Abstract
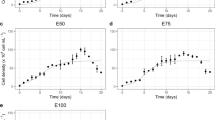
The present study artificially enhanced wild colonial Microcystis buoyancy by controlling light regime and colonial morphology with various contents of Ca2+ and tightly-bound extracellular polysaccharides (TB-EPS) extracted from cyanobacterial blooms in medium. A series of laboratory culture experiments indicated that Microcystis exhibited higher buoyancy (Relative buoyancy of 25% [RB25]: 59 ± 12%) when precultured without light irradiance for 24 hours compared with continuous lighting condition (RB25: 49 ± 7.8%). Depletion of cellular carbohydrate would be the major cause. The preculture with the addition of Ca2+ (250 mg/L) into medium in dark condition promoted colony formation and colony size expansion, which enhanced the buoyancy of Microcystis (RB25: 88 ± 7.4%), although the addition of TB-EPS (200 mg/L) together with Ca2+ caused less efficiency of the buoyancy (RB25: 55 ± 5.6%). Microcystis was originally wrapped with a large amount of TB-EPS, and negatively charged carboxy groups were considered to be present on the TB-EPS surface. Carboxy groups between the added TB-EPS and the TB-EPS wrapping Microcystis would be repulsed each other, which caused the undevelopment of colonies as well as decline of buoyancy. Furthermore, flotation velocity experiments have confirmed that the preculture of Microcystis at Ca2+ = 500 mg/L, in particular, had a significant acceleration of the flotation velocity (2.0 × 10–1 cm/s), and consequently promoting colony size expansion. From these results, the present study suggests that controlling light and Ca2+ concentration in solution could enhance the buoyant efficiency of Microcystis, which might become a promising method for the removal of cyanobacterial blooms.








Similar content being viewed by others
References
Abeynayaka D, Atsuzawa K, Tokunaga M, Yaneko Y (2022) Effect of aqueous metal ion solutions containing copper on cyanobacterial cells: behavior of metal ions and changes in cell structure. J Japan Inst Copp 61(1):206–212. https://doi.org/10.34562/jic.61.1_206
Ahn C, Park M, Joung S, Kim H, Jang K, Oh H (2003) Growth inhibition of cyanobacteria by ultrasonic radiation: Laboratory and enclosure studies. Environ Sci Technol 37(13):3031–3037. https://doi.org/10.1021/es034048z
Anderson DM, Glibert PM, Burkholder JM (2002) Harmful algal blooms and eutrophication: nutrient sources, composition, and consequences. Estuaries Coasts 25:704–726
Cheng Y, Xiaoqing S, Miao Y, Yifei T, Weijun Z (2019) Determining the effects of polyaluminum chloride alkalinities and dosage treatments on various microalgal growth phases for the treatment of microalgae-laden water. Sep Purif Technol 209(31):202–210. https://doi.org/10.1016/j.seppur.2018.07.046
Coral LA, Zamyadi A, Barbeau B, Bassetti FJ, Lopolli FR, Prevost M (2013) Oxidation of Microcystis aeruginosa and Anabaena flos-aquae by ozone: impacts on cell integrity and chlorination by-product formation. Water Res 47(9):2983–2994. https://doi.org/10.1016/j.watres.2013.03.012
Dubois M, Gilles KA, Hamilton JK, Rebers PA, Smith F (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 28(3):350–356. https://doi.org/10.1021/ac60111a017
Figueiredo J, Pereira M, Freitas M, Orfao J (1999) Modification of the surface chemistry of activated carbons. Carbon 37(9):1379–1389. https://doi.org/10.1016/s0008-6223(98)00333-9
Graham JL, Loftin KA, Meyer MT, Ziegler AC (2010) Cyanotoxin mixtures and teste-and-odor compounds in cyanobacterial blooms from the Midwestern United States. Environ Sci Technol 44(19):7361–7368. https://doi.org/10.1021/es1008938
Gu P, Li Q, Zhang W, Zheng Z, Luo X (2020) Effects of different metal ions (Ca, Cu, Pb, Cd) on formation of cyanobacterial blooms. Ecotoxicol Environ Saf 189:109976. https://doi.org/10.1016/j.ecoenv.2019.109976
Jia Y, Thomas K (2000) Adsorption of cadmium ions on oxygen surface sites in activated carbon. Langmuir 16(3):1114–1122. https://doi.org/10.1021/la990436w
Jochimsen EM, Carmichael WW (1998) Liver failure and death after exposure to microcystins at a hemodialysis center in Brazil. N Engl J Med 338:873–878. https://doi.org/10.1056/NEJM199803263381304
Kim K, Park C, Yoon Y, Hwang S (2018) Harmful cyanobacterial material production in the North Han River (South Korea): genetic potential and temparature-dependent properties. Int J Environ Res Public Health 15(3):444. https://doi.org/10.3390/ijerph15030444
Kromkamp JC, Mur LR (1984) Buoyant density changes in the cyanobacterium Microcystis aeruginosa due to changes in the cellular carbohydrate content. FEMS Microbiol Lett 25(1):105–109. https://doi.org/10.1111/j.1574-6968.1984.tb01384.x
Li M, Zhu W, Guo L, Hu J, Chen H, Xiao M (2016) To increase size or decrease density? Different Microcystis species has different choice to form blooms. Sci Rep 6:37056. https://doi.org/10.1038/srep37056
Ma J, Brookes JD, Qin B (2014) Environmental factors controlling colony formation in blooms of the cyanobacteria Microcystis spp. in Lake Taihu. China Harmful Algae 31:136–142. https://doi.org/10.1016/j.hal.2013.10.016
Masuki S, Wakabayashi K, Takahashi S, Bessyo H, Toshima K, Kunii H (2012) Investigation of vertical migration of blue-green algae in in stratified dam reservoir. J Japan Soc Water Environ 35(9):151–157
Nakamura T, Adachi Y, Suzuki M (1993) Flotation and sedimentation of a single microcystis floc collected from surface bloom. Water Res 27(6):979–983. https://doi.org/10.1016/0043-1354(93)90061-L
Omori K, Datta T, Amano Y, Machida M (2018) Effects of different types of extracellular poly saccharides isolated from cyanobacterial blooms on the colony formation of unicellular Microcystis aeruginosa. Environ Sci Pollut Res 26:3741–3750. https://doi.org/10.1007/s11356-018-3892-z
Piel T, Sandrini G, White E, Xu T, Schuurmans J, Huisman J, Visser P (2020) Suppressing cyanobacteria with hydrogen peroxide is more effective at high light intensities. Toxins 12(1):18. https://doi.org/10.3390/toxins12010018
Pouria S, Andrade A, Barbosa J, Cavalcanti RL, Barreto VT, Ward CJ, Preiser W, Poon GK, Neild GH, Codd GA (1998) Fatal microcystin intoxication in haemodialysis unit in Caruaru. Braz Lancet 352(9121):21–26. https://doi.org/10.1016/s0140-6736(97)12285-1
Sato M, Omori K, Datta T, Amano Y, Machida M (2017) Influence of extracellular polysaccharides and calcium ion on colony formation of unicellular Microcystis aeruginosa. Environ Eng Sci 34(3):149–157. https://doi.org/10.1089/ees.2016.0135
Song W, Xie Y, Chen Q, Wang W, Li X (2022) Investigation of polyaluminum chloride (PACl) coagulation to remove cyanobacteria from maintenance to decay stage: erformance and mechanism. J Environ Chem Eng 10(5):108395. https://doi.org/10.1016/j.jece.2022.108395
Tan X, Shu X, Guo J, Parajuli K, Zhang X, Duan Z (2018) Effects of low-frequency ultrasound on Microcystis aeruginosa from cell inactivation to disruption. Bull Environ Contam Toxicol 101:117–123. https://doi.org/10.1007/s00128-018-2348-y
Wang YW, Zhao J, Li SS, Zhang LH, Wu M (2011) Effects of calcium levels on colonial aggregation and buoyancy of Microcystis aeruginosa. Current Microbiol 62:679–683. https://doi.org/10.1007/s00284-010-9762-7
Wei K, Jung S, Amano Y, Machida M (2019) Control of the buoyancy of Microcystis aeruginosa via colony formation induced by regulating extracellular polysaccharides and cationic ions. SN Appl Sci 1:1573. https://doi.org/10.1007/s42452-019-1637-3
Xiao M, Li M, Reynolds CS (2018) Colony formation in the cyanobacterium Microcystis. Biol Rev 93(3):1399–1420. https://doi.org/10.1111/brv.12401
Xu HC, Yu GH, Jiang HL (2013) Investigation on extracellular polymeric substances from mucilaginous cyanobacterial blooms in eutrophic fresh water lakes. Chemosphere 93(1):75–81. https://doi.org/10.1016/j.chemosphere.2013.04.077
Xu H, Lv H, Liu X, Wang P, Jiang H (2016) Electrolyte cations binding with extracellular polymeric substances enhanced Microcystis aggregation: implication for Microcystis bloom formation in eutrophic freshwater lakes. Environ Sci Technol 50(17):9034–9043. https://doi.org/10.1021/acs.est.6b00129
Zhang M, Wang Y, Wang Y, Li M, Zhang D, Qiang Z, Pan X (2019) Efficient elimination and re-growth inhibition of harmful bloom-forming cyanobacteria using surface-functionalized microbubbles. Water Res 161(15):473–485. https://doi.org/10.1016/j.watres.2019.06.035
Zhu W, Dai X, Li M (2014) Relationship between extracellular polysaccharide (EPS) content and colony size of Microcystis is colonial morphology dependent. Biochem Syst Ecol 55:346–350. https://doi.org/10.1016/j.bse.2014.04.0
Acknowledgements
The present study was supported in part by Grants-in-Aid for Scientific Research (C) from the Japan Society for the Promotion of Science (KAKENHI Grant No. JP21K04311). We are deeply thankful to Prof. Dr. Reiko Uruma, the head of Safety and Health Organization, Chiba University, for her encouragement on our study.
Funding
No competing financial interests exist. Japan Society for the Promotion of Science, JP21K04311, Yoshimasa Amano
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest directly relevant to the content of this article.
Additional information
Editorial responsibility: Josef Trögl.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Masumoto, A., Amano, Y. & Machida, M. Enhancement of cyanobacterial blooms buoyancy by controlling extracellular polysaccharides content and cation concentration under light-limited condition. Int. J. Environ. Sci. Technol. 21, 1245–1254 (2024). https://doi.org/10.1007/s13762-023-05014-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-023-05014-4