Abstract
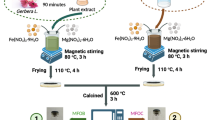
Pistachio shell powder-supported MnO2 nanostructure-based eco-friendly nanocomposite (nMPP) was prepared via one-pot redox precipitation method and was characterized by FTIR, XRD, SEM, TEM, BET, TGA/DTA, and XPS techniques. SEM and TEM analysis revealed the pseudo-spherical and nanorod morphologies of the synthesized nano-MnO2 and found agglomerated on the pistachio biomass. The nMPP contains nearly 41% Mn as MnO2 (w/w %) dispersed onto the pistachio shell biomass as confirmed from EDX, TGA, and AAS analysis. The nMPP exhibits multi-process crystal violet (CV) removal phenomenon under different pH of aqueous dye solution. Under acidic pH, nMPP caused oxidative degradation of CV by in situ formed.OH radicals; while under the neutral pH, CV undergoes monolayer adsorption onto the surface of nMPP as confirmed from Langmuir adsorption isotherm fit with maximum equilibrium adsorption value of 148.7 mg.g−1. The nMPP nanomaterial exhibits a synergistic effect between adsorption efficiencies of pistachio shell biomass and nano-MnO2 for the effective removal of toxic CV dye. The maximum saturation adsorption and rate constant (k2) value obtained from the pseudo-second-order kinetic fit model were 119.13 mg.g−1 and 5.0 × 10–4 g.mg−1 min−1, respectively.
Graphical abstract














Similar content being viewed by others
Availability of data and materials
The data used for this manuscript have been cited with proper references. All the data used to write the results are included in manuscript and can be produced by the corresponding author on reasonable request.
Change history
26 May 2022
A Correction to this paper has been published: https://doi.org/10.1007/s13762-022-04290-w
Abbreviations
- PP:
-
Pistachio shell powder
- nMPP:
-
Nanomanganese oxide on pistachio shell powder
- FTIR:
-
Fourier transform infrared
- XRD:
-
X-ray powder diffraction
- FESEM:
-
Field emission scanning electron microscopy
- EDX:
-
Energy-dispersive X-ray
- BET:
-
Brunauer–Emmett–Teller
- BJH:
-
Barrett–Joyner–Halenda
- XPS:
-
X-ray photoelectron spectroscopy
- TGA:
-
Thermogravimetric analysis
- DTA:
-
Differential thermal analysis
References
Abdelrahman EA, Hegazey RM, Kotp YH, Alharbi A (2019) Facile synthesis of Fe2O3 nanoparticles from Egyptian insecticide cans for efficient photocatalytic degradation of methylene blue and crystal violet dyes. Spectrochim Acta A Mol Biomol Spectrosc 222:117195. https://doi.org/10.1016/j.saa.2019.117195
Abdi M, Balagabri M, Karimi H, Hossini H, Rastegar SO (2020) Degradation of crystal violet (CV) from aqueous solutions using ozone, peroxone, electroperoxone, and electrolysis processes: a comparison study. Appl Water Sci 10:1–10. https://doi.org/10.1007/s13201-020-01252-w
Abdulaziz K (2021) Removal of crystal violet dye from aqueous solution with pistachio shell powder: optimization of process by Taguchi method. Erzincan Üniv Fen Bilim Enst derg 14:357–369. https://doi.org/10.18185/erzifbed.878694.
Abdullah NH, Shameli K, Abdullah EC, Abdullah LC (2020) Low cost and efficient synthesis of magnetic iron oxide/activated sericite nanocomposites for rapid removal of methylene blue and crystal violet dyes. Mater Charact 163:110275. https://doi.org/10.1016/j.matchar.2020.110275
Afroze S, Sen TK (2018) A review on heavy metal ions and dye adsorption from water by agricultural solid waste adsorbents. Water Air Soil Poll 229:1–50. https://doi.org/10.1007/s11270-018-3869-z
Ahmad W, Khan A, Ali N, Khan S, Uddin S, Malik S, Ali N, Khan H, Khan H, Bilal M (2021) Photocatalytic degradation of crystal violet dye under sunlight by chitosan-encapsulated ternary metal selenide microspheres. Environ Sci Poll Res 28:8074–8087. https://doi.org/10.1007/s11356-020-10898-7
Alharbi A, Shah RK, Sayqal A, Alshikh ZA, Algethami FK, Naglah AM, Almehizia AA, Youssef HM (2020) Facile hydrothermal synthesis of glutamine-assisted tin oxide nanorods for efficient photocatalytic degradation of crystal violet dye. Int J Environ Anal Chem. https://doi.org/10.1080/03067319.2020.1836172
Ali A, Idris R (2017) Utilization of low-cost activated carbon from rapid synthesis of microwave pyrolysis for WC nanoparticles preparation. Adv Mater Lett 8:70–76. https://doi.org/10.5185/amlett.2017.6964
Avramova I, Radoykova TH, Valchev I, Mehandjiev D (2018) X-ray photoelectron spectroscopy investigations of lignocellulosic materials. Bulg Chem Commun 50:411–416. https://doi.org/10.1080/02773819809350122
Awad AM, Jalab R, Benamor A, Nasser MS, Ba-Abbad MM, El-Naas M, Mohammad AW (2020) Adsorption of organic pollutants by nanomaterial-based adsorbents: an overview. J Mol Liq 301:112335. https://doi.org/10.1016/j.molliq.2019.112335
Bahiraei A, Behin J (2020) Sonochemical immobilization of MnO2 nanoparticles on NaP-zeolite for enhanced Hg(II) adsorption from water. J Environ Chem Eng 8:103790. https://doi.org/10.1016/j.jece.2020.103790
Banerjee M, Bar N, Basu RK, Das SK (2018) Removal of Cr (VI) from its aqueous solution using green adsorbent pistachio shell: a fixed bed column study and GA-ANN modeling. Water Conserv Sci Eng 3:19–31. https://doi.org/10.1007/s41101-017-0039-x
Banerjee M, Basu RK, Das SK (2019) Adsorptive removal of Cu (II) by pistachio shell: Isotherm study, kinetic modelling and scale-up designing-continuous mode. Environ Technol Innov 15:100419. https://doi.org/10.1016/j.eti.2019.100419
Barakat MA, Kumar R, Seliem MK, Selim AQ, Mobarak M, Anastopoulos I, Giannakoudakis D, Barczak M, Bonilla-Petriciolet A, Mohamed EA (2020) Exfoliated clay decorated with magnetic iron nanoparticles for crystal violet adsorption: modeling and physicochemical interpretation. Nanomaterials 10:1454. https://doi.org/10.3390/nano10081454
Bernard S, Chazal P, Mazet M (1997) Removal of organic compounds by adsorption on pyrolusite (β-MnO2). Water Res 31:1216–1222. https://doi.org/10.1016/S0043-1354(96)00149-2
Bordbar M, Mortazavimanesh N (2018) Biosynthesis of waste pistachio shell supported silver nanoparticles for the catalytic reduction processes. IET Nanobiotechnol 12:939–945. https://doi.org/10.1049/iet-nbt.2017.0266
Chahinez HO, Abdelkader O, Leila Y, Tran HN (2020) One-stage preparation of palm petiole-derived biochar: Characterization and application for adsorption of crystal violet dye in water. Environ Technol Innov 19:100872. https://doi.org/10.1016/j.eti.2020.100872
Chaudhary B (2020) Chemistry of synthetic dyes: a review. J Interdiscipl Cycle Res 12:390–396
Chen CC, Fan HJ, Jang CY, Jan JL, Lin HD, Lu CS (2006) Photooxidative N-de-methylation of crystal violet dye in aqueous nano-TiO2 dispersions under visible light irradiation. J Photochem Photobiol A Chem 184:147–154. https://doi.org/10.1016/j.jphotochem.2006.04.008
Chen F, Fang P, Gao Y, Liu Z, Liu Y, Dai Y (2012) Effective removal of high-chroma crystal violet over TiO2-based nanosheet by adsorption–photocatalytic degradation. Chem Eng J 204:107–113. https://doi.org/10.1016/j.cej.2012.07.030
Cheng K, Winter WT, Stipanovic AJ (2012) A modulated-TGA approach to the kinetics of lignocellulosic biomass pyrolysis/combustion. Polym Degrad Stab 97:1606–1615. https://doi.org/10.1016/j.polymdegradstab.2012.06.027
Dai Y, Wang X, Dai Q, Li D (2012) Effect of Ce and La on the structure and activity of MnOx catalyst in catalytic combustion of chlorobenzene. Appl Catal B Environ 111:141–149. https://doi.org/10.1016/j.apcatb.2011.09.028
Demissie H, An G, Jiao R, Ritigala T, Lu S, Wang D (2021) Modification of high content nanocluster-based coagulation for rapid removal of dye from water and the mechanism. Sep Purif Technol 259:117845. https://doi.org/10.1016/j.seppur.2020.117845
Dey S, Kumar VP (2020) The performance of highly active manganese oxide catalysts for ambient conditions carbon monoxide oxidation. Curr Opin Green Sustain Chem 3:100012. https://doi.org/10.1016/j.crgsc.2020.100012
Dhanalakshmi M, Prabavathi SL, Saravanakumar K, Jones BF, Muthuraj V (2020) Iridium nanoparticles anchored WO3 nanocubes as an efficient photocatalyst for removal of refractory contaminants (crystal violet and methylene blue). Chem Phys Lett 745:137285. https://doi.org/10.1016/j.cplett.2020.137285
Diao H, Zhang Z, Liu Y, Song Z, Zhou L, Duan Y, Zhang J (2020) Facile fabrication of carboxylated cellulose nanocrystal–MnO2 beads for high-efficiency removal of methylene blue. Cellulose 27:7053–7066. https://doi.org/10.1007/s10570-020-03260-0
El-Azazy M, El-Shafie AS, Ashraf A, Issa AA (2019) Eco-structured biosorptive removal of basic fuchsin using pistachio nutshells: A definitive screening design-Based approach. Appl Sci 9:4855. https://doi.org/10.3390/app9224855
Elella MH, Sabaa MW, Abd ElHafeez E, Mohamed RR (2019) Crystal violet dye removal using crosslinked grafted xanthan gum. Int J Biol Macromol 137:1086–1101. https://doi.org/10.1016/j.ijbiomac.2019.06.243
Elmaci G, Ozgenc G, Atakay M, Salih B, Zumreoglu-Karan B (2017) Degradation of crystal violet dye from waters by layered MnO2 and nanocomposite-MnO2@MnFe2O4 catalysts. Hacettepe J Biol Chem 4:573–580. https://doi.org/10.15671/HJBC.2018.199
Gabal MA, Al-Harthy EA, Al Angari YM, Salam MA (2014) MWCNTs decorated with Mn0.8Zn0.2Fe2O4 nanoparticles for removal of crystal-violet dye from aqueous solutions. Chem Eng J 255:156–164. https://doi.org/10.1016/j.cej.2014.06.019
Garces Goncalves Jr PR, De Abreu HA, Duarte HA (2018) Stability, structural, and electronic properties of Hausmannite (Mn3O4) surfaces and their interaction with water. J Phys Chem C 122:20841–20849. https://doi.org/10.1021/acs.jpcc.8b06201
Gök O, Mesutoğlu ÖC (2016) Application of pistachio shell (PSS) as low-cost adsorbent for the removal of Pb(II) from aqueous solution. Int J Ecosyst Ecol Sci (IJEES) 6:389–394
Guerra E, Llompart M, Garcia-Jares C (2018) Analysis of dyes in cosmetics: challenges and recent developments. Cosmetics 5:47. https://doi.org/10.3390/cosmetics5030047
Heidari AA, Mahdavi H (2021) Polyethylene coated with MnO2 nanoparticles as thin film composite membranes for organic solvent nanofiltration. ACS Appl Nano Mater 4:2768–2782. https://doi.org/10.1021/acsanm.0c03409
Husnain SM, Asim U, Yaqub A, Shahzad F, Abbas N (2020) Recent trends of MnO2-derived adsorbents for water treatment: a review. New J Chem 44:6096–6120. https://doi.org/10.1039/C9NJ06392G
Igwegbe CA, Ighalo JO, Ghosh S, Ahmadi S, Ugonabo VI (2021) Pistachio (Pistacia vera) waste as adsorbent for wastewater treatment: a review. Biomass Convers Biorefin. https://doi.org/10.1007/s13399-021-01739-9
Jamal R, Zhang L, Wang M, Zhao Q, Abdiryim T (2016) Synthesis of poly(3,4-propylenedioxythiophene)/MnO2 composites and their applications in the adsorptive removal of methylene blue. Prog Nat Sci Mater Int 26:32–40. https://doi.org/10.1016/j.pnsc.2016.01.001
Jeong JM, Park SH, Park HJ, Jin SB, Son SG, Moon JM, Suh H, Choi BG (2021) Alternative ultrathin assembling of exfoliated manganese dioxide and nitrogen doped carbon layers for high mass loading supercapacitors with outstanding capacitance and impressive rate capability. Adv Funct Mater 31:2009632. https://doi.org/10.1002/adfm.202009632
Jiani L, Zhicheng X, Hao X, Dan Q, Zhengwei L, Wei Y, Yu W (2020) Pulsed electrochemical oxidation of acid Red G and crystal violet by PbO2 anode. J Environ Chem Eng 8:103773. https://doi.org/10.1016/j.jece.2020.103773
Kazayawoko M, Balatinecz JJ, Woodhams RT, Sodhi RN (1998) X-ray photoelectron spectroscopy of lignocellulosic materials treated with maleated polypropylenes. J Wood Chem Technol 18:1–26. https://doi.org/10.1080/02773819809350122
Kumar A, Singh R, Upadhyay SK, Kumar S, Charaya MU (2021a) Biosorption: the removal of toxic dyes from industrial effluent using phytobiomass-a review. Plant Archives 21:1320–1325. https://doi.org/10.51470/PLANTARCHIVES.2021a.v21.S1.207
Kumar S, Brar RS, Babu JN, Dahiya A, Saha S, Kumar A (2021b) Synergistic effect of pistachio shell powder and nano-zerovalent copper for chromium remediation from aqueous solution. Environ Sci Poll Res 28:63422–63436. https://doi.org/10.1007/s11356-021-15285-4
Kyi PP, Quansah JO, Lee CG, Moon JK, Park SJ (2020) The removal of crystal violet from textile wastewater using palm kernel shell-derived biochar. Appl Sci 10:2251. https://doi.org/10.3390/app10072251
Li X, Zhang L, Yang Z, Wang P, Yan Y, Ran J (2020) Adsorption materials for volatile organic compounds (VOCs) and the key factors for VOCs adsorption process: a review. Sep Purif Technol 235:116213. https://doi.org/10.1016/j.seppur.2019.116213
Liang Z, Zhao Z, Sun T, Shi W, Cui F (2017) Enhanced adsorption of the cationic dyes in the spherical CuO/meso-silica nano composite and impact of solution chemistry. J Colloid Interface Sci 485:192–200. https://doi.org/10.1016/j.jcis.2016.09.028
Lin J, Xue C, Guo S, Owens G, Chen Z (2021) Effects of green synthesized and commercial nZVI on crystal violet degradation by Burkholderia vietnamiensis C09V: dose-dependent toxicity and biocompatibility. Chemosphere 279:130612. https://doi.org/10.1016/j.chemosphere.2021.130612
Ma J, Wang C, Xi W, Zhao Q, Wang S, Qiu M, Wang J, Wang X (2021) Removal of radionuclides from aqueous solution by manganese dioxide-based nanomaterials and mechanism research: a review. ACS ES&T Eng 1:685–705. https://doi.org/10.1021/acsestengg.0c00268
Mahamallik P, Saha S, Pal A (2015) Tetracycline degradation in aquatic environment by highly porous MnO2 nanosheet assembly. Chem Engin J 276:155–165. https://doi.org/10.1016/j.cej.2015.04.064
Mani S, Bharagava RN (2016) Exposure to crystal violet, its toxic, genotoxic and carcinogenic effects on environment and its degradation and detoxification for environmental safety. Rev Environ Contam Toxicol 237:71–104. https://doi.org/10.1007/978-3-319-23573-8_4
Manickam M, Singh P, Issa TB, Thurgate S, De Marco R (2004) Lithium insertion into manganese dioxide electrode in MnO2/Zn aqueous battery: Part I. A Preliminary Study. J Power Sour 130:254–259. https://doi.org/10.1016/j.jpowsour.2004.06.049
Mansor ES, Abdallah H, Shaban AM (2020) Fabrication of high selectivity blend membranes based on poly vinyl alcohol for crystal violet dye removal. J Environ Chem Eng 8:103706. https://doi.org/10.1016/j.jece.2020.103706
Mittal H, Al Alili A, Morajkar PP, Alhassan SM (2021) Graphene oxide crosslinked hydrogel nanocomposites of xanthan gum for the adsorption of crystal violet dye. J Mol Liq 323:115034. https://doi.org/10.1016/j.molliq.2020.115034
Miyah Y, Lahrichi A, Idrissi M, Anis K, Kachkoul R, Idrissi N, Lairini S, Nenov V, Zerrouq F (2017) Removal of cationic dye “crystal violet” in aqueous solution by the local clay. J Mater Environ Sci 8:3570–3582
Mohammed AA, Al-Musawi TJ, Kareem SL, Zarrabi M, Al-Ma’abreh AM (2020) Simultaneous adsorption of tetracycline, amoxicillin, and ciprofloxacin by pistachio shell powder coated with zinc oxide nanoparticles. Arab J Chem 13:4629–4643. https://doi.org/10.1016/j.arabjc.2019.10.010
Mohanty S, Moulick S, Maji SK (2020) Adsorption/photodegradation of crystal violet (basic dye) from aqueous solution by hydrothermally synthesized titanate nanotube (TNT). J Water Process Eng 37:101428. https://doi.org/10.1016/j.jwpe.2020.101428
Mori K, Iguchi S, Takebe S, Nakayama M (2015) A thin film sorbent of layered organo-MnO2 for the extraction of p-aminoazobenzene from aqueous solution. J Mater Chem A 3:6470–6476. https://doi.org/10.1039/C4TA06725H
Naseem T, Baig MM, Warsi MF, Hussain R, Agboola PO, Waseem M (2020) Mesoporous silica prepared via a green route: A comparative study for the removal of crystal violet from wastewater. Mater Res Express 8:015005. https://doi.org/10.1088/2053-1591/abd45d
Nayak S, Prasad SR, Mandal D, Das P (2020) Carbon dot cross-linked polyvinylpyrrolidone hybrid hydrogel for simultaneous dye adsorption, photodegradation and bacterial elimination from waste water. J Hazard Mater 392:122287. https://doi.org/10.1016/j.jhazmat.2020.122287
Nichols G, Byard S, Bloxham MJ, Botterill J, Dawson NJ, Dennis A, Diart V, North NC, Sherwood JD (2002) A review of the terms agglomerate and aggregate with a recommendation for nomenclature used in powder and particle characterization. J Pharm Sci 91:2103–2109. https://doi.org/10.1002/jps.10191
Osmari TA, Gallon R, Schwaab M, Barbosa-Coutinho E, Severo JB Jr, Pinto JC (2013) Statistical analysis of linear and non-linear regression for the estimation of adsorption isotherm parameters. Adsorp Sci Technol 31:433–458. https://doi.org/10.1260%2F0263-6174.31.5.433
Ozulumba TJ (2016) A comparative study of the adsorption of crystal violet and malachite green from synthetic waste water by peanut and coconut husk powders (Doctoral dissertation).
Panimalar S, Uthrakumar R, Selvi ET, Gomathy P, Inmozhi C, Kaviyarasu K, Kennedy J (2020) Studies of MnO2/g-C3N4 hetrostructure efficient of visible light photocatalyst for pollutants degradation by sol-gel technique. Surf Interfaces 20:100512. https://doi.org/10.1016/j.surfin.2020.100512
Popa N, Visa M, Patachia S (2020) Influence of the Cd2+ Ions on the crystal violet dye adsorption from aqueous solutions by activated charcoal obtained from pine cones. Solar Energy Convers Commun. https://doi.org/10.1007/978-3-030-55757-7_23
Puneetha J, Kottam N, Rathna A (2021) Investigation of photocatalytic degradation of crystal violet and its correlation with bandgap in ZnO and ZnO/GO nanohybrid. Inorg Chem Commun 125:108460. https://doi.org/10.1016/j.inoche.2021.108460
Putri KN, Keereerak A, Chinpa W (2020) Novel cellulose-based biosorbent from lemongrass leaf combined with cellulose acetate for adsorption of crystal violet. Int J Biol Macromol 156:762–772. https://doi.org/10.1016/j.ijbiomac.2020.04.100
Rahdar S, Shikh L, Ahmadi S (2018) Removal of reactive blue 19 dye using a combined sonochemical and modified pistachio shell adsorption processes from aqueous solutions. Iranian J Health Sci 6:8–20. https://doi.org/10.18502/jhs.v6i3.193
Rahmat M, Rehman A, Rahmat S, Bhatti HN, Iqbal M, Khan WS, Bajwa SZ, Rahmat R, Nazir A (2019) Highly efficient removal of crystal violet dye from water by MnO2 based nanofibrous mesh/photocatalytic process. J Mater Res Technol 8:5149–5159. https://doi.org/10.1016/j.jmrt.2019.08.038
Rani A, Reddy R, Sharma U, Mukherjee P, Mishra P, Kuila A, Sim LC, Saravanan P (2018) A review on the progress of nanostructure materials for energy harnessing and environmental remediation. J Nanostruct Chem 8:255–291. https://doi.org/10.1007/s40097-018-0278-1
Remucal CK, Ginder-Vogel M (2014) A critical review of the reactivity of manganese oxides with organic contaminants. Environ Sci Process Impacts 16:1247–1266. https://doi.org/10.1039/C3EM00703K
Roy DC, Biswas SK, Saha AK, Sikdar B, Rahman M, Roy AK, Prodhan ZH, Tang SS (2018) Biodegradation of crystal violet dye by bacteria isolated from textile industry effluents. PeerJ 6:e5015. https://doi.org/10.7717/peerj.5015
Sacco O, Matarangolo M, Vaiano V, Libralato G, Guida M, Lofrano G, Carotenuto M (2018) Crystal violet and toxicity removal by adsorption and simultaneous photocatalysis in a continuous flow micro-reactor. Sci Total Environ 644:430–438. https://doi.org/10.1016/j.scitotenv.2018.06.388
Saha S, Pal A (2014) Microporous assembly of MnO2 nanosheets for malachite green degradation. Sep Purif Technol 134:26–36. https://doi.org/10.1016/j.seppur.2014.07.021
Satapathy MK, Das P (2014) Optimization of crystal violet dye removal using novel soil-silver nanocomposite as nanoadsorbent using response surface methodology. J Environ Chem Eng 2:708–714. https://doi.org/10.1016/j.jece.2013.11.012
Şentürk İ, Alzein M (2020) Adsorptive removal of basic blue 41 using pistachio shell adsorbent-performance in batch and column system. Sustain Chem Pharm 16:100254. https://doi.org/10.1016/j.scp.2020.100254
Shafizadeh F (1982) Introduction to pyrolysis of biomass. J Anal Appl Pyrolysis 3:283–305. https://doi.org/10.1016/0165-2370(82)80017-X
Shaker KS, AbdAlsalm AH (2018) Synthesis and characterization nano structure of MnO2 via chemical method. Eng Technol J. https://doi.org/10.30684/etj.36.9A.1
Sharma S, Kaur A (2018) Various methods for removal of dyes from industrial effluents-a review. Indian J Sci Technol 11:1–21. https://doi.org/10.17485/ijst/2018/v11i12/120847
Singh KP, Gupta S, Singh AK, Sinha S (2011) Optimizing adsorption of crystal violet dye from water by magnetic nanocomposite using response surface modeling approach. J Hazard Mater 186:1462–1473. https://doi.org/10.1016/j.jhazmat.2010.12.032
Singh M, Bharadwaj K, Dey ES, Dicko C (2020) Sonication enhances the stability of MnO2 nanoparticles on silk film template for enzyme mimic application. Ultrason Sonochem 64:105011. https://doi.org/10.1016/j.ultsonch.2020.105011
Sohal N, Maity B, Shetti NP, Basu S (2021) Biosensors based on MnO2 nanostructures: a review. ACS Appl Nano Mater 4:2285–2302. https://doi.org/10.1021/acsanm.0c03380
Stone AT, Morgan JJ (1984) Reduction and dissolution of manganese(III) and manganese(IV) oxides by organics. 1. Reaction with Hydroquinone Environ Sci Technol 18:450–456. https://doi.org/10.1021/es00124a011
Taghizadeh A, Rad-Moghadam K (2018) Green fabrication of Cu/pistachio shell nanocomposite using Pistacia Vera L. hull: An efficient catalyst for expedient reduction of 4-nitrophenol and organic dyes. J Clean Prod 198:1105–1119. https://doi.org/10.1016/j.jclepro.2018.07.042
Tang Y, Zheng S, Cao S, Xue H, Pang H (2020) Advances in the application of manganese dioxide and its composites as electrocatalysts for the oxygen evolution reaction. J Mater Chem A 8:18492–18514. https://doi.org/10.1039/D0TA05985D
Tkaczyk A, Mitrowska K, Posyniak A (2020) Synthetic organic dyes as contaminants of the aquatic environment and their implications for ecosystems: a review. Sci Total Environ 717:137222. https://doi.org/10.1016/j.scitotenv.2020.137222
Toupin M, Brousse T, Bélanger D (2002) Influence of microstucture on the charge storage properties of chemically synthesized manganese dioxide. Chem Mater 14:3946–3952. https://doi.org/10.1021/cm020408q
Turan NG, Mesci B (2011) Use of pistachio shells as an adsorbent for the removal of zinc(II) ion. Clean-Soil Air Water 39:475–481. https://doi.org/10.1002/clen.201000297
Uddin MK, Mashkoor F, AlArifi IM, Nasar A (2021) Simple one-step synthesis process of novel MoS2@bentonite magnetic nanocomposite for efficient adsorption of crystal violet from aqueous solution. Mater Res Bull 139:111279. https://doi.org/10.1016/j.materresbull.2021.111279
Ullah R, Liu C, Panezai H, Gul A, Sun J, Wu X (2020) Controlled crystal phase and particle size of loaded-TiO2 using clinoptilolite as support via hydrothermal method for degradation of crystal violet dye in aqueous solution. Arab J Chem 13:4092–4101. https://doi.org/10.1016/j.arabjc.2019.06.011
Vyavahare G, Jadhav P, Jadhav J, Patil R, Aware C, Patil D, Gophane A, Yang YH, Gurav R (2019) Strategies for crystal violet dye sorption on biochar derived from mango leaves and evaluation of residual dye toxicity. J Clean Prod 207:296–305. https://doi.org/10.1016/j.jclepro.2018.09.193
Wei W, Guo K, Kang X, Zhang J, Li C, Fang J (2021) Complete removal of organoarsenic by the UV/permanganate process via HO• oxidation and in situ-formed manganese dioxide adsorption. ACS ES&T Eng 1:794–803. https://doi.org/10.1021/acsestengg.1c00004
Wei Y, Huang Y, Yu Y, Gao R, Yu W (2018) The surface chemical constituent analysis of poplar fibrosis veneers during heat treatment. J Wood Sci 64:485–500. https://doi.org/10.1007/s10086-018-1732-x
Wu SJ, Liou TH, Mi FL (2009) Synthesis of zero-valent copper-chitosan nanocomposites and their application for treatment of hexavalent chromium. Bioresour Technol 100:4348–4353. https://doi.org/10.1016/j.biortech.2009.04.013
Wu YH, Ma YL, Sun YG, Xue K, Ma QL, Ma T, Ji WX (2020) Graded synthesis of highly ordered MCM-41 and carbon/zeolite composite from coal gasification fine residue for crystal violet removal. J Clean Prod 277:123186. https://doi.org/10.1016/j.jclepro.2020.123186
Zhang YX, Guo XL, Huang M, Hao XD, Yuan Y, Hua C (2015) Engineering birnessite-type MnO2 nanosheets on fiberglass for pH-dependent degradation of methylene blue. J Phys Chem Solids 83:40–46. https://doi.org/10.1016/j.jpcs.2015.03.015
Yang J, Ao Z, Wu H, Zhang S (2021a) Immobilization of chitosan-templated MnO2 nanoparticles onto filter paper by redox method as a retrievable Fenton-like dip catalyst. Chemosphere 268:128835. https://doi.org/10.1016/j.chemosphere.2020.128835
Yang R, Fan Y, Ye R, Tang Y, Cao X, Yin Z, Zeng Z (2021b) MnO2 based materials for environmental applications. Adv Mater 33:2004862. https://doi.org/10.1002/adma.202004862
Yeganeh MM, Kaghazchi T, Soleimani M (2006) Effect of raw materials on properties of activated carbons. Chem Eng Technol 29:1247–1251. https://doi.org/10.1002/ceat.200500298
Yin B, Zhang S, Jiao Y, Liu Y, Qu F, Wu X (2014) Facile synthesis of ultralong MnO2 nanowires as high performance supercapacitor electrodes and photocatalysts with enhanced photocatalytic activities. CrystEngComm 16:9999–10005. https://doi.org/10.1039/C4CE01302F
Yuan X, Zhou F, Man R, Huang J (2019) Dendritic post-cross-linked resin for the adsorption of crystal violet from aqueous solution. J Chem Thermodyn 130:235–242. https://doi.org/10.1016/j.jct.2018.09.030
Yue X, Zhao J, Shi H, Chi Y, Salam M (2020) Preparation of composite adsorbents of activated carbon supported MgO/MnO2 and adsorption of Rhodamine B. Water Sci Technol 81:906–914. https://doi.org/10.2166/wst.2020.172
Yusuf M, Song K, Geng S, Fazhi X (2020) Adsorptive removal of anionic dyes by graphene impregnated with MnO2 from aqueous solution. Colloids Surf A Physicochem Eng Asp 595:124667. https://doi.org/10.1016/j.colsurfa.2020.124667
Zhao C, Chen W (2019) A review for tannery wastewater treatment: some thoughts under stricter discharge requirements. Environ Sci Poll Res 26:26102–26111. https://doi.org/10.1007/s11356-019-05699-6
Zhou L, Huang Y, Qiu W, Sun Z, Liu Z, Song Z (2017) Adsorption properties of nano-MnO2-biochar composites for copper in aqueous solution. Molecules 22:173. https://doi.org/10.3390/molecules22010173
Acknowledgements
The authors would like to thanks the Akal University for providing research facilities and Central Instrumentation Laboratory, Central University of Punjab, for providing the physicochemical depiction facility for the characterization of materials.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
All authors contributed equally in preparation of this manuscript. Sandeep Kumar. This is corresponding author of the manuscript. He contributed in this manuscript via designing/supervising the work, data interpretation, and writing the manuscript with major input from all co-authors. Ravinderdeep Singh Brar. Author is a Ph.D. scholar and contributed with nanocomposite synthesis, and experimental data collection for the manuscript. J Nagendra Babu. Author contributed via supervision of work, data interpretation, and providing critical feedback for writing this manuscript. Amarjeet Dahiya. Author is research scholar and assists in experimental data collection for the manuscript. Sandip Saha. Author contributed by data interpretation, figures design, and helped in research writings. Kalpana. Author contributed by acquiring and interpretation of characterization data.
Corresponding author
Ethics declarations
Competing interests
The authors have no competing interests.
Consent for publication
Authors provide their consent for the publication of this manuscript.
Ethics approval and consent to participate
Not applicable.
Additional information
Editorial responsibility: Senthil Kumar Ponnusamy.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kumar, S., Brar, R.S., Saha, S. et al. Synergistic effect of eco-friendly pistachio shell biomass on nano-MnO2 for crystal violet removal: kinetic and equilibrium studies. Int. J. Environ. Sci. Technol. 20, 5123–5140 (2023). https://doi.org/10.1007/s13762-022-04212-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-022-04212-w