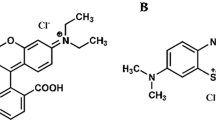
Abstract
In this research, two biocomposite materials, namely Amberlite IRA-400 Cl resin/A. campestris and Amberlite 4200 Cl resin/A. campestris, were used to perform the biosorption of Brilliant Blue G from aqueous solution. The biosorption of dye was evaluated by using a batch system. In addition, the effect of some physicochemical and kinetic parameters of the medium such as pH, temperature, contact time and initial concentration was determined. In this research, pH (3–11), adsorbent dosage (0.3 g), initial dye concentration (10–150 mg L−1), temperature (298–318 K) and contact time (10–95 min) were investigated. In the 95th minute, biosorption reaches a maximum of 137.18 mg/g at the highest temperature (318 K). Brilliant Blue G biosorption increased for both materials and at pH 11 values reached 149 mg/g on IRA-400 Cl resin/A. campestris and 142 mg/g on IRA-4200 Cl resin/A. campestris. The characterization of adsorbents was done by scanning electron microscopy, element detection X-ray and Fourier transform infrared spectroscopy. The adsorption isotherms were examined for the biosorption process. The Harkins–Jura isotherm shows better fit than the other isotherms for IRA-4200 Cl resin/A. campestris, and the Freundlich isotherm is a better fit in the temperature range studied than the other isotherms for IRA-400 Cl resin/A. campestris biocomposite. Thermodynamic values such as enthalpy, change in entropy and Gibbs free energy were measured, and the biosorption process was found to be spontaneous and endothermic. As a result, IRA-400 Cl resin/A. campestris is a suitable biosorbent and has more typical proximity than IRA-4200 Cl resin/A. campestris biocomposite for biosorption of Brilliant Blue G from aqueous solutions.





Similar content being viewed by others
References
Alver E, Metin AÜ (2012) Anionic dye removal from aqueous solutions using modified zeolite: adsorption kinetics and isotherm studies. Chem Eng J 9(67):200–202. https://doi.org/10.1016/j.cej.2012.06.038
Arslan I, Balcioǧlu IA, Bahnemann DW (2000) Advanced chemical oxidation of reactive dyes in simulated dyehouse effluents by ferrioxalate Fenton/UV-A and TiO2/UV-A processes. Dyes Pigm 47(3):207–218. https://doi.org/10.1016/S0143-7208(00)00082-6
Dahri MK, Kooh MRR, Lim LBL (2015) Application of Casuarinas equisetifolia needle for the removal of methylene blue and malachite green dyes from aqueous solution. Alex Eng J 54:1253–1263. https://doi.org/10.1016/j.aej.2015.07.005
Forgacs E, Cserhati T, Oros G (2004) Removal of synthetic dyes from wastewaters: a review. Environ Int 30(7):953–971. https://doi.org/10.1016/j.envint.2004.02.001
Freundlich HMF (1906) Over the adsorption in solution. J Phys Chem 57(385471):1100–1107
Fu Y, Viraraghavan T (2001) Fungal decolorization of dye wastewaters: a review. Bioresour Technol 79:251–262. https://doi.org/10.1016/S0960-8524(01)00028-1
Gupta VK, Mittal A, Krishnan L (2004) Adsorption kinetics and column operations for the removal and recovery of malachite green from wastewater using bottom ash. Sep Purif Technol 40(1):87–96. https://doi.org/10.1016/j.seppur.2004.01.008
Gupta VK, Alok M, Lisha K, Jyoti M (2006) Adsorption treatment and recovery of the hazardous dye, Brilliant Blue FCF, over bottom ash and de-oiled soya. J Colloid Interface Sci 293(1):16–26. https://doi.org/10.1016/j.jcis.2005.06.021
Holkar CR, Jadhav AJ, Pinjari DV, Mahamuni NM, Pandit AB (2016) A critical review on textile wastewater treatments: possible approaches. J Environ Manag 182:351–366. https://doi.org/10.1016/j.jenvman.2016.07.090
Huang R, Liu Q, Huo J, Yang B (2017) Adsorption of methyl orange onto protonated cross-linked chitosan. Arab J Chem 10(1):24–32. https://doi.org/10.1016/j.arabjc.2013.05.017
Hunge YM, Yadav AA, Mahadik MA, Mathe VL, Bhosale CH (2018) A highly efficient visible-light responsive sprayed WO3/FTO photoanode for photoelectrocatalytic degradation of brilliant blue. J Taiwan Inst Chem E 85:273–281. https://doi.org/10.1016/j.jtice.2018.01.048
Jaafari J, Barzanouni H, Mazloomi S, Farahani NAA, Sharafi K, Soleimani P, Haghighat GA (2020) Effective adsorptive removal of reactive dyes by magnetic chitosan nanoparticles: kinetic, isothermal studies and response surface methodology. Int J Biol Macromol 164:344–355. https://doi.org/10.1016/j.ijbiomac.2020.07.042
Jia HL, Chen YC, Ji L, Lin LX, Guan MY, Yang Y (2019) Cosensitization of porphyrin dyes with new X type organic dyes for efficient dye-sensitized solar cells. Dyes Pigm 163:589–593. https://doi.org/10.1016/j.dyepig.2018.12.048
Jiang T, Liang Y, He Y, Wang Q (2015) Activated carbon/NiFe2O4 magnetic composite: a magnetic adsorbent for the adsorption of methyl orange. J Environ Chem Eng 3(3):1740–1751. https://doi.org/10.1016/j.jece.2015.06.020
Jura G, Harkins WD (1946) Surfaces of solids. XIV. A unitary thermodynamic theory of the adsorption of vapors on solids and of insoluble films on liquid subphases. J Am Chem Soc 68(10):1941–1952. https://doi.org/10.1021/ja01214a022
Khambhaty Y, Mody K, Basha S (2012) Efficient removal of Brilliant Blue G (BBG) from aqueous solutions by marine Aspergillus wentii: kinetics, equilibrium and process design. Ecol Eng 41:74–83. https://doi.org/10.1016/j.ecoleng.2012.01.002
Khouni I, Marrot B, Amar RB (2012) Treatment of reconstituted textile wastewater containing a reactive dye in an aerobic sequencing batch reactor using a novel bacterial consortium. Sep Purif Technol 87:110–119. https://doi.org/10.1016/j.seppur.2011.11.030
Kıvanç MR, Yönten V (2020) A statistical optimization of methylene blue removal from aqueous solutions by Agaricus campestris using multi-step experimental design with response surface methodology: isotherm, kinetic and thermodynamic studies. Surf Interfaces 18:100414. https://doi.org/10.1016/j.surfin.2019.100414
Kousha M, Daneshvar E, Dopeikar H, Taghavi D, Bhatnagar A (2012) Box-Behnken design optimization of Acid Black 1 dye biosorption by different brown macroalgae. Chem Eng J 179:158–168. https://doi.org/10.1016/j.cej.2011.10.073
Labanda J, Sabate J, Llorens J (2009) Modeling of the dynamic adsorption of an anionic dye through ion-exchange membrane adsorber. Membr Sci 340(1–2):234–240. https://doi.org/10.1016/j.memsci.2009.05.036
Langmuir I (1918) The adsorption of gases on plane surfaces of glass, mica and platinum. J Am Chem Soc 40(9):1361–1403. https://doi.org/10.1021/ja02242a004
Laskar N, Kumar U (2019) Removal of Brilliant Green dye from water by modified Bambusa Tulda: adsorption isotherm, kinetics and thermodynamics study. Int J Environ Sci Te 16(3):1649–1662
Linhui Z, Chengdong G, Bin Z, Zhentao Z, Rui Y, Yaoji T, Jing Y (2017) Adsorption of dyes onto sodium alginate graft poly (acrylic acid-co-2-acrylamide-2-methyl propane sulfonic acid)/kaolin hydrogel composite. Polym Compos 25(8):627–634. https://doi.org/10.1177/096739111702500808
Liu CH, Wu JS, Chiu HC, Suen SY, Chu KH (2007) Removal of anionic reactive dyes from water using anion exchange membranes as adsorbers. Water Res 41(7):1491–1500. https://doi.org/10.1016/j.watres.2007.01.023
Mahmoodi NM (2016) Photocatalytic degradation of textile dyes using ozonation and magnetic nickel ferrite nanoparticle. Prog Color Color Coat 9(3):163–171
Moyo W, Chaukura N, Msagati TA, Mamba BB, Heijman SG, Nkambule TT (2019) The properties and removal efficacies of natural organic matter fractions by South African drinking water treatment plants. J Environ Chem Eng 7(3):103101. https://doi.org/10.1016/j.jece.2019.103101
Naghipour D, Taghavi K, Ashournia M, Jaafari J, Arjmand Movarrekh R (2020) A study of Cr(VI) and NH4 + adsorption using greensand (glauconite) as a low-cost adsorbent from aqueous solutions. Water Environ J 34(1):45–56. https://doi.org/10.1111/wej.12440
Naushad M, Abdullah AL, Othman Z, Rabiul Awual M, Alfadul SM, Ahamad T (2016) Adsorption of rose Bengal dye from aqueous solution by amberlite Ira-938 resin: kinetics, isotherms, and thermodynamic studies. Desalin Water Treat 57(29):13527–13533. https://doi.org/10.1080/19443994.2015.1060169
Önal Y, Akmil-Başar C, Sarıcı-Özdemir Ç (2007) Investigation kinetics mechanisms of adsorption malachite green onto activated carbon. J Hazard Mater 146(1–2):194–203. https://doi.org/10.1016/j.jhazmat.2006.12.006
Öztürk G (2018). Farklı adsorbanlar kullanılarak atıksulardan reaktif boyaların gideriminde adsorpsiyon koşullarının araştırılması, Yüksek Lisans Tezi, Bilecik Şeyh Edebali Üniversitesi, Fen Bilimleri Enstitüsü
Ozturk D, Yilmaz AE (2020) Investigation of electrochemical degradation of Basic Red 13 dye in aqueous solutions based on COD removal: numerical optimization approach. Int J Environ Sci Tecnol. https://doi.org/10.1007/s13762-020-02692-2
Ozturk D, Sahan T, Bayram T, Erkus A (2017) Application of response surface methodology (RSM) to optimize the adsorption conditions of cationic Basic Yellow 2 onto pumice samples as a new adsorbent. Fresenius Environ Bull 26:3285-5
Pang H, Wu Y, Wang X, Hu B, Wang X (2019) Recent advances in composites of graphene and layered double hydroxides for water remediation: a review. Chem Asian J 14(15):2542–2552. https://doi.org/10.1002/asia.201900493
Reis LGT, Robaina NF, Pacheco WF, Cassella RJ (2011) Separation of malachite green and methyl green cationic dyes from aqueous medium by adsorption on Amberlite XAD-2 and XAD-4 resins using sodium dodecylsulfate as carrier. Chem Eng J 171:532–540. https://doi.org/10.1016/j.cej.2011.04.024
Sarma GK, Gupta SS, Bhattacharyya KG (2019) Removal of hazardous basic dyes from aqueous solution by adsorption onto kaolinite and acid-treated kaolinite: kinetics, isotherm and mechanistic study. SN Appl Sci 1(3):211. https://doi.org/10.1007/s42452-019-0216y
Şen F, Demirbaş Ö, Çalımlı MH, Aygün A, Alma MH, Nas MS (2018) The dye removal from aqueous solution using polymer composite films. Appl Water Sci 8(7):206. https://doi.org/10.1007/s13201-018-0856-x
Shi Li G, Wang D, Feng C, Tang H (2007) Removal of direct dyes by coagulation: the performance of preformed polymeric aluminum species. J Hazard Mater 143(1–2):567–574. https://doi.org/10.1016/j.jhazmat.2006.09.076
Sinha S, Behera SS, Das S, Basu A, Mohapatra RK, Murmu BM, Dhal NK, Tripathy SK, Parhi PK (2018) Removal of Congo Red dye from aqueous solution using Amberlite IRA-400 in batch and fixed bad reactors. Chem Eng Commun 205(4):432–444. https://doi.org/10.1080/00986445.2017.1399366
Sugashini S, Begum KMS (2013) Optimization using central composite design (CCD) for the biosorption of Cr(VI) ions by cross linked chitosan carbonized rice husk (CCACR). Clean Technol Environ 15(2):293–302. https://doi.org/10.1007/s10098-012-0512-3
Temkin M, Pyzhev V (1940) Kinetics of ammonia synthesis on promoted iron catalysts. Acta Phys Chem URSS 12:327–356
Torgut G, Tanyol M, Biryan F, Pihtili G, Demirelli K (2017) Application of response surface methodology for optimization of Remazol Brilliant Blue R removal onto a novel polymeric adsorbent. J Taiwan Inst Chem E 80:406–414. https://doi.org/10.1016/j.jtice.2017.07.030
Wang L (2012) Application of activated carbon derived from ‘waste’bamboo culms for the adsorption of azo disperse dye: kinetic, equilibrium and thermodynamic studies. J Environ Manag 102:79–87. https://doi.org/10.1016/j.jenvman.2012.02.01
Yener J, Kopac T, Doğu G, Doğu T (2006) Adsorption of Basic Yellow 28 from aqueous solutions with clinoptilolite and amberlite. J Colloid Interface Sci 294:255–264. https://doi.org/10.1016/j.jcis.2005.07.040
Yonten V, Tanyol M, Yildirim N, Yildirim NC, Ince M (2016) Optimization of Remazol Brilliant Blue R dye removal by novel biosorbent P. eryngii immobilized on Amberlite XAD-4 using response surface methodology. Desalin Water Treat 57(33):15592–15602. https://doi.org/10.1080/19443994.2015.1070760
Yönten V, Alp H, Yildirim N, Cikcikoğlu Y, Nuran OA (2017) Investigation of optimum conditions for efficient COD reduction in synthetic sulfamethazine solutions by Pleurotus eryngii var. ferulae using response surface methodology. J Taiwan Inst Chem E 80(80):349–355. https://doi.org/10.1016/j.jtice.2017.07.021
Zhang M, Yu Z, Yu H (2020) Adsorption of Eosin Y, methyl orange and brilliant green from aqueous solution using ferroferric oxide/polypyrrole magnetic composite. Polym Bull 77(2):1049–1066. https://doi.org/10.1007/s00289-019-02792-1
Acknowledgements
The authors would like to express sincerely their gratitude to Yuzuncu Yil University (Faculty of Engineering Research Laboratories) for their analytical assistance in carrying out our laboratory studies.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editorial responsibility: Shahid Hussain.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yönten, V., Özgüven, A., Ahmed, A.A. et al. Effect of some medium parameters on Brilliant Blue G biosorption by Amberlite resin/Agaricus campestris. Int. J. Environ. Sci. Technol. 18, 1709–1718 (2021). https://doi.org/10.1007/s13762-020-02954-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-020-02954-z