Abstract
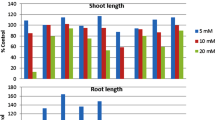
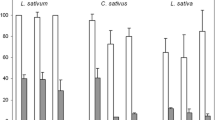
Phenolic acid is a very important class of allelochemicals with allelopathic weed control activity. In this study, three benzoic acid derivatives (syringic, 4-hydroxybenzoic, and vanillic acids), three cinnamic acid derivatives (cinnamic, 4-hydroxycinnamic, and ferulic acids) were tested, and high-performance liquid chromatography was used to conduct a dynamic analysis on the changes in the concentration of phenolic acids in a bioassay based on the initial concentration and test time. The results showed that the concentration of individual phenolic acids and a solution of mixed phenolic acids decreased to a certain extent irrespective of environment, i.e., bioassay (4–7 days) or a rice-growing environment, and a significant decrease in concentration was measured after 48 h. Based on the above results, the laboratory bioassay was conducted using a fresh solution of phenolic acids every 48 h. The results showed that the instability of phenolic acid could affect its weed control activity, and this effect was more significant for high concentrations of phenolic acids. On the other hand, changing the solution did not have a significant impact on the weed control activity of phenolic acids in the natural environment (pH 6.50), in which allelopathic rice release phenolic acids. These results reveal the instability of phenolic acids could significantly reduce the inhibition rate on the growth index for receptor plants in an indoor bioassay.




Similar content being viewed by others
References
Amy MC, William CK, Cheng HH, Konrad H (2004) Sorption–desorption of phenolic acids as affected by soil properties. Biol Fertil Soils 39:235–242
Bhadoria PBS (2011) Allelopathy: a natural way towards weed management. A J Exp Agr 1:7–20
Blum U (2004) Fate of phenolic allelochemicals in soils—the role of soil and rhizosphere microorganisms. Allelopathy: Chemistry and Mode of Action of Allelochemicals. CRC Press, Cambridge, pp 57–76
Blum U, Wentworth TR, Klein K, Worsham AD, King LD, Gerig TM, Lyu SW (1991) Phenolic acid content of soils from wheat-no till, wheat–conventional till, and fallow–conventional till soybean cropping systems. J Chem Ecol 17:1045–1068
Cecile B, Xiaohan Y, Leslie AW (2003) The role of root exudates and allelochemicals in the rhizosphere. Plant Soil 256:67–83
Cheema ZA, Khaliq A (2000) Use of sorghum allelopathic properties to control weeds in irrigated wheat in a semi arid region of Punjab. Agr Ecosyst Environ 79:105–112
Chung IM, Kim KH, Ahn JK, Chun SC, Kim CS, Kim JT, Kim SH (2002) Screening of allelochemicals on barnyardgrass (Echinochloa crus-galli) and identification of potentially allelopathic compounds from rice (Oryza sativa) variety hull extracts. Crop Prot 21:913–920
Duke SO, Dayan FE, Romagni JG, Rimando A (2000) Natural products as sources of herbicides: current status and future trends. Weed Res 40:99–111
Francisco AM, Molinillo JMG, Rosa MV, Galindo JCG (2007) Allelopathy—a natural alternative for weed control. Pest Manag Sci 63:327–348
Kohli RK, Batish D, Singh HP (1998) Allelopathy and its implications in agroecosystems. J Crop Prod 1:169–202
Mann J (1987) In secondary metabolism, 2nd edn. Clarendon Press, Oxford
Mattice J, Lavy T, Skulman B, Dilday RH (1998) Searching for allelochemicals in rice that control ducksalad. In: Proceedings of 1998 Workshop on Allelopathy in Rice. International Rice Research Institute, Manila, pp 81–98
Olofsdotter M, Navarez D, Rebulanan M, Streibig JC (1999) Weed suppressing rice cultivars—does allelopathy play a role? Weed Res 39:441–454
Oueslati O (2003) Allelopathy in two durum wheat (Triticum durum L.) varieties. Agr Ecosyst Environ 96:161–163
Rice EL (1984) In allelopathy, 2nd edn. Academic Press, Orlando
Rimando A, Olofsdotter M, Duke SO (2001) Searching for rice allelochemicals an example of bioassay-guide isolation. Agron J 93:16–20
Seal AN, Pratley JE, Haig T, An M (2004a) Identification and quantitation of compounds in a series of allelopathic and non-allelopathic rice root exudates. J Chem Ecol 30:1647–1662
Seal AN, Haig T, Pratley JE (2004b) Evaluation of putative allelochemicals in rice roots exudates for their role in the suppression of arrowhead root growth. J Chem Ecol 30:1663–1678
Seigler DS (1996) Chemistry and mechanisms of allelopathic interactions. Agron J 88:876–885
Singh HP, Kohli RK, Batish DR (2001) Allelopathy in agro-ecosystems: an overview. J Crop Prod 4:1–41
Siqueira JO, Nair MG, Hammerschmidt R, Safir GR, Putnam AR (1991) Significance of phenolic compounds in plant–soil–microbial systems. Crit Rev Plant Sci 10:63–121
Smith SK, Ley RE (1999) Microbial competition and soil structure limit the expression of allelopathy. In: Principles and practices in plant ecology: allelochemical interactions. CRC Press, Boca Raton, pp 339–351
Wu H, Pratley J, Lemerle D, Haig T, An M (2001) Screening methods for the evaluation of crop allelopathic potential. Bot Rev 67:403–412
Zheng XY, Ye RJ, He YB, Zhou XC, Lin RY, Lin WX (2013) Determination of 10 phenolic acids in root exudates of rice using solid-phase extraction and high performance liquid chromatography. J Yunnan Univ (Natu Sci Edi) 35:219–224
Acknowledgments
This work was supported by National Natural Science Foundation of China (Grant No. 31070447 and No. 31370380); Natural Science Foundation of Fujian Province (Grant No. 2012J01077); and Key Program of Ecology (6108c0502, 6112c0601) of Fujian Province.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, J., Zhang, Q., Hu, W. et al. Stability of phenolic acids and the effect on weed control activity. J Korean Soc Appl Biol Chem 58, 919–926 (2015). https://doi.org/10.1007/s13765-015-0124-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13765-015-0124-9