Abstract
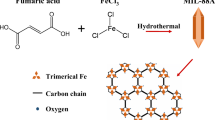
In this study, mesoporous metal organic framework (UiO-66-M) and Fe-UiO-66-M were synthesized by the thermal solvent method at 120 °C and ambient pressure. The as-synthesized Fe-UiO-66-M samples exhibit a small nanoparticle size (20–30 nm), high surface area (1040 m2/g) and medium pore diameter (5.35–5.95 nm). The TGA, EDS and XPS analysis results demonstrated the formation of missing bonding defects in Fe-UiO-66-M and UiO-66-M samples. The UiO-66-M and Fe-UiO-66-M samples were applied to adsorb As(V) over a wide pH range (pH = 1–11). Results have shown that As(V) adsorption capacity reached 337 mg/g and the removal rate of As(V) reached 94.14% at optimal pH (pH = 3). The factors which affected the As(V) adsorption process, such as initial As(V) concentration, pH, adsorption mechanism and adsorption kinetics, were also studied. To our knowledge, the As(V) adsorption capacity of Fe-UiO-66-M material was higher than that of previously reported adsorbents (5–303 mg/g). The high As(V) adsorption efficiency of Fe-UiO-66-M materials was due to the porous crystal structure, and the formation of a mesostructure with a large pore size is favorable for the diffusion process dispersion and contact of the reactants. In addition, the defect formation in the UiO-66 structure increased the electrostatic interaction and the construction of As–O–Zr and Fe–O–As bonds in the UiO-66 structure, thus promoting the As adsorption process of the synthesized material.









Similar content being viewed by others
References
Abu Tarboush BJ et al (2018) Metal-organic framework-74 for ultratrace arsenic removal from water: experimental and density functional theory studies. ACS Appl Nano Mater 1(7):3283–3292. https://doi.org/10.1021/acsanm.8b00501
Assaad N, Sabeh G, Hmadeh M (2020) Defect control in zr-based metal-organic framework nanoparticles for arsenic removal from water. ACS Appl Nano Mater 3(9):8997–9008. https://doi.org/10.1021/acsanm.0c01696
Atallah H et al (2018) A highly stable indium based metal organic framework for efficient arsenic removal from water. Dalton Trans 47(3):799–806. https://doi.org/10.1039/c7dt03705h
Berardozzi E et al (2021) ‘Comparison of arsenate adsorption from neutral ph aqueous solutions using two different iron-trimesate porous solids: kinetics, equilibrium isotherms, and synchrotron x-ray absorption experiments. J Inorg Organomet Polym Mater 31(3):1185–1194. https://doi.org/10.1007/s10904-020-01774-5
Bužek D, Demel J, Lang K (2018) Zirconium metal-organic framework uio-66: stability in an aqueous environment and its relevance for organophosphate degradation. Inorg Chem 57(22):14290–14297. https://doi.org/10.1021/acs.inorgchem.8b02360
Chang ZW, Lee YJ, Lee DJ (2019) Adsorption of hydrogen arsenate and dihydrogen arsenate ions from neutral water by UiO-66-NH2. J Environ Manage 247(June):263–268. https://doi.org/10.1016/j.jenvman.2019.06.068
Cliffe MJ et al (2014) Correlated defect nanoregions in a metal-organic framework. Nat Commun 5(May):1–8. https://doi.org/10.1038/ncomms5176
Donohue MD, Aranovich GL (1998) Classification of gibbs adsorption isotherms. Adv Coll Interface Sci 76–77:137–152. https://doi.org/10.1016/S0001-8686(98)00044-X
Dung NT et al (2021) Photocatalytic removal of Rhodamine B in water using g-C3N4/MIL-53(Fe) material under LED visible light with persulfate activation. Korean J Chem Eng 38(10):2034–2046. https://doi.org/10.1007/s11814-021-0846-9
Eltaweil AS et al (2020) Fabrication of UiO-66/MIL-101(Fe) binary MOF/carboxylated-GO composite for adsorptive removal of methylene blue dye from aqueous solutions. RSC Adv R Soc Chem 10(32):19008–19019. https://doi.org/10.1039/d0ra02424d
Feng Y et al (2019) Tailoring the properties of UiO-66 through defect engineering: a review. Ind Eng Chem Res 58(38):17646–17659. https://doi.org/10.1021/acs.iecr.9b03188
Gu Y et al (2018) (2019) ‘Facile fabrication of composition-tunable Fe/Mg bimetal-organic frameworks for exceptional arsenate removal.’ Chem Eng J 357:579–588. https://doi.org/10.1016/j.cej.2018.09.174
Ha VTT et al (2022) ‘Highly sensitive electrochemical sensor based on zirconium oxide-decorated gold nanoflakes nanocomposite 2,4-dichlorophenol detection. J Appl Electrochem 52(3):607–616. https://doi.org/10.1007/s10800-021-01655-w
Hao L et al (2018) Hierarchically porous UiO-66: Facile synthesis, characterization and application. Chem Commun 54(83):11817–11820. https://doi.org/10.1039/c8cc05895d
He X et al. (2019) Exceptional adsorption of arsenic by zirconium metal-organic frameworks: Engineering exploration and mechanism insight. J Colloid Interf Sci 539: 223–234. https://doi.org/10.1016/j.jcis.2018.12.065.
Hou S et al (2018) Green synthesis and evaluation of an iron-based metal-organic framework MIL-88B for efficient decontamination of arsenate from water. Dalton Trans 47(7):2222–2231. https://doi.org/10.1039/c7dt03775a
Hu C et al (2021) ‘Hydroxylation and sodium intercalation on g-C 3 N 4 for photocatalytic removal of gaseous formaldehyde’, Carbon. Elsevier Ltd 175:467–477. https://doi.org/10.1016/j.carbon.2021.01.112
Huo JB et al (2018) (2019) ‘Direct epitaxial synthesis of magnetic Fe3O4@UiO-66 composite for efficient removal of arsenate from water.’ Micropor Mesopor Mater Elsevier 276:68–75. https://doi.org/10.1016/j.micromeso.2018.09.017
Jian M et al. (2015) Adsorptive removal of arsenic from aqueous solution by zeolitic imidazolate framework-8 (ZIF-8) nanoparticles. Colloids Surf A: Physicochem Eng Aspects. Elsevier B.V. 465: 67–76. https://doi.org/10.1016/j.colsurfa.2014.10.023.
Jomova K et al (2011) Arsenic: Toxicity, oxidative stress and human disease. J Appl Toxicol 31(2):95–107. https://doi.org/10.1002/jat.1649
Li Z et al. (2011) Removal of arsenic from water using Fe-exchanged natural zeolite. J Hazard Mater. Elsevier B.V., 187(1–3): 318–323. https://doi.org/10.1016/j.jhazmat.2011.01.030.
Li Z et al. (2019) Adsorption behavior of arsenicals on MIL-101(Fe): The role of arsenic chemical structures. J Colloid Interf Sci. Elsevier Inc., 554: 692–704. https://doi.org/10.1016/j.jcis.2019.07.046.
Liu T et al (2019) Highly efficient and rapid removal of arsenic(iii) from aqueous solutions by nanoscale zero-valent iron supported on a zirconium 1,4-dicarboxybenzene metal-organic framework (UiO-66 MOF). RSC Adv R Soc Chem 9(67):39475–39487. https://doi.org/10.1039/c9ra08595e
Mahanta N, Chen JP (2013) A novel route to the engineering of zirconium immobilized nano-scale carbon for arsenate removal from water. J Mater Chem A 1(30):8636–8644. https://doi.org/10.1039/c3ta10858a
Miyamoto M et al (2015) In situ solvothermal growth of highly oriented Zr-based metal organic framework UiO-66 film with monocrystalline layer. CrystEngComm R Soc Chem 17(18):3422–3425. https://doi.org/10.1039/c5ce00462d
Moghaddam ZS et al. (2018) Synthesis of UiO-66-OH zirconium metal-organic framework and its application for selective extraction and trace determination of thorium in water samples by spectrophotometry. Spectrochimica Acta - Part A: Mol Biomol Spectro. Elsevier B.V. 194: 76–82. https://doi.org/10.1016/j.saa.2018.01.010.
Mohan D, Pittman CU (2007) Arsenic removal from water/wastewater using adsorbents-A critical review. J Hazard Mater 142(1–2):1–53. https://doi.org/10.1016/j.jhazmat.2007.01.006
Navarrete-Magaña M et al. (2021) Improved photocatalytic oxidation of arsenic (III) with WO3/TiO2 nanomaterials synthesized by the sol-gel method. J Environ Manage 282. https://doi.org/10.1016/j.jenvman.2020.111602.
Nguyen MB, Hong Nhung VT et al (2020a) An electrochemical sensor based on copper-based metal-organic framework-reduced graphene oxide composites for determination of 2,4-dichlorophenol in water. RSC Adv R Soc Chem 10(69):42212–42220. https://doi.org/10.1039/d0ra06700h
Nguyen MB, Le GH et al (2020b) Novel nano-Fe2O3-Co3O4Modified dolomite and its use as highly efficient catalyst in the ozonation of ammonium solution. J Nanomater. https://doi.org/10.1155/2020/4593054
Nguyen TH et al (2022) Single-step removal of arsenite ions from water through oxidation-coupled adsorption using Mn/Mg/Fe layered double hydroxide as catalyst and adsorbent. Chemosphere Elsevier Ltd 295:133370. https://doi.org/10.1016/j.chemosphere.2021.133370
Nguyen MB et al. (2021) Bimetallic Ag-Zn-BTC / GO composite as highly efficient photocatalyst in the photocatalytic degradation of reactive yellow 145 dye in water. J Hazard Mater. Elsevier B.V. 420: 126560. https://doi.org/10.1016/j.jhazmat.2021.126560.
Pang D et al. (2020) Highly efficient removal of As(V) using metal–organic framework BUC-17. SN Appl Sci. Springer International Publishing, 2(2). https://doi.org/10.1007/s42452-020-1981-3.
Shen C et al (2019) Catalytic MOF-loaded cellulose sponge for rapid degradation of chemical warfare agents simulant. Carbohyd Polym Elsevier Ltd 213:184–191. https://doi.org/10.1016/j.carbpol.2019.02.044
Somjit V et al (2022) Hydroxylation of UiO-66 metal-organic frameworks for high Arsenic(III) removal efficiency. Inorg Chem 61(29):11342–11348. https://doi.org/10.1021/acs.inorgchem.2c01513
Song L et al (2019) (2020) ‘Photothermal graphene/UiO-66-NH2 fabrics for ultrafast catalytic degradation of chemical warfare agent simulants. J Hazard Mater Elsevier 393:122332. https://doi.org/10.1016/j.jhazmat.2020.122332
Sun J et al. (2019) Preparation of Fe–Co based MOF-74 and its effective adsorption of arsenic from aqueous solution. J Environ Sci (China). Elsevier B.V., 80, pp. 197–207. https://doi.org/10.1016/j.jes.2018.12.013.
Vaiano V et al. (2014) Enhanced photocatalytic oxidation of arsenite to arsenate in water solutions by a new catalyst based on MoOx supported on TiO2. Appl Catal B: Environ. Elsevier B.V., 160–161(1), pp. 247–253. https://doi.org/10.1016/j.apcatb.2014.05.034.
Van Le D et al (2022) Synthesis of a UiO-66/g-C 3 N 4 composite using terephthalic acid obtained from waste plastic for the photocatalytic degradation of the chemical warfare agent simulant, methyl paraoxon. RSC Adv R Soc Chem 12(35):22367–22376. https://doi.org/10.1039/d2ra03483b
Vermoortele F et al (2013) Synthesis modulation as a tool to increase the catalytic activity of metal-organic frameworks: the unique case of UiO-66(Zr). J Am Chem Soc 135(31):11465–11468. https://doi.org/10.1021/ja405078u
Vo TK et al. (2021) Formation of structural defects within UiO-66(Zr)-(OH)2 framework for enhanced CO2 adsorption using a microwave-assisted continuous-flow tubular reactor. Micropor Mesopor Mater. Elsevier Inc., 312(August 2020), p. 110746. https://doi.org/10.1016/j.micromeso.2020.110746.
Vu TA et al (2015) Arsenic removal from aqueous solutions by adsorption using novel MIL-53(Fe) as a highly efficient adsorbent. RSC Adv R Soc Chem 5(7):5261–5268. https://doi.org/10.1039/c4ra12326c
Vu HT et al (2020) ‘Synthesis and application of novel nano Fe-BTC/GO composites as highly efficient photocatalysts in the dye degradation. Topics Catal Springer, US 63(11–14):1046–1055. https://doi.org/10.1007/s11244-020-01289-w
Wang C et al (2015) ‘Superior removal of arsenic from water with zirconium metal-organic framework UiO-66. Sci Rep Nat Publ Group 5:1–10. https://doi.org/10.1038/srep16613
Wang Y et al (2017) Missing-node directed synthesis of hierarchical pores on a zirconium metal-organic framework with tunable porosity and enhanced surface acidity: Via a microdroplet flow reaction. J Mater Chem A 5(42):22372–22379. https://doi.org/10.1039/c7ta06060b
Wu H et al (2013) Unusual and highly tunable missing-linker defects in zirconium metal-organic framework UiO-66 and their important effects on gas adsorption. J Am Chem Soc 135(28):10525–10532. https://doi.org/10.1021/ja404514r
Xiang W et al (2020) The metal–organic framework UiO-66 with missing-linker defects: a highly active catalyst for carbon dioxide cycloaddition. Appl Energy Elsevier 277(February):115560. https://doi.org/10.1016/j.apenergy.2020.115560
Xu C et al (2019) Turning on visible-light photocatalytic C-H oxidation over metal-organic frameworks by introducing metal-to-cluster charge transfer. J Am Chem Soc 141(48):19110–19117. https://doi.org/10.1021/jacs.9b09954
Xu R et al (2020) Hierarchically porous UiO-66 with tunable mesopores and oxygen vacancies for enhanced arsenic removal. J Mater Chem A 8(16):7870–7879. https://doi.org/10.1039/c9ta13747e
Yang Q et al (2018) Ru/UiO-66 catalyst for the reduction of nitroarenes and tandem reaction of alcohol oxidation/knoevenagel condensation. ACS Omega 3(4):4199–4212. https://doi.org/10.1021/acsomega.8b00157
Yang B et al. (2021) Preparation of a spindle δ-MnO2@Fe/Co-MOF-74 for effective adsorption of arsenic from water. Colloids Surf A: Physicochem Eng Aspects. Elsevier B.V., 629(August), p. 127378. https://doi.org/10.1016/j.colsurfa.2021.127378.
Zhang R et al (2018) α-Fe 2 O 3 nanoclusters confined into UiO-66 for efficient visible-light photodegradation performance. Appl Surf Sci Elsevier 466:956–963. https://doi.org/10.1016/j.apsusc.2018.10.048
Zhang Z-J et al (2021) Amine-functionalized UiO-66 as a fluorescent sensor for highly selective detecting volatile organic compound biomarker of lung cancer. J Solid State Chem Elsevier Ltd 305:122623. https://doi.org/10.1016/j.jssc.2021.122623
Zhang X et al. (2019) Enhanced adsorption performance of gaseous toluene on defective UiO-66 metal organic framework: Equilibrium and kinetic studies. J Hazard Mater. Elsevier B.V. 365: 597–605. https://doi.org/10.1016/j.jhazmat.2018.11.049.
Acknowledgements
This research is funded by Intiture of Chemistry-Vietnam Academy of Science and Technology (VAST) under Grant Number CSCL06.05/22-23.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The author declare that they have no conflicts of interests.
Additional information
Editorial responsibility: Mohamed F. Yassin.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hai, N.D., Nguyen, M.B., Tan, V.M. et al. Formation of structural defects within Fe-UiO-66 for effective adsorption of arsenic from water. Int. J. Environ. Sci. Technol. 20, 10075–10088 (2023). https://doi.org/10.1007/s13762-022-04712-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-022-04712-9