Abstract
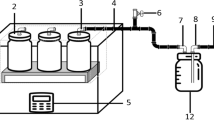
The aim of this laboratory-scale study is to propose a practical method for better mixing and mercaptan removal from an industrial feed. The role of proper mixing for mercaptan removal from an industrial LPG stream using a commercial mercaptan scavenger has been investigated in the laboratory scale. The LPG obtained from the Ilam Gas Treating Company includes a very high concentration of mercaptan, and intense environmental pollution will occur during carrying this product to other refineries. The effect of the mixer length, including 20 and 40 cm, and element pitches, including 1, 2 and 3 mm, as well as the LPG stream flow rates, including 2, 18 and 30 mL/s, on the mercaptan removal efficiency and the mass transfer coefficient was investigated using UOP 163 standard method. The mercaptan removal efficiency was enhanced 15.2, 18.1 and 19.7% for a mixer length of 20 cm and element pitch 1 mm at 2, 18 and 30 mL/s flow rates, respectively. Similarly, for the mixer length of 40 cm and element pitch 1 mm, the largest enhancement 23% of the mercaptan removal efficiency was obtained. In addition, the mass transfer coefficient will increase as the flow rate increases, and the mixer pitch decreases.








Similar content being viewed by others
Abbreviations
- C in :
-
Concentrations of mercaptan in the inlet streams (mg/m3)
- C out :
-
Concentrations of mercaptan in the outlet streams (mg/m3)
- C * :
-
C* is the equilibrium concentration of the mercaptan (mg/m3)
- E :
-
Removal efficiency (−)
- K L a :
-
Volumetric mass transfer coefficient (1/s)
- Q inj :
-
Injection volumetric flow rate (mL/s)
- Q main :
-
Main volumetric flow rate (mL/s)
- t m :
-
Residence time of mixture two stream (s)
- V :
-
Total volume of mixing line (m3)
- in:
-
Inlet
- out:
-
Outlet
- inj:
-
Injection
References
Al-Atabi M (2011) Design and assessment of a novel static mixer. Can J Chem Eng 89(3):550–554
Assmann N, von Rohr PR (2011) Extraction in microreactors: intensification by adding an inert gas phase. Chem Eng Process 50(8):822–827
Bertsch A, Heimgartner S, Cousseau P, Renaud P (2001) Static micromixers based on large-scale industrial mixer geometry. Lab Chip 1(1):56–60
Chatti I, Ghorbel A, Grange P, Colin JM (2002) Oxidation of mercaptans in light oil sweetening by cobalt (II) phthalocyanine–hydrotalcite catalysts. Catal Today 75(1–4):113–117
Fang JZ, Lee DJ (2001) Micromixing efficiency in static mixer. Chem Eng Sci 56(12):3797–3802
Hobbs DM, Muzzio FJ (1998) Reynolds number effects on laminar mixing in the Kenics static mixer. Chem Eng J 70(2):93–104
Jaworski Z, Pianko-Oprych P (2002) Two-phase laminar flow simulations in a Kenics static mixer: standard Eulerian and Lagrangian approaches. Chem Eng Res Des 80(8):910–916
Lang E, Drtina P, Streiff F, Fleischli M (1995) Numerical simulation of the fluid flow and the mixing process in a static mixer. Int J Heat Mass Transf 38(12):2239–2250
Legrand J, Morançais P, Carnelle G (2001) Liquid-liquid dispersion in an SMX-Sulzer static mixer. Chem Eng Res Des 79(8):949–956
Liu S, Hrymak AN, Wood PE (2006) Design modifications to SMX static mixer for improving mixing. AIChE J 52(1):150–157
Mohan VM, Kumar CS, Sujatha V, Prasad PR, Sarveswarao S (2010) Mass transfer correlation development for the presence of entry region coil as swirl promoter in tube. Int J Therm Sci 49(2):356–364
Rezvani MA, Zonoz FM (2015) An organic–inorganic hybrid compound constructed by polytungsto-vanadosilicate and hexadecyltrimethyl ammonium as an efficient catalyst for demercaptanization of crude oil. J Ind Eng Chem 22:83–91
Song HS, Han SP (2005) A general correlation for pressure drop in a Kenics static mixer. Chem Eng Sci 60(21):5696–5704
Sujatha V, Sarma CB, Raju GJ (1977) Studies on ionic mass transfer with coaxially placed helical tapes on a rod in forced convection flow. Chem Eng Process 36(1):67–73
Tajima H, Nagata T, Yamasaki A, Kiyono F, Masuyama T (2007) Formation of HFC-134a hydrate by static mixing. J Pet Sci Eng 56(1–3):75–81
Tsaoulidis D, Dore V, Angeli P, Plechkova NV, Seddon KR (2013) Dioxouranium (VI) extraction in microchannels using ionic liquids. Chem Eng J 227:151–157
Van Wageningen WF, Kandhai D, Mudde RF, Van Den Akker HE (2004) Dynamic flow in a Kenics static mixer: an assessment of various CFD methods. AIChE J 50(8):1684–1696
Yongping Z, Shengui J, Weihong X, Changlin C (2008) Adsorption of mercaptan from model gasoline on 13X loaded with Zn2+. Can J Chem Eng 86(2):186–191
You S, Higuchi N, Ueda T (2009) Macroscopic estimation method of the mixedness of Kenics type static mixer. Korean J Chem Eng 26(6):1497–1503
Zhang Y, Liu Z, Ren S, Wang W, Shen B (2014) Study on the basic centers and active oxygen species of solid-base catalysts for oxidation of iso-mercaptans. Appl Catal A Gen 473:125–131
Acknowledgements
The authors wish to express their thanks to the Ilam Gas Treating Company (IGTC) of Iran for preparing the LPG and mercaptan scavenger as well as the mercaptan measurement facility.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors certify that they have NO affiliations with or involvement in any organization or entity with any financial interest, or non-financial interest in the subject matter or materials discussed in this manuscript, and there is no conflict of interest concerning this study.
Additional information
Editorial Responsibiility: Agnieszka Galuszka.
Rights and permissions
About this article
Cite this article
Parvareh, A., Parvizi, F. Laboratory-scale study of mercaptan removal from LPG in the presence of passive mixing tools. Int. J. Environ. Sci. Technol. 17, 3199–3208 (2020). https://doi.org/10.1007/s13762-019-02561-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-019-02561-7