Abstract
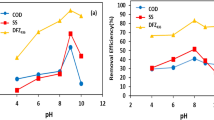
The inappropriate discharge of industrial effluents into water bodies causes significant damage to the environment. Proper treatment and disposal are crucial for the conservation of aquatic ecosystems. Advanced oxidative processes are effective alternatives for treating complex effluents, including those generated in the production of paints. The Fenton process is particularly notable among advanced oxidative processes because of its high ability to oxidize persistent contaminants using hydrogen peroxide as an oxidizing agent and ferrous ions as a catalyst. The purpose of this study was to optimize the Fenton process and substitute hydrogen peroxide with peracetic acid and ferrous ions with ferric together with a coagulation process known as the Fenton-type coagulation process for treating paint production effluents. To assess the turbidity removal efficiency, varying concentrations of peracetic acid and ferric chloride were tested. Significant results were obtained with a concentration of 200 mgL−1 of ferric chloride and 15 mgL−1 of peracetic acid when the reagents were added simultaneously, resulting in a removal of 98.4%. For the chemical oxygen demand parameters, the reduction reached 76.8%, total solids were reduced by 85%, color removal was 98.6%, total coliforms were reduced by 96.6%, and Escherichia coli reduction reached 96.4%, accompanied by an increase in the sedimentation speed.







Similar content being viewed by others
References
Aboulhassan MA, Souabi S, Yaacoubi A, Baudu M (2006) Improvement of paint effluents coagulation using natural and synthetic coagulant aids. J Haz Mat 138:40–45. https://doi.org/10.1016/j.jhazmat.2006.05.040
Andersen FA, Brečević L (1992) Infrared spectra of amorphous and crystalline calcium carbonate. ChemInform 23
Ao X, Eloranta J, Huang C, Santoro D, Sun W, Lu Z, Li C (2021) Peracetic acid-based advanced oxidation processes for decontamination and disinfection of water: a review. Water Res 188:116479. https://doi.org/10.1016/j.watres.2020.116479
APHA (2017) Standard methods for examining water and wastewater, 23rd edn. American Public Health Association, Washington, DC
Baba Y, Yatagai T, Harada T, Kawase Y (2015) Hydroxyl radical generation in the photo-Fenton process: e of carboxylic acids on iron redox cycling. Chem Eng J 277:229–241. https://doi.org/10.1016/j.cej.2015.04.103
Bokare AD, Choi W (2014) Review of iron-free Fenton-like systems for activating H2O2 in advanced oxidation processes. J Haz Mat 275:121–135. https://doi.org/10.1016/j.jhazmat.2014.04.054
Cavallini GS, Vidal CM, de Souza JB (2016) Post-treatment of the anaerobic reactor effluent using coagulation/oxidation followed by double filtration. Environ Sci Pollut Res 23:6244–6252. https://doi.org/10.1007/s11356-015-5836-1
Cavallini GS, Vidal CM, de Souza JB (2015) Fenton coagulation/oxidation using Fe2+ and Fe3+ ions and peracetic acid for the treatment of wastewater. Orbital 7(3):253–263. https://doi.org/10.17807/orbital.v7i3.626
Da Silva WP, Carlos TD, Cavallini GS, Pereira DH (2020) Peracetic acid: Structural elucidation for applications in wastewater treatment. Water Res 168. https://doi.org/10.1016/j.watres.2019.115143
Garand E, Wende T, Goebbert DJ, Bergmann R, Meijer G, Neumark DM (2010) Infrared spectroscopy of hydrated bicarbonate anion clusters: HCO3−(H2O). J Am Chem Soc 132:849–856. https://doi.org/10.1021/ja9093132
Grube M, Lin JG, Lee PH, Kokorevicha S (2006) Evaluation of sewage sludge-based compost by FTIR spectroscopy. Geoderma 130:324–333. https://doi.org/10.1016/j.geoderma.2005.02.005
Herney-Ramirez J, Vicente MA, Madeira LM (2010) Heterogenous photo-fenton oxidation with pillared clay-based catalysts for wastewater treatment: a review. Appl Catal B 98:10–26. https://doi.org/10.1016/j.apcatb.2010.05.004
Joshi S, Kalyanasundaram S, Balasubramanian V (2013) Quantitative analysis of sodium carbonate and sodium bicarbonate in solid mixtures using fourier-transform infrared spectroscopy (FT-IR). Appl Spectrosc 67:841–845. https://doi.org/10.1366/12-06915
Larkin PJ (2018) Chapter 6: IR and Raman Spectra–Structure correlations: characteristic group frequencies. In: Larkin PJ (edn) Infrared and Raman Spectroscopy, 2nd edn.Elsevier, Amsterdam, pp 85–134
Lin R, Li Y, Yong T, Cao W, Wu J, Shen Y (2022) Synergistic effects of oxidation, coagulation, and adsorption in the integrated Fenton-based process for wastewater treatment: a review. J Environ Manag 306:114460
Mishra NS, Reddy R, Kuila A, Rani A, Mukherjee P, Nawaz A, Pichiah S (2017) A review on advanced oxidation processes for effective water treatment. Curr World Environ 12:3. https://doi.org/10.12944/CWE.12.3.02
Nair KS, Manu B, Azhoni A (2021) Sustainable treatment of paint industry wastewater: Current techniques and challenges. J Environ Man 296:113105
Nicholas N (2023) Innovation in industrial wastewater treatment—sustainable water reuse strategies applied to the paint and pigment dye industry. Genes. https://www.linkedin.com/pulse/innovation-industrial-wastewater-treatment-water-reuse-nicholas. Accessed on 24 April 2023
Oriekhova O, Stoll S (2014) Investigation of FeCl3 induced coagulation processes using electrophoretic measurement, nanoparticle tracking analysis and dynamic light scattering: Importance of pH and colloid surface charge. Col Surf A Phys and Eng Asp 461:212–219. https://doi.org/10.1016/j.colsurfa.2014.07.049
Parveen MF, Umapathy S, Dhanalakshmi V, Anbarasan R (2010) Synthesis and characterizations of nanosized iron(II) hydroxide and iron(II) hydroxide/poly(vinyl alcohol) nanocomposite. J of Appl Poly Sci 118:1728–1737. https://doi.org/10.1002/app.32296
Pinto PS, Lanza GD, Ardisson JD, Lago RM (2019) Controlled dehydration of Fe(OH)3 to Fe2O3: developing mesopores with complexing iron species for the adsorption of β-lactam antibiotics. J Braz Chem Soc 30
Rahmani K, Ahmadi M, Rahmani A, Rahmani H (2017) Removal of benzotriazole by Photo-Fenton like process using nano zero-valent iron: response surface methodology with a Box-Behnken design. Pol J Chem Tech 19:104–112. https://doi.org/10.1515/pjct-2017-0015
Rémazeilles C, Refait P (2009) Fe(II) hydroxycarbonate Fe2(OH)2CO3 (chukanovite) as iron corrosion product: synthesis and study by Fourier Transform Infrared Spectroscopy. Polyhedron 28:749–756. https://doi.org/10.1016/j.poly.2008.12.034
Roberts JD, Caserio MC (1977) Basic principles of organic chemistry. WA Benjamin. ISBN: 9780805383294
Sayın FE, Karatas O, Özbay I, Gengec E, Khataee A (2022) Treatment of real printing and packaging wastewater by combination of coagulation with Fenton and photo-Fenton processes. Chemosphere 306:135539. https://doi.org/10.1016/j.chemosphere.2022.135539
STATISTA (2023) Chemicals & Resources. https://www.statista.com/statistics/1042799/paints-and-coatings-market-volume-worldwide/. Accessed on 06 Abril 2023
Vernekar D, Jagadeesan D (2015) Tunable acid–base bifunctional catalytic activity of FeOOH in an orthogonal tandem reaction. Cat Sci & Tech 5:4029–4038. https://doi.org/10.1039/C5CY00361J
Viktoryová N, Szarka A, Hrouzková S (2022) Recent developments and emerging trends in paint industry wastewater treatment methods include. Appl Sci 12:10678. https://doi.org/10.3390/app122010678
Wang HL, Cui JY, Jiang WF (2011) Synthesis, characterization and flocculation activity of novel Fe(OH)3–polyacrylamide hybrid polymer. Mat Chem and Phys 130:993–999. https://doi.org/10.1016/j.matchemphys.2011.08.027
Wang X, Andrews L (2006) Infrared Spectra of M(OH)1,2,3 (M = Mn, Fe, Co, Ni) molecules in solid argon and the character of first row transition metal hydroxide bonding. J Phys Chem A 110:10035–10045. https://doi.org/10.1021/jp0624698
Xie P, Chen Y, Ma J, Zhang X, Zou J, Wang Z (2016) A mini review of preoxidation to improve coagulation. Chemosphere 155:550–563. https://doi.org/10.1016/j.chemosphere.2016.04.003
Zhang F, Li Y, Zhaojian L, Tong W, Huang Y (2021) Experimental investigation on the oxidation of printing ink wastewater under hydrothermal flames. J Environ Chem Eng 9:106745. https://doi.org/10.1016/j.jece.2021.106745
Zhu L, Hu T, Li S, Nugroho YK, Li B, Cao J, Show P, Hiltunen E (2020) Effects of operating parameters on algae Chlorella vulgaris biomass harvesting and lipid extraction using metal sulfates as flocculants. Biomass Bioenergy 132:105433. https://doi.org/10.1016/j.biombioe.2019.105433
Acknowledgements
The authors thank Conselho Nacional de Desenvolvimento Científico Tecnológico (CNPq-Brazil), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES-Brazil), Thech Desinfecção and the Graduate Program in Biodiversity and Biotechnology-BIONORTE. Renato A. Sarmento thanks CNPq, through the Research Productivity programme (Project 306011/2022-0), and Universal Project 405875/2021-5). We would like to thank Editage (www.editage.com) for English language editing.
Funding
Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Coordination for the Improvement of Higher Education Personnel – Brazil (CAPES), Financing Code 001 CAPES; PROPESQ and the Federal University of Tocantins; CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnológico, Brazil) Apoio à Formação de Doutores em Areas Estratégicas, project number: 142525/2020–0; Tocantins Research Support Foundation—FAPT; and PDPG-POSDOC (Programa de Desenvolvimento da Pós-Graduação (PDPG) Pós-Doutorado Estratégico—Postgraduate Development Program (PGDP) Strategic Postdoctoral) No. 88887.798251/2022–00. Project nº 400859/2019–0—Training of human resources in plant production and biotechnology and biodiversity in the Amazon.
Author information
Authors and Affiliations
Contributions
Thayrine Dias Carlos: Investigation, Writing—Original Draft; Renato de Almeida Sarmento: Supervision, Writing—Revision & Editing, Funding acquisition; Nelson Luis Gonçalves Dias de Souza: Writing—Revision & Editing; Angelo Pallini: Writing—Revision & Editing; Grasiele Soares Cavallini: Conceptualization, Supervision, Wrinting—Revision & Editing.
Corresponding author
Ethics declarations
Conflict of interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Editorial responsibility: Samareh Mirkia.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Carlos, T.D., de Souza, N.L.G.D., Pallini, A. et al. Paint effluent treatment through the fenton-type coagulation process with peracetic acid and iron ions. Int. J. Environ. Sci. Technol. (2024). https://doi.org/10.1007/s13762-024-05645-1
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13762-024-05645-1