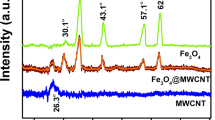
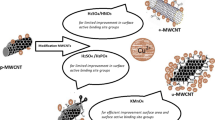
Abstract
Herein, cobalt (0) metals loaded on multiwalled carbon nanotube nanoadsorbents were synthesized by a straightforward ultrasonic technique, and the removal efficiency of cobalt (0) multiwalled carbon nanotube nanoadsorbents were evaluated in the adsorption of methylene blue at various experimental conduction in an ultrasonic bath. Nanoadsorbents amount, solution pH, ultrasonic power, methylene blue concentrations, temperature, and H2O2 concentrations were studied as experimental parameters in the study. The synthesized cobalt (0) multiwalled carbon nanotube nanoadsorbents were characterized using some advanced spectroscopic techniques. The adsorption mechanism of methylene blue using the as-synthesized nanoadsorbents was evaluated by investigating some kinetic models including intra-particles diffusion, the pseudo-first-order, and the second-order models. The experimental findings of methylene blue adsorption by cobalt multiwalled carbon nanotube was found to be compatible with the pseudo-second-order model. Additionally, some kinetic activation studies like Gibbs free energy, enthalpy, and activation energy for the methylene blue adsorption were investigated. The enthalpy of the process was found to be 25.29 kJ mol−1 that shows the adsorption process is endothermic. The maximum adsorption efficiency of the nanoadsorbents was found to be 1.04 × 10−3 mol g−1 (324.34 mg g−1). The comparisons of results to the previous studies revealed that the synthesized nanoparticles were found to be very effective for the removal of methylene blue from the aquatic mediums.













Similar content being viewed by others
References
Akova A, Ustun G (2000) Activity and adsorption of lipase from Nigella sativa seeds on Celite at different pH values. Biotechnol Lett 22:355–359. https://doi.org/10.1023/A:1005668500716
Ali I, Asim M, Khan TA (2012) Low cost adsorbents for the removal of organic pollutants from wastewater. J Environ Manag 113:170–183
Alkan M, Çelikçcapa S, Demirbaş Ö, Dogan M (2005) Removal of reactive blue 221 and acid blue 62 anionic dyes from aqueous solutions by sepiolite. Dye Pigment 65:251–259. https://doi.org/10.1016/j.dyepig.2004.07.018
Alkan M, Demirbaş Ö, Doǧan M (2007) Adsorption kinetics and thermodynamics of an anionic dye onto sepiolite. Microporous Mesoporous Mater 101:388–396. https://doi.org/10.1016/j.micromeso.2006.12.007
Alkan M, Doğan M, Turhan Y et al (2008) Adsorption kinetics and mechanism of maxilon blue 5G dye on sepiolite from aqueous solutions. Chem Eng J 139:213–223. https://doi.org/10.1016/J.CEJ.2007.07.080
Arrechea S, Guerrero-Gutiérrez EMA, Velásquez L et al (2020) Effect of additions of multiwall carbon nanotubes (MWCNT, MWCNT-COOH and MWCNT-Thiazol) in mechanical compression properties of a cement-based material. Materialia 11:100739. https://doi.org/10.1016/j.mtla.2020.100739
Avcı A, İnci İ, Baylan N (2020) Adsorption of ciprofloxacin hydrochloride on multiwall carbon nanotube. J Mol Struct 1206:127711. https://doi.org/10.1016/j.molstruc.2020.127711
Ayranci R, Demirkan B, Sen B et al (2019) Use of the monodisperse Pt/Ni@rGO nanocomposite synthesized by ultrasonic hydroxide assisted reduction method in electrochemical nonenzymatic glucose detection. Mater Sci Eng C 99:951–956. https://doi.org/10.1016/j.msec.2019.02.040
Babu DS, Srivastava V, Nidheesh PV, Kumar MS (2019) Detoxification of water and wastewater by advanced oxidation processes. Sci Total Environ 696:133961
Bae S, Kim D, Lee W (2013) Degradation of diclofenac by pyrite catalyzed Fenton oxidation. Appl Catal B Environ 134–135:93–102. https://doi.org/10.1016/j.apcatb.2012.12.031
Bagal MV, Lele BJ, Gogate PR (2013) Removal of 2,4-dinitrophenol using hybrid methods based on ultrasound at an operating capacity of 7 L. Ultrason Sonochem 20:1217–1225. https://doi.org/10.1016/j.ultsonch.2013.01.015
Bai L, Li Z, Zhang Y et al (2015) Synthesis of water-dispersible graphene-modified magnetic polypyrrole nanocomposite and its ability to efficiently adsorb methylene blue from aqueous solution. Chem Eng J 279:757–766. https://doi.org/10.1016/j.cej.2015.05.068
Boukhalfa N, Boutahala M, Djebri N, Idris A (2019) Maghemite/alginate/functionalized multiwalled carbon nanotubes beads for methylene blue removal: adsorption and desorption studies. J Mol Liq 275:431–440. https://doi.org/10.1016/j.molliq.2018.11.064
Çalımlı MH, Demirbaş Ö, Aygün A et al (2018) Immobilization kinetics and mechanism of bovine serum albumin on diatomite clay from aqueous solutions. Appl Water Sci 8:1–12. https://doi.org/10.1007/s13201-018-0858-8
Carneiro PA, Umbuzeiro GA, Oliveira DP, Zanoni MVB (2010) Assessment of water contamination caused by a mutagenic textile effluent/dyehouse effluent bearing disperse dyes. J Hazard Mater 174:694–699. https://doi.org/10.1016/j.jhazmat.2009.09.106
Castellari M, Versari A, Fabiani A et al (2001) Removal of ochratoxin A in red wines by means of adsorption treatments with commercial fining agents. J Agric Food Chem 49:3917–3921. https://doi.org/10.1021/jf010137o
Chatterjee S, Chatterjee T, Woo SH (2010) A new type of chitosan hydrogel sorbent generated by anionic surfactant gelation. Bioresour Technol 101:3853–3858. https://doi.org/10.1016/j.biortech.2009.12.089
Chen H, Li J, Shao D et al (2012) Poly(acrylic acid) grafted multiwall carbon nanotubes by plasma techniques for Co(II) removal from aqueous solution. Chem Eng J 210:475–481. https://doi.org/10.1016/j.cej.2012.08.082
Çiçek F, Özer D, Özer A, Özer A (2007) Low cost removal of reactive dyes using wheat bran. J Hazard Mater 146:408–416. https://doi.org/10.1016/j.jhazmat.2006.12.037
Demir E, Sen B, Sen F (2017) Highly efficient Pt nanoparticles and f-MWCNT nanocomposites based counter electrodes for dye-sensitized solar cells. Nano-Struct Nano-Objects 11:39–45. https://doi.org/10.1016/j.nanoso.2017.06.003
Demirbaş Ö, Nas M (2016a) Kinetics and mechanism of the adsorption of methylene blue from aqueous solution onto turkish green clay. Arch Curr Res Int 6:1–10. https://doi.org/10.9734/acri/2016/30677
Demirbaş Ö, Nas MS (2016b) Kinetics and thermodynamics properties of catalase onto diatomite. Asian J Chem Sci 1:1–15
Demirbaş O, Alkan M, Doğan M (2002) The removal of victoria blue from aqueous solution by adsorption on a low-cost material. Adsorption 8:341–349. https://doi.org/10.1023/A:1021589514766
Demirbaş Ö, Alkan M, Demirbaş A (2015) Adsorption of casein onto some oxide minerals and electrokinetic properties of these particles. Microporous Mesoporous Mater 204:197–203. https://doi.org/10.1016/j.micromeso.2014.11.012
Demirbaş Ö, Nas MS, Manickam S (2016) Article no.ACRI.30677 Reviewers: (1) Atiya Firdous, Jinnah University for Women. Zimbabwe Orig Res Artic Demirbaş Nas 6:30677. https://doi.org/10.9734/ACRI/2016/30677
Demirbaş Ö, Çalımlı MH, Demirkan B et al (2019a) Thermodynamics, kinetics, and adsorption properties of biomolecules onto carbon-based materials obtained from food wastes. Bionanoscience. https://doi.org/10.1007/s12668-019-00628-w
Demirbaş Ö, Çalımlı MH, Demirkan B et al (2019b) The kinetic parameters of adsorption of enzymes using carbon-based materials obtained from different food wastes. Bionanoscience 9:749–757. https://doi.org/10.1007/s12668-019-00635-x
Doǧan M, Alkan M (2003) Adsorption kinetics of methyl violet onto perlite. Chemosphere 50:517–528. https://doi.org/10.1016/S0045-6535(02)00629-X
Doǧan M, Alkan M, Demirbaş Ö et al (2006) Adsorption kinetics of maxilon blue GRL onto sepiolite from aqueous solutions. Chem Eng J 124:89–101. https://doi.org/10.1016/j.cej.2006.08.016
Fu W, Zhang W (2018) Microwave-enhanced membrane filtration for water treatment. J Membr Sci 568:97–104. https://doi.org/10.1016/j.memsci.2018.09.064
Gandhi N, Sirisha D, Sekhar KBC (2016) Adsorption of fluoride (F-) from aqueous solution by using pineapple (Ananas comosus) peel and orange (Citrus sinensis) peel powders. Int J Bioremediat Biodegrad 4:55–67. https://doi.org/10.12691/ijebb-4-2-4
Gong J, Liu J, Jiang Z et al (2015) A facile approach to prepare porous cup-stacked carbon nanotube with high performance in adsorption of methylene blue. J Colloid Interface Sci 445:195–204. https://doi.org/10.1016/j.jcis.2014.12.078
Hassani A, Soltani RDC, Karaca S, Khataee A (2015) Preparation of montmorillonite-alginate nanobiocomposite for adsorption of a textile dye in aqueous phase: isotherm, kinetic and experimental design approaches. J Ind Eng Chem 21:1197–1207. https://doi.org/10.1016/j.jiec.2014.05.034
Huang R, Fang Z, Yan X, Cheng W (2012) Heterogeneous sono-Fenton catalytic degradation of bisphenol A by Fe3O4 magnetic nanoparticles under neutral condition. Chem Eng J 197:242–249. https://doi.org/10.1016/j.cej.2012.05.035
Irani E, Amoli-Diva M (2020) Hybrid adsorption–photocatalysis properties of quaternary magneto-plasmonic ZnO/MWCNTs nanocomposite for applying synergistic photocatalytic removal and membrane filtration in industrial wastewater treatment. J Photochem Photobiol A Chem 391:112359. https://doi.org/10.1016/j.jphotochem.2020.112359
Janoš P, Šmídová V (2005) Effects of surfactants on the adsorptive removal of basic dyes from water using an organomineral sorbent-iron humate. J Colloid Interface Sci 291:19–27. https://doi.org/10.1016/j.jcis.2005.04.065
Jaseela PK, Garvasis J, Joseph A (2019) Selective adsorption of methylene blue (MB) dye from aqueous mixture of MB and methyl orange (MO) using mesoporous titania (TiO2)–poly vinyl alcohol (PVA) nanocomposite. J Mol Liq 286:110908. https://doi.org/10.1016/j.molliq.2019.110908
Jin X, Yu B, Lin J, Chen Z (2016) Integration of biodegradation and nano-oxidation for removal of PAHs from aqueous solution. ACS Sustain Chem Eng 4:4717–4723. https://doi.org/10.1021/acssuschemeng.6b00933
Jin L, Zhao X, Qian X, Dong M (2018) Nickel nanoparticles encapsulated in porous carbon and carbon nanotube hybrids from bimetallic metal-organic-frameworks for highly efficient adsorption of dyes. J Colloid Interface Sci 509:245–253. https://doi.org/10.1016/j.jcis.2017.09.002
Jose M, Harsha N, Suhailath K et al (2016) Hydrogen phosphate anions modified hydrogen titanate nanotubes for methylene blue adsorption from aqueous solution: validating novel method of predicting adsorption capacity. J Environ Chem Eng 4:1295–1307. https://doi.org/10.1016/j.jece.2016.01.042
Joshi S, Garg VK, Kataria N, Kadirvelu K (2019) Applications of Fe3O4@AC nanoparticles for dye removal from simulated wastewater. Chemosphere 236:124280. https://doi.org/10.1016/j.chemosphere.2019.07.011
Kanakkillam SS, Krishnan B, Avellaneda DA, Shaji S (2020) Surfactant free stable cobalt oxide nanocolloid in water by pulsed laser fragmentation and its thin films for visible light photocatalysis. Colloids Surf A Physicochem Eng Asp 594:124657. https://doi.org/10.1016/j.colsurfa.2020.124657
Kannan N, Sundaram MM (2001) Kinetics and mechanism of removal of methylene blue by adsorption on various carbons—a comparative study. Dye Pigment 51:25–40. https://doi.org/10.1016/S0143-7208(01)00056-0
Khataee AR, Vahid B, Behjati B, Safarpour M (2013) Treatment of a dye solution using photoelectro-Fenton process on the cathode containing carbon nanotubes under recirculation mode: investigation of operational parameters and artificial neural network modeling. Environ Prog Sustain Energy 32:557–563. https://doi.org/10.1002/ep.11657
Khataee A, Saadi S, Safarpour M, Joo SW (2015) Sonocatalytic performance of Er-doped ZnO for degradation of a textile dye. Ultrason Sonochem 27:379–388. https://doi.org/10.1016/j.ultsonch.2015.06.010
Khataee A, Kayan B, Kalderis D et al (2017) Ultrasound-assisted removal of Acid Red 17 using nanosized Fe3O4-loaded coffee waste hydrochar. Ultrason Sonochem 35:72–80. https://doi.org/10.1016/j.ultsonch.2016.09.004
Kumar KV, Ramamurthi V, Sivanesan S (2005) Modeling the mechanism involved during the sorption of methylene blue onto fly ash. J Colloid Interface Sci 284:14–21. https://doi.org/10.1016/j.jcis.2004.09.063
Li H, Li TT, Qian J et al (2020) CuCo2S4 integrated multiwalled carbon nanotube as high-performance electrocatalyst for electroreduction of nitrogen to ammonia. Int J Hydrog Energy 45:14640–14647. https://doi.org/10.1016/j.ijhydene.2020.03.198
Luo W, Zhu L, Wang N et al (2010) Efficient removal of organic pollutants with magnetic nanoscaled BiFeO3 as a reusable heterogeneous Fenton-like catalyst. Environ Sci Technol 44:1786–1791. https://doi.org/10.1021/es903390g
Mahdavi Talarposhti A, Donnelly T, Anderson GK (2001) Colour removal from a simulated dye wastewater using a two-phase anaerobic packed bed reactor. Water Res 35:425–432. https://doi.org/10.1016/s0043-1354(00)00280-3
Mishra AK, Arockiadoss T, Ramaprabhu S (2010) Study of removal of azo dye by functionalized multi walled carbon nanotubes. Chem Eng J 162:1026–1034. https://doi.org/10.1016/j.cej.2010.07.014
Mongalo NI, Dikhoba PM, Soyingbe SO, Makhafola TJ (2018) Antifungal, anti-oxidant activity and cytotoxicity of South African medicinal plants against mycotoxigenic fungi. Heliyon. https://doi.org/10.1016/j.heliyon.2018.e00973
Nas MS, Calimli MH, Burhan H et al (2019) Synthesis, characterization, kinetics and adsorption properties of Pt-Co@GO nano-adsorbent for methylene blue removal in the aquatic mediums using ultrasonic process systems. J Mol Liq 296:112100. https://doi.org/10.1016/j.molliq.2019.112100
Othman NH, Alias NH, Shahruddin MZ et al (2018) Adsorption kinetics of methylene blue dyes onto magnetic graphene oxide. J Environ Chem Eng 6:2803–2811. https://doi.org/10.1016/j.jece.2018.04.024
Özer A, Özer D, Ekiz HI (2005) The equilibrium and kinetic modelling of the biosorption of copper(II) ions on cladophora crispata. Adsorption 10:317–326. https://doi.org/10.1007/s10450-005-4817-y
Patil MP, Jin X, Simeon NC et al (2018) Anticancer activity of sasa borealis leaf extract-mediated gold nanoparticles. Artif Cells Nanomed Biotechnol 46:82–88. https://doi.org/10.1080/21691401.2017.1293675
Pineda X, Quintana GC, Herrera AP, Sánchez JH (2020) Preparation and characterization Of magnetic cellulose fibers modified with cobalt ferrite nanoparticles. Mater Chem Phys. https://doi.org/10.1016/j.matchemphys.2020.122778
Qian F, Sun X, Liu Y (2013) Removal characteristics of organics in bio-treated textile wastewater reclamation by a stepwise coagulation and intermediate GAC/O3 oxidation process. Chem Eng J 214:112–118. https://doi.org/10.1016/j.cej.2012.09.130
Rajabi M, Mahanpoor K (2017) Removal of dye molecules from aqueous solution by carbon nanotubes and carbon nanotube functional groups: critical review. RSC Adv. pubs.rsc.org
Rakap M, Özkar S (2010) Hydrogen generation from the hydrolysis of ammonia-borane using intrazeolite cobalt(0) nanoclusters catalyst. Int J Hydrog Energy 35:3341–3346. https://doi.org/10.1016/j.ijhydene.2010.01.138
Ramakrishna KR, Viraraghavan T (1997) Dye removal using low cost adsorbents. Wat Sci Technol 36:189–196
Rubin E, Rodriguez P, Herrero R et al (2005) Removal of Methylene Blue from aqueous solutions using as biosorbent Sargassum muticum: an invasive macroalga in Europe. J Chem Technol Biotechnol 80:291–298. https://doi.org/10.1002/jctb.1192
Savk A, Sen B, Demirkan B et al (2018) Graphene oxide-chitosan furnished monodisperse platinum nanoparticles as importantly competent and reusable nanosorbents for methylene blue removal. Chitosan. books.google.com
Sen B, Kuzu S, Demir E et al (2017) Highly monodisperse RuCo nanoparticles decorated on functionalized multiwalled carbon nanotube with the highest observed catalytic activity in the dehydrogenation of dimethylamine-borane. Int J Hydrog Energy 42:23292–23298. https://doi.org/10.1016/j.ijhydene.2017.06.032
Simon G, Meziane L, Courty A et al (2016) Low wavenumber Raman scattering of cobalt nanoparticles self-organized in 3D superlattices far from surface plasmon resonance. J Raman Spectrosc 47:248–251. https://doi.org/10.1002/jrs.4782
Tan IAW, Ahmad AL, Hameed BH (2008) Adsorption of basic dye using activated carbon prepared from oil palm shell: batch and fixed bed studies. Desalination 225:13–28. https://doi.org/10.1016/j.desal.2007.07.005
Tang Y, Zhao Y, Lin T et al (2019) Adsorption performance and mechanism of methylene blue by H3PO4-modified corn stalks. J Environ Chem Eng 7:103398. https://doi.org/10.1016/j.jece.2019.103398
Tekin N, Dinçer A, Demirbaş Ö, Alkan M (2006) Adsorption of cationic polyacrylamide onto sepiolite. J Hazard Mater 134:211–219. https://doi.org/10.1016/j.jhazmat.2005.11.005
Tissera ND, Wijesena RN, Yasasri H et al (2020) Fibrous keratin protein bio micro structure for efficient removal of hazardous dye waste from water: surface charge mediated interfaces for multiple adsorption desorption cycles. Mater Chem Phys 246:122790. https://doi.org/10.1016/j.matchemphys.2020.122790
Vadivelan V, Vasanth Kumar K (2005) Equilibrium, kinetics, mechanism, and process design for the sorption of methylene blue onto rice husk. J Colloid Interface Sci 286:90–100. https://doi.org/10.1016/j.jcis.2005.01.007
Wang W, Jiao T, Zhang Q et al (2015) Hydrothermal synthesis of hierarchical core–shell manganese oxide nanocomposites as efficient dye adsorbents for wastewater treatment. RSC Adv. https://doi.org/10.1039/C5RA08678G
Wu Q, Zhang H, Zhou L et al (2016) Synthesis and application of rGO/CoFe2O4 composite for catalytic degradation of methylene blue on heterogeneous Fenton-like oxidation. J Taiwan Inst Chem Eng 67:484–494. https://doi.org/10.1016/j.jtice.2016.08.004
Yagub MT, Sen TK, Afroze S, Ang HM (2014) Dye and its removal from aqueous solution by adsorption: a review. Adv Colloid Interface Sci 209:172–184
Yasar A, Khalil S, Tabinda AB, Malik A (2013) Comparison of cost and treatment efficiency of solar assisted advance oxidation processes for textile dye bath effluent. Korean J Chem Eng 30:131–138. https://doi.org/10.1007/s11814-012-0110-4
Yu L, Jiang L, Wang S et al (2018) Pectin microgel particles as high adsorption rate material for methylene blue: performance, equilibrium, kinetic, mechanism and regeneration studies. Int J Biol Macromol 112:383–389. https://doi.org/10.1016/j.ijbiomac.2018.01.193
Zhang G, Gao Y, Zhang Y, Guo Y (2010) Fe2O3-pillared rectorite as an efficient and stable Fenton-like heterogeneous catalyst for photodegradation of organic contaminants. Environ Sci Technol 44:6384–6389. https://doi.org/10.1021/es1011093
Zhou T, Che G, Ding L et al (2019) Recent progress of selective adsorbents: from preparation to complex sample pretreatment. Trends Anal Chem 121:115678
Acknowledgements
I would like to thank to Igdir University (2018-05) for funding.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editorial responsibility: S. Mirkia.
Rights and permissions
About this article
Cite this article
Çalımlı, M.H. Magnetic nanocomposite cobalt-multiwalled carbon nanotube and adsorption kinetics of methylene blue using an ultrasonic batch. Int. J. Environ. Sci. Technol. 18, 723–740 (2021). https://doi.org/10.1007/s13762-020-02855-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-020-02855-1