Abstract
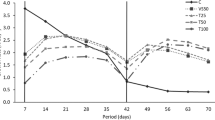
Biodegradation of crude oil hydrocarbon by microorganisms in seawater is generally slow because of the harsh environmental condition due to high salinity. The aim of this study was to compare sawdust (SD) and oil palm empty fruit bunch wastes as suitable carrier material to immobilise hydrocarbon-degrading bacterial consortium culture to accelerate and improve crude oil degradation in seawater. The consortium culture was found able to tolerate salinity up to 3 %, where the degradation of crude oil was not inhibited (p > 0.05). In artificial seawater, suspension of bacterial consortium culture was able to degrade 83.3 ± 3.00 % of crude oil within 8 weeks, which indicated the possibility of using consortium culture in seawater. When tested in seawater, suspension of consortium culture managed to degrade 47.7 ± 1.53 % of crude oil in 8 weeks. In order to improve the performance of consortium culture, immobilisation of consortium culture onto SD and oil palm empty fruit bunch was successfully undertaken when formation of biofilm layers was observed under scanning electron microscope. Immobilising consortium culture onto oil palm empty fruit bunch and SD was shown to increase crude oil biodegradation to 68.7 ± 4.04 and 62.3 ± 5.51 % in 8 weeks, respectively. This study demonstrated immobilisation of consortium culture onto SD and oil palm empty fruit bunch can be utilised as ready-to-use seeds to improve and accelerate crude oil biodegradation in seawater.





Similar content being viewed by others
References
Abdulkarim SM, Fatimah AB, Anderson JG (2009) Effect of salt concentrations on the growth of heat-stressed and unstressed Escherichia coli. J Food Agric Environ 7:51–54
Asker MMS, Shawky BT (2010) Structural characterization and antioxidant activity of an extracellular polysaccharide isolated from Brevibacterium otitidis BTS 44. Food Chem 123:315–320
Atlas RM (1981) Microbial degradation of petroleum hydrocarbons: an environmental perspective. Microb Rev 45:180–209
Atlas RM (1995) Petroleum biodegradation and oil spill bioremediation. Mar Pollut Bull 31:178–182
Austin B (1993) Microbiological methods: marine microbiology. Cambridge University Press, Cambridge, p 24
Bao M, Wang L, Sun P, Cao L, Zou J, Li Y (2012) Biodegradation of crude oil using an efficient microbial consortium in a simulated marine environment. Mar Pollut Bull 64:1177–1185
Bertrand JC, Bianchi M, Mallah MA, Acquaviva M, Mille G (1993) Hydrocarbon biodegradation and hydrocarbon clastic bacterial communities composition grown in seawater as a function of sodium chloride concentration. J Exp Mar Biol 168:125–138
Bishop PL (1983) Marine pollution and its control. McGraw-Hill Book Company, New York
Csonka NL (1989) Physiological and genetic responses of bacteria to osmotic stress. Microb Rev 53:121–147
Díaz MP, Boyd KG, Grigson SJW, Burgess JG (2002) Biodegradation of crude oil across a wide range of salinities by an extremely halotolerant bacterial consortium MPD-M, immobilized onto polypropylene fibers. Biotechnol Bioeng 79:145–153
Etoumi A (2007) Microbial treatment of waxy crude oils for mitigation of wax precipitation. J Petrol Sci Eng 55:111–121
Fellie EA, Sannasi P, Wong KK, Salmijah S, Kader J (2012) Tolerance and biodegradation of benzene, toluene, ethylbenzene and xylene (BTEX) by a metal acclimatized bacterial consortium culture. Res J Biotechnol 7:52–58
Gentili AR, Cubitto MA, Ferrero M, Rodriguez MS (2006) Bioremediation of crude oil polluted seawater by a hydrocarbon degrading bacterial strain immobilised on chitin and chitosan flakes. Int Biodeter Biodegrad 57:222–228
Hamzah A, Chia-Wei P, Nur Faizah AB, Kok-Kee W (2013) Biodegradation of crude oil by constructed bacterial consortia and the constituent single bacteria isolated from Malaysia. Bioremediat J 17:1–10
Harayama S, Kishira H, Kasai Y, Shutsubo K (1999) Petroleum biodegradation in marine environment. J Mol Microbiol Biotechnol 1(1):63–70
Hazaimeh M, Sahilah AM, Palsan SA, Wong KK, Salmijah S (2014) Enhanced crude oil hydrocarbon degradation by self-immobilized bacterial consortium culture on sawdust and oil palm empty fruit bunch. Ann Microbiol. doi:10.1007/s13213-014-0821-3
International Standard ISO 9377-2 (2000) Water quality-determination of hydrocarbon oil index. Part 2: method using solvent extraction and gas chromatography. http://www.norden.org/en/publications/publikationer/2003-516/at_download/publicationfile/. Accessed 16 Dec 2013
International Tanker Owners Pollution Federation Limited (ITOPF) (2011) Use of dispersants to treat oil spills. http://www.itopf.com/spill-response/clean-up-and-response/. Accessed 16 Oct 2012
Ko SH, Lebeault JM (1999) Effect of a mixed culture on cooxidation during the degradation of saturated hydrocarbon mixture. J Appl Microbiol 87:72–79
Kok-Kee W, Brid Q, Salmijah S (2013) Degradation of crude oil in the presence of lead (Pb) and cadmium (Cd) by a metal-adapted consortium culture. Adv Environ Biol 7:577–585
Ławniczak L, Kaczorek E, Olszanowski A (2010) The influence of cell immobilization by biofilm forming on the biodegradation capabilities of bacterial consortia. World J Microbiol Biotechnol. doi:10.1007/s11274-010-0566-5
Liu CW, Chang WN, Liu HS (2009) Bioremediation of n-alkanes and the formation of biofloccules by Rhodococcus erythropolis NTU-1 under various saline conditions and seawater. Biochem Eng J 45:69–75
Lliròs M, Gaju N, de Oyteza TG, Grimalt JO, Esteve I, Martinez-Alonso M (2008) Microcosms experiments of oil degradation by microbial mats. II. The changes in microbial species. Sci Total Environ 393:39–49
Mahjoubi M, Jaouani A, Guesmi A, Ben Amor S, Jouini A, Cherif H, Najjari A, Boudabous A, Koubaa N, Cherif A (2013) Hydrocarbonoclastic bacteria isolated from petroleum contaminated sites in Tunisia: isolation, identification and characterization of the biotechnological potential. New Biotechnol 30(6):723–733
Mielke HW, Wang G, Gonzales CR, Powell ET, Le B, Quach VN (2004) PAHs and metals in the soils of inner-city and suburban New Orleans, Louisiana, USA. Environ Toxicol Pharmacol 18:243–247
Mille G, Almallah M, Bianchi M, Van Wambeke F, Bertran JC (1991) Effect of salinity on petroleum biodegradation. Fresenius J Anal Chem 339:788–791
Minai-Tehrani D, Herfatmanesh A, Azari-Dehkordi F, Minooi S (2006) Effect of salinity on biodegradation of aliphatic fractions of crude oil in soil. Pak J Biol Sci 9:1531–1535
Mumtaz T, Abd-aziz S, Abdul Rahman NA, Phang LY, Shirai Y, Hassan MA (2008) Pilot-scale of recovery of low molecular weight organic acids from anaerobically treated palm oil mill effluent (POME) with energy integrated system. Afr J Biotechnol 21:3900–3905
Obuekwe CO, Al-Muttawa EM (2001) Self-immobilized bacterial cultures with potential for application as ready-to-use seeds for petroleum bioremediation. Biotechnol Lett 23:1025–1032
Podorozhko EA, Lozinsky VI, Ivshina IB, Kuyukina MS, Krivorutchko AB, Philp JC, Cunningham CJ (2008) Hydrophobised sawdust as a carrier for immobilisation of the hydrocarbon-oxidizing bacterium Rhodococcus ruber. Bioresour Technol 99:2001–2008
Rahman SHA, Choudhury JP, Ahmad AL, Kamaruddin AH (2007) Optimization studies on acid hydrolysis of oil palm empty fruit bunch fiber for production of xylose. Bioresour Technol 98:554–559
Sannasi P, Kader J, Othman O, Salmijah S (2009) Physical growth and biomass characterization of bacterial cells exposed to Cd(II), Cr(VI), Ni(II) and Pb(II). J Environ Res Dev 4:8–18
Sathishkumar M, Binupriya AR, Baik S, Yun S (2008) Biodegradation of crude oil by individual bacterial strains and a mixed bacterial consortium isolated from hydrocarbon contaminated areas. Clean 36:92–96
Sorkhoh NA, Ghannoum MA, Ibrahim AS, Stretton RJ, Radwan SS (1990) Crude oil and hydrocarbon-degrading strains of Rhodococcus rhodochrous isolated from soil and marine environments in Kuwait. Environ Pollut 65:1–17
Sutherland IW (2001) Biofilm exopolysaccharides: a strong and sticky framework. Microbiology 147:3–9
Tumaĭkina I, Turkovskaia OV, Ignatov VV (2008) Degradation of hydrocarbons and their derivatives by a microbial association based on Canadian pondweed. Prikl Biokhim Mikrobiol 44:422–429
Viggor S, Juhanson J, Joessar M, Mitt M, Truu J, Vedler E, Heinaru A (2013) Dynamic changes in the structure of microbial communities in Baltic Sea coastal seawater microcosms modified by crude oil, shale oil or diesel fuel. Microbiol Res 168(7):415–427
Vila J, Nieto JM, Mertens J, Springael D, Grifoll M (2010) Microbial community structure of a heavy fuel oil-degrading marine consortium: linking microbial dynamics with polycyclic aromatic hydrocarbon utilization. FEMS Microbiol Ecol 73:349–362
Vreeland RH (1987) Mechanisms of halotolerance in microorganisms. Crit Rev Microbiol 14:311–356
Ward DM, Brock TA (1978) Hydrocarbon biodegradation in hypersaline environments. Appl Environ Microbiol 35:353–359
Wentzel A, Ellingsen TE, Kotlar HK, Zotchev SB, Throne-Holst M (2007) Bacterial metabolism of long-chain n-alkanes. Appl Microbiol Biotechnol 76:1209–1221
Yakimov MM, Timmis KN, Golyshin PN (2007) Obligate oil degrading bacteria. Curr Opin Biotechnol 18:257–266
Zhen-Yu W, Ying X, Hao-Yun W, Jian Z, Dong-Mei G, Feng-Min L, Xing B (2012) Biodegradation of crude oil in contaminated soils by free and immobilized microorganisms. Pedosphere 22:717–725
Acknowledgments
The work described in this paper was supported by Exxon-Mobil STGL-008-2006 and the Malaysian Ministry of Science, Technology and Innovation (MOSTI), Escience Fund 06-01-02-SF0469.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kok Kee, W., Hazaimeh, H., Mutalib, S.A. et al. Self-immobilised bacterial consortium culture as ready-to-use seed for crude oil bioremediation under various saline conditions and seawater. Int. J. Environ. Sci. Technol. 12, 2253–2262 (2015). https://doi.org/10.1007/s13762-014-0619-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-014-0619-7