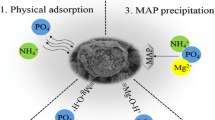
Abstract
Water pollution and purification are significant issues currently facing the world. Phosphorous is one of the pollutants which degrades water quality and is a cause of concern for eutrophication of water. This paper discusses the synthesis of a novel renewable resource-based nanocomposite, its use in phosphorus remediation, and its regeneration. The nanocomposite is synthesized from waste pine wood chips with stepwise chemical treatment. The synthetic method is environmental friendly and economically viable. Harmful chemicals are not used in the synthesis of the nanocomposite, which represents a significant shift from existing technologies. These nanocomposites are found to be useful for the removal of phosphate from contaminated waters, mainly agricultural wastes, mine drainage, and seawater. The superiority of using nanocomposites for adsorption is mainly associated with the positive charge as well as the presence of iron oxide nanoparticles on the surface of the media. Scanning electron microscopy and X-ray photoelectron spectroscopy were performed to determine the size and the elements present on the surface of the nanocomposite. Kinetic studies and adsorption isotherm were conducted which helped in proposing a mechanism for adsorption of phosphate on the media. Results indicate that phosphorus levels can be brought down from 1000 parts per billion to at least 10 parts per billion from stock solutions. The phosphorus could also be desorbed and the media regenerated for reuse at least 100 times without loss of phosphorus removal efficiency.








Similar content being viewed by others
References
Arias CA, Del Bubba M, Brix H (2001) Phosphorus removal by sands for use as media in subsurface flow constructed reed beds. Water Res 35(5):1159–1168. https://doi.org/10.1016/S0043-1354(00)00368-7
Blaney LM, Cinar S, SenGupta AK (2007) Hybrid anion exchanger for trace phosphate removal from water and wastewater. Water Res 41(7):1603–1613. https://doi.org/10.1016/j.watres.2007.01.008
Bond PL, Erhart R, Wagner M, Keller J, Blackall LL (1999) Identification of some of the major groups of bacteria in efficient and nonefficient biological phosphorus removal activated sludge systems. Appl Environ Microbiol 65(9):4077–4084
Chen L, Huang X, Zhang Y, Yuan D (2015) A new polymeric ionic liquid-based magnetic adsorbent for the extraction of inorganic anions in water samples. J Chromatograph A 1403:37–44. https://doi.org/10.1016/j.chroma.2015.05.021
Chitrakar R, Tezuka S, Sonoda A, Sakane K, Ooi K, Hirotsu T (2006) Phosphate adsorption on synthetic goethite and akaganeite. J Colloild Interface Sci 298(2):602–608. https://doi.org/10.1016/j.jcis.2005.12.054
de Vicente I, Merino-Martos A, Guerrero F, Amores V, de Vicente J (2011) Chemical interferences when using high gradient magnetic separation for phosphate removal: consequences for lake restoration. J Hazard Mater 192(3):995–1001. https://doi.org/10.1016/j.jhazmat.2011.05.090
Delaney P, McManamon C, Hanrahan JP, Copley MP, Holmes JD, Morris MA (2011) Development of chemically engineered porous metal oxides for phosphate removal. J Hazard Mater 185(1):382–391. https://doi.org/10.1016/j.jhazmat.2010.08.128
Dogan M, Alkan M, Turkyilmaz A, Ozdemir Y (2004) Kinetics and mechanism of removal of methylene blue by adsorption onto perlite. J Hazard Mater 109(1–3):141–148. https://doi.org/10.1016/j.jhazmat.2004.03.003
Eberhardt TL, Min S, Han JS (2006) Phosphate removal by refined aspen wood fiber treated with carboxymethyl cellulose and ferrous chloride. Bioresour Technol 97(18):2371–2376. https://doi.org/10.1016/j.biortech.2005.10.040
Guzman L, Gunawan G, Viswanathan T (2012) Removal of phosphorus from contaminated wastewater using an iron-containing quaternized wood nanocomposite. Int J Green Nanotechnol 4(3):207–214. https://doi.org/10.1080/19430892.2012.706005
Hebeish A, Higazy A, El-Shafei A, Sharaf S (2010) Synthesis of carboxymethyl cellulose (CMC) and starch-based hybrids and their applications in flocculation and sizing. Carbohydr Polym 79(1):60–69. https://doi.org/10.1016/j.carbpol.2009.07.022
Ho YS, McKay G (1999) Pseudo-second order model for sorption processes. Process Biochem 34(5):451–465. https://doi.org/10.1016/S0032-9592(98)00112-5
Jiang J, Graham NJD (1998) Pre-polymerized inorganic coagulants and phosphorus removal by coagulation: a review. Water SA 24(3):237–244
Juang R, Chen M (1997) Application of the Elovich equation to the kinetics of metal sorption with solvent-impregnated resins. Ind Eng Chem Res 36(3):813–820. https://doi.org/10.1021/IE960351F
Kim J, Mann J, Kwon S (2006) Enhanced adsorption and regeneration with lignocellulose-based phosphorus removal media using molecular coating nanotechnology. J Environ Sci Health, Part A 41(1):87–100. https://doi.org/10.1080/10934520500299570
Kumar KV (2006) Linear and nonlinear regression analysis for the sorption kinetics of methylene blue onto activated carbon. J Hazard Mater 137(3):1538–1544. https://doi.org/10.1016/j.jhazmat.2006.04.036
Mane VS, Mall ID, Srivastava VC (2007) Kinetic and equilibrium isotherm studies for the adsorptive removal of brilliant green dye from aqueous solution by rice husk ash. J Environ Manag 84(4):390–400. https://doi.org/10.1016/j.jenvman.2006.06.024
Mwabonje ON, Jiang J (2010) A trial of using solvent extraction for phosphorus recovery. J Water Resour Protect 2(9):830–838. https://doi.org/10.4236/jwarp.2010.29098
Parsons SA, Smith JA (2008) Phosphorus removal and recovery from municipal wastewaters. Elements 4(2):109–112. https://doi.org/10.2113/GSELEMENTS.4.2.109
Ramasahayam SK, Gunawan G, Finlay C, Viswanathan T (2012) Renewable resource-based magnetic nanocomposites for removal and recovery of phosphorus from contaminated waters. Water Air Soil Pollut 223(8):4853–4863. https://doi.org/10.1007/s11270-012-1241-2
Robalds A, Dreijalte L, Bikovens O, Klavins M (2016) A novel peat-based biosorbent for the removal of phosphate from synthetic and real wastewater and possible utilization of spent sorbent in land application. Desalin Water Treat 57(28):13285–13294. https://doi.org/10.1080/19443994.2015.1061450
Sengupta S, Pandit A (2011) Selective removal of phosphorus from wastewater combined with its recovery as a solid-phase fertilizer. Water Res 45(11):3318–3330. https://doi.org/10.1016/j.watres.2011.03.044
Smith VH (2003) Eutrophication of freshwater and coastal marine ecosystems: a global problem. Environ Sci Pollut Res Int 10(2):126–139. https://doi.org/10.1065/espr2002.12.142
Vijayakumar G, Tamilarasan R, Dharmendirakumar M (2012) Adsorption, kinetic, equilibrium and thermodynamic studies on the removal of basic dye rhodamine-B from aqueous solution by the use of natural adsorbent perlite. J Mater Environ Sci 3(1):157–170
Viswanathan T (2010) US8647512
Viswanathan T (2012) US8574337
Viswanathan T (2013) US9095837
Viswanathan T (2014) US8790615
Yeoman S, Stephenson T, Lester JN, Perry R (1988) The removal of phosphorus during wastewater treatment: a review. Environ Pollut 49(3):183–233. https://doi.org/10.1016/0269-7491(88)90209-6
Zhang D, Wei S, Kaila C, Su X, Wu J, Karki AB, Young DP, Guo Z (2010) Carbon-stabilized iron nanoparticles for environmental remediation. Nanoscale 2(6):917–919. https://doi.org/10.1039/c0nr00065e
Acknowledgement
The authors wish to thank Dr. Fumiya Watanabe and Dr. Shawn Bourdo, Center for Integrative Nanotechnology Sciences (CINS) at the University of Arkansas at Little Rock for SEM and XPS studies. We would also like to acknowledge AbTech Industries, Inc., for financial support to this current work and EPA SBIR program for funding Phase I (EPA Contract Number: EPD12027) and Phase II (EPA Contract Number: EPD13044) projects for the development by Synanomet, LLC., of the media used in this study.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editorial responsibility: M. Abbaspour.
Rights and permissions
About this article
Cite this article
Nakarmi, A., Kim, J., Toland, A. et al. Novel reusable renewable resource-based iron oxides nanocomposites for removal and recovery of phosphate from contaminated waters. Int. J. Environ. Sci. Technol. 16, 4293–4302 (2019). https://doi.org/10.1007/s13762-018-2058-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-018-2058-3