Abstract
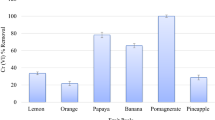
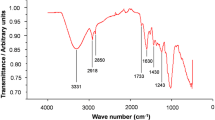
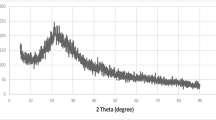
Removal of Eu(III) from aqueous solutions was studied using pomegranate peel in raw and modified form as potential biosorbent. Low-cost chemical modifications of the material using acidic and alkaline reagents under moderate conditions were applied to improve its sorption ability. The sorption study was performed in the metal concentration range Cinit:5–300 mg L−1, regarding the effect of sorbent dosage, competitive ions, temperature and contact time. It was revealed that the biosorbent modified by alkaline reagent, exhibited considerable sorption capacity with qmax value 83.3 mg g−1 for europium. Langmuir and Freundlich linear isotherm equations were satisfactorily applied as well as simulation of the kinetics data at various temperatures using pseudo-first- and pseudo-second-order equations and thermodynamic data in order to explore the sorption mechanism. Meanwhile, characterization of the biosorbents before and after sorption was performed by SEM–EDS, XRD and FTIR while chemical modification of the biosorbents surface functional groups, revealed the involvement of carboxyl, hydroxyl and phosphate groups in biosorption. Desorption experiments demonstrated the environmental compatibility of the tested materials which could be used as potential sorbents in environmental technology.









Similar content being viewed by others
References
Anagnostopoulos VA, Symeopoulos BD (2013) Sorption of europium by malt spent rootlets, a low cost biosorbent: effect of pH, kinetics and equilibrium studies. J Radioanal Nucl Chem 295:7–13. https://doi.org/10.1007/s10967-012-1956-y
Anastopoulos I, Massas I, Ehaliotis C (2015) Use of residues and by-products of the olive-oil production chain for the removal of pollutants from environmental media: a review of batch biosorption approaches. J Environ Sci Heal Part A Toxic Hazardous Subst Environ Eng 50:677–718. https://doi.org/10.1080/10934529.2015.1011964
Atkins A, Overton T, Rourke J, Weller M, Armstrong F (2006) Shriver & atkins inorganic chemistry, 4th edn. Oxford University Press, Oxford
Attia LA, Youssef MA, Abdel Moamen OA (2021) Feasibility of radioactive cesium and europium sorption using valorized punica granatum peel: kinetic and equilibrium aspects. Sep Sci Technol 56:217–232. https://doi.org/10.1080/01496395.2019.1708111
Bampaiti A, Yusan S, Aytas S, Pavlidou E, Noli F (2016) Investigation of uranium biosorption from aqueous solutions by Dictyopteris polypodioides brown algae. J Radioanal Nucl Chem 307:1335–1343. https://doi.org/10.1007/s10967-015-4289-9
Ben-Ali S, Jaouali I, Souissi-Najar S, Ouederni A (2017) Characterization and adsorption capacity of raw pomegranate peel biosorbent for copper removal. J Clean Prod 142:3809–3821. https://doi.org/10.1016/j.jclepro.2016.10.081
Bhatnagar A, Minocha AK (2010) Biosorption optimization of nickel removal from water using Punica granatum peel waste. Colloids Surf B 76:544–548. https://doi.org/10.1016/j.colsurfb.2009.12.016
Chubar N, Carvalho JR, Neiva Correia MJ (2004) Heavy metals biosorption on cork biomass: effect of the pre-treatment. Colloids Surf A Physicochem Eng Asp 238:51–58. https://doi.org/10.1016/j.colsurfa.2004.01.039
Diniz V, Volesky B (2005) Biosorption of La, Eu and Ybusing Sargassum biomass. Water Res 39:239–247
El-Ashtoukhy ESZ, Amin NK, Abdelwahab O (2008) Removal of lead (II) and copper (II) from aqueous solution using pomegranate peel as a new adsorbent. Desalination 223:162–173. https://doi.org/10.1016/j.desal.2007.01.206
Elsayed AE, Osman DI, Attia SK, Ahmed HM, Shoukry EM, Mostafa YM, Taman AR (2020) A study on the removal characteristics of organic and inorganic pollutants from wastewater by low cost biosorbent. Egypt J Chem 63(4):16–21. https://doi.org/10.21608/ejchem.2019.15710.1950
EPA (2021) SW-846 Test method 1311: toxicity characteristic leaching procedure, Accessed: Jul. 26, 2021. [Online]. Available: https://www.epa.gov/hw-sw846/sw-846-test-method-1311-toxicity-characteristic-leaching-procedure.
Femina Carolin C, Senthil Kumar P, Saravanan A, Janet Joshiba G, Naushad Mu (2017) Efficient techniques for the removal of toxic heavy metals from aquatic environment: a review. J Environ Chem Eng 5(3):2782–2799
Freitas GR, Silva MGC, Vieira MGA (2019) Biosorption technology for removal of toxic metals: a review of commercial biosorbents and patents. Environ Sci Pollut Res 26:19097–19118. https://doi.org/10.1007/s11356-019-05330-8
Freundlich HMF (1906) Over the adsorption in solution. J Phy Chem 57:385–470
Ho YS, Mckay G, Hong T, Bay W, Kong H, Hong T (2000) Separation & purification reviews kinetics of pollutant sorption by biosorbents: review. Sep Purif Rev 29(2):189–232
Kapashi E, Kapnisti M, Dafnomili A, Noli F (2019) Aloe Vera as an effective biosorbent for the removal of thorium and barium from aqueous solutions. J Radioanal Nucl Chem 321:217–226. https://doi.org/10.1007/s10967-019-06558-x
Kapnisti MG, Noli FG, Papastergiadis ES, Pavlidou EG (2018) Exploration of the parameters affecting the europium removal from aqueous solutions by novel synthesized titanium phosphates. J Environ Chem Eng 6:3408–3417. https://doi.org/10.1016/j.jece.2018.05.010
Langmuir I (1917) The constitution and fundamental properties of solids and liquids. Part II.-liquids. J Franklin Inst 184:721. https://doi.org/10.1016/s0016-0032(17)90088-2
Lagergren SK (1898) About the theory of so-called adsorption of soluble substances. Sven Vetenskapsakad Hingarl 24:1–39. Available: https://ci.nii.ac.jp/naid/10016440244. Accessed 26 Apr 2021 [Online]
Liu Y, Liu YJ (2008) Biosorption isotherms, kinetics and thermodynamics. Sep Purif Technol 61:229–242. https://doi.org/10.1016/j.seppur.2007.10.002
Mahmood-Ul-Hassan M, Suthar V, Ahmad R, Yousra M (2018) Biosorption of metal ions on lignocellulosic materials:batch and continuous-flow process studies. Environ Monit Assess. https://doi.org/10.1007/s10661-018-6674-7
Manobala T, Shukla SK, Rao TS, Kumar MD (2021) Kinetic modelling of the uranium biosorption by deinococcus radiodurans biofilm. Chemosphere 269:128722. https://doi.org/10.1016/j.chemosphere.2020.128722
Min SH, Han JS, Shin EW, Park JK (2004) Improvement of cadmium ion removal by base treatment of juniper fiber. Water Res 38:1289–1295. https://doi.org/10.1016/j.watres.2003.11.016
Nemr NA (2009) Potential of pomegranate husk carbon for Cr(VI) removal from wastewater: kinetic and isotherm studies. J Hazard Mater 161:132–141. https://doi.org/10.1016/j.jhazmat.2008.03.093
Nharingo T, Moyo M (2016) Application of Opuntia ficus-indica in bioremediation of wastewaters. A critical review. J Environ Manage 166:55–72. https://doi.org/10.1016/j.jenvman.2015.10.005
Noli F, Kapnisti M, Buema G, Harja M (2016) Retention of barium and europium radionuclides from aqueous solutions on ash-based sorbents by application of radiochemical techniques. Appl Radiat Isot 116:102–109. https://doi.org/10.1016/j.apradiso.2016.07.012
Noli F, Kapashi E, Kapnisti M (2019) Biosorption of uranium and cadmium using sorbents based on Aloe vera wastes. J Environ Chem Eng. https://doi.org/10.1016/j.jece.2019.102985
Panda GC, Das SK, Chatterjee S, Maity PB, Bandopadhyay TS, Guha AK (2006) Adsorption of cadmium on husk of Lathyrus sativus: physico-chemical study. Colloids Surf B 50:49–54. https://doi.org/10.1016/j.colsurfb.2006.03.022
Philip L, Iyengar L, Venkobachar C (2000) Biosorption of U, La, Pr, Nd, Eu and Dy by Pseudomonas aeruginosa. J Ind Microbiol Biotechnol 25:1–7
Prodromou M, Pashalidis I (2016) Europium adsorption by non-treated and chemically modified Opuntia ficus indica cactus fibres in aqueous solutions. Desalin Water Treat 57:5079–5088. https://doi.org/10.1080/19443994.2014.1002431
Puigdomenech I (2006) HYDRA (hydrochemical equilibrium-constant database) medusa (make equilibrium diagrams using sophisticated algorithms) programs. Royal Institute of Technology, Stockholm
Rafiaee S, Samani MR, Toghraie D (2020) Removal of hexavalent chromium from aqueous media using pomegranate peels modified by polymeric coatings: effects of various composite synthesis parameters. Synth Met. https://doi.org/10.1016/j.synthmet.2020.116416
Runde W (2000) The chemical interactions of actinides in the environment. Los Alamos Science Number 26. https://fas.org/sgp/othergov/doe/lanl/pubs/00818040.pdf
Sayed SA, Rabie KA, Salama IE (2005) Studies on europium separation from a middle rare earth concentrate by in situ zinc reduction technique. Sep Purif Technol 46(3):145–154. https://doi.org/10.1016/j.seppur.2005.05.006
Schwantes D, Gonçalves AC, Campagnolo MA, Tarley CRT, Dragunski DC, de Varennes A, dos Santos Silva AK, Conradi E (2018) Chemical modifications on pinus bark for adsorption of toxic metals. J Environ Chem Eng 6:1271–1278. https://doi.org/10.1016/j.jece.2018.01.044
Shakoor MB, Niazi NK, Bibi I, Shahid M, Sharif F, Bashir S, Shaheen SM, Wang H, Tsang DCW, Ok YS, Rinklebe J (2018) Arsenic removal by natural and chemically modified water melon rind in aqueous solutions and groundwater. Sci Total Environ 645:1444–1455. https://doi.org/10.1016/j.scitotenv.2018.07.218
Tai P, Zhao Q, Su D, Li P, Stagnitti F (2010) Biological toxicity of lanthanide elements on algae. Chemosphere 80:1031–1035. https://doi.org/10.1016/j.chemosphere.2010.05.030
Vijayaraghavan K, Balasubramanian R (2010) Single and binary biosorption of cerium and europium onto crab shell particles. Chem Eng J 163:337–343. https://doi.org/10.1016/j.cej.2010.08.012
Vijayaraghavan K, Sathishkumar M, Balasubramanian R (2010) Biosorption of lanthanum, cerium, europium, and ytterbium by a brown marine alga turbinaria conoides. Ind Eng Chem Res 49(9):4405–4411
Acknowledgements
M.S.A. Busari Nasiru wants to thank the staff of the radiochemichal laboratory of the Chemistry Department-AUTh for supporting his research during the ERASMUS mobility program.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
FN contributed to the study conception and design as well as to the writing of the manuscript. Materials preparation, experiments, data collection and analysis were performed by MSABN and PT. EP contributed to the materials characterization (SEM–EDS and XRD techniques). All the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Editorial responsibility: Samareh Mirkia.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Noli, F., Busari Nasiru, M.S.A., Tsamos, P. et al. Eu(III) removal from aqueous solutions using raw and modified pomegranate peel as biosorbents. Int. J. Environ. Sci. Technol. 20, 3091–3100 (2023). https://doi.org/10.1007/s13762-022-04207-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-022-04207-7