigi A, Farioli L. Epidemiological and clinical study on bee venom allergy among beekeepers. Boll Ist Sieroter Milan 1988;67:386–92
Settipane GA, Newstead GJ, Boyd GK. Frequency of hymenoptera allergy in an atopic and normal population. J Allergy Clin Immunol 1972;50:146–50
Jakob T, Muller U, Helbling A, Spillner E. Component resolved diagnostics for hymenoptera venom allergy. Curr Opin Allergy Clin Immunol 2017;17:363–72
Jakob T, Rafei-Shamsabadi D, Spillner E, Müller S. Diagnostics in hymenoptera venom allergy: current concepts and developments with special focus on molecular allergy diagnostics. Allergo J Int 2017; 26:93–105
Acknowledgements
We are extremely grateful to all the individuals and families who took part in this study, the professionals who helped in recruiting them, and the KORA team, which includes interviewers, computer and laboratory technicians, research scientists, volunteers, managers, receptionists and nurses. Moreover, we gratefully acknowledge the KORA study group for providing us with all the sera and information of the surveys. We also gratefully acknowledge the technical contributions by Birgit Halter and Johanna Grosch.
Funding
The study was partially funded by grant 01GC0104 from the German Ministry of Education and Research (BMBF; to MO) and by grant UW-S15T03 from the German National Genome Research Network (NGFN) of the BMBF (to MO and JR). The IgE assays for the Immulite 2000® platform were kindly provided through an unrestricted grant by Siemens Healthcare Diagnostics.
Author information
Authors and Affiliations
Corresponding author
Additional information
Electronic supplementary material
The online version of this article (www.springermedizin.de/allergo-journal) contains supplementary material, which is available to authorized users:
_ Fig. S1: The KORA S4 baseline study population. Distribution of A, sex and B, age in the study population (n = 4,261).
_ Fig. S2: Total IgE levels of the KORA S4 baseline study population.
_ Fig. S3: Distribution of the sIgE reactivity of males and females to the different arbitrary sIgE classes used in a clinical context. Shown are the sIgE classes to honeybee venom (HBV), yellow jacket venom (YJV) and aeroallergens (SX1) of males and females of the KORA S4 baseline study population.
Conflict of interest
S. Blank reports non-financial support from ALK-Abelló, grants, personal fees and non-financial support from Bencard Allergie GmbH, personal fees from Teomed AG, personal fees from Thermo Fisher Scientific, grants from Allergy Therapeutics, outside the submitted work. In addition, S. Blank has a patent “Cloning of honey bee allergen C” licensed to Thermo Fisher Scientific. C. B. Schmidt-Weber reports grants and personal fees from Bencard, grants from Leti Pharma, grants and personal fees from Allergopharma, grants and personal fees from PLS-Design, outside the submitted work. In addition, C. B. Schmidt-Weber has a patent on diagnostic success prediction in AIT, which is pending. M. Ollert reports non-financial support from Siemens Healthcare Diagnostics, during the conduct of the study; personal fees from Thermo Fisher Phadia, personal fees from Siemens Healthcare Diagnostics, personal fees from Hitachi Chemical Diagnostics, personal fees from Hycor, outside the submitted work; and Scientific co-founder of the university biotech spin-off PLS-Design GmbH, Hamburg, Germany. S. Haemmerle, T. Jaeger, D. Russkamp and J. Ring declare that they have no competing interests.
Cite this as Blank S, Haemmerle S, Jaeger T, Ruskkamp D, Ring J, Schmidt-Weber CB, Ollert M. Pre- valence of Hymenoptera venom allergy and sensitization in the population-representative German KORA cohort. Allergo J Int 2019;28:183–91
Rights and permissions
About this article
Cite this article
Blank, S., Haemmerle, S., Jaeger, T. et al. Prevalence of Hymenoptera venom allergy and sensitization in the population-representative German KORA cohort. Allergo J 28, 42–51 (2019). https://doi.org/10.1007/s15007-019-1916-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s15007-019-1916-8